Chemical and spectroscopy characterization of a compost from food waste applying the hot composting Berkeley method
- Laboratório de Química Biológica do Solo, Departamento de Solos, Instituto de Agronomia, Universidade Federal Rural de Rio de Janeiro (UFRRJ), Seropédica-Rio de Janeiro, Brazil
- Empresa Brasileira de Pesquisa Agropecuária (EMBRAPA), Seropédica-Rio de Janeiro, Brazil

Received: 2020-04-13
Accepted: 2021-07-14
Published in Issue 2022-03-05
Copyright (c) -1 Marco Antonio De Almeida Leal, Marco Antônio Leal, Danielle França De Oliveira, Andrés Calderín García (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
HTML views: 29
PDF views: 395
Abstract
Purpose This study aims to characterize the compost produced under low input conditions in thirty days of composting from food wastes applying the hot composting Berkeley method (HCBM) by 13C NMR CP/MAS techniques combined with evaluation of chemometric, physical and chemical parameters.
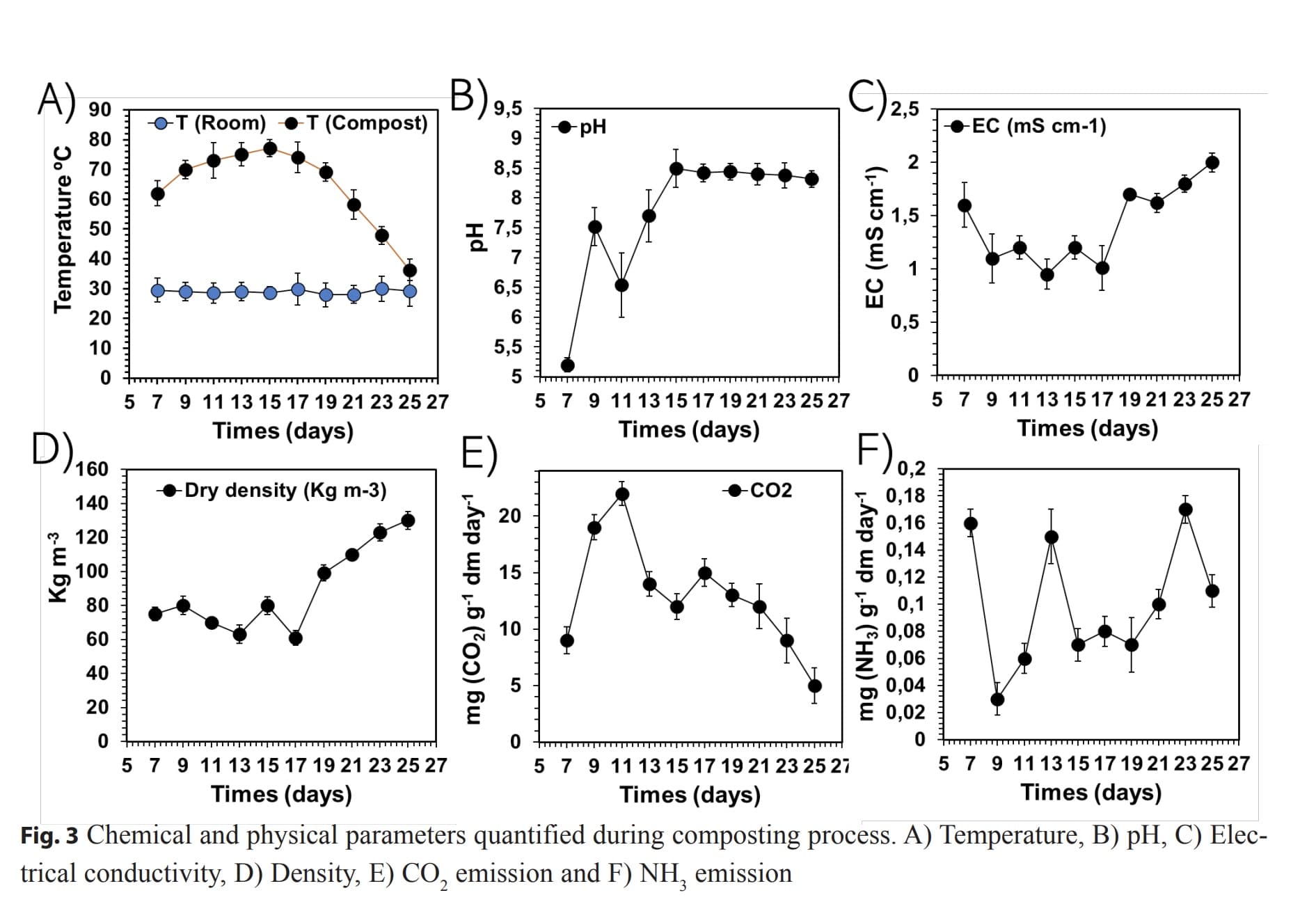
Method The composting process from food waste was carried out following the recommendations of the Hot Composting Berkeley method. At 7, 9, 11, 13, 15, 17, 19, 21, 23 and 25 days turning was carried out, totaling eleven turns and the temperature, pH, electrical conductivity, dry density, potential CO2 emission and potential NH3 emission were quantified. The metal content in the compost was determined after 25 days. The structural modifications of compost process were monitored by 13C-NMR CP MAS spectroscopic and chemometrics analysis.
Results Composting takes place immediately and continues for 15 days through transformation to more labile lipid, carbohydrate and proteinaceous molecules (CAlkyl-H,R; CAlkyl</sub>-O; CAlkyl-O,N) with increasing temperature and pH. After 15 days, there is a CO2 and NH3 emission reduction as well as preservation of recalcitrant molecules with more aromatic and nonpolar characteristics (CAromatic-H,R; CAromatic-O,N).
Conclusion The compost obtained at 25 days of composting by applying HCBM has greater hydrophobicity and aromaticity and is rich in mineral elements important for plant nutrition. The application of HCBM to food waste allowed to obtain a stabilized compost in ecological production conditions.
- Hot composting Berkeley method to food waste allowed to obtain a stabilized compost.
- Composting occurred through an initial transformation of polar and labile compounds.
- After 15 days, the preservation of recalcitrant and molecules occurred.
- The final compost (30 days) was hydrophobic and aromatic.
- Final compost was stabilized and rich in mineral elements (Ca2+, Mg2+, Mn2+, and Zn2+).
Keywords
- Composting,
- Gas emission,
- Humification,
- Spectroscopy

 10.30486/ijrowa.2021.1897439.1052
10.30486/ijrowa.2021.1897439.1052