Human Sperm Cryopreservation Using Polyvinyl Alcohol as a Cryoprotectant
- Department of Biology, Science and Research Branch, Islamic Azad University, Tehran, Iran
- Department of Andrology, Reproductive Biomedicine Research Center, Royan Institute for Reproductive Biomedicine, ACECR, Tehran, Iran
- Department of Animal Science, College of Agriculture, Tarbiat Modares University, Tehran, Iran
- Department of Biology, Damghan Branch, Islamic Azad University of Damghan, Iran

Published in Issue 2024-12-30
Copyright (c) 2024 Atefe Mohsennezhad, Maryam Ghobeh, Mohsen Sharafi, Saeed Zandiyeh, Marjan Sabbaghian (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Abstract
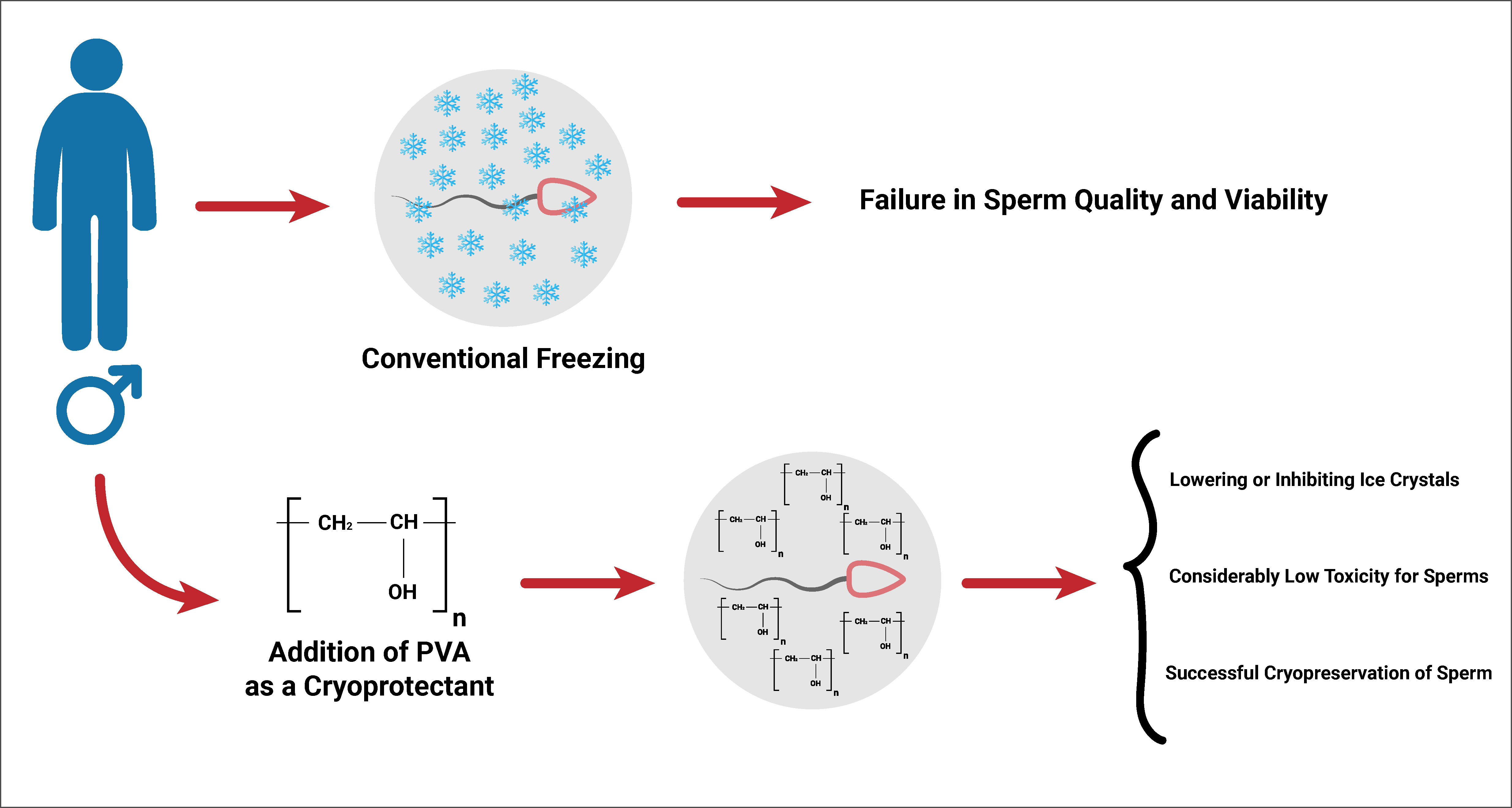
Cryopreservation is a procedure commonly used worldwide to keep a broad range of living cells and tissues, such as sperm, oocytes, and embryos, at extremely low temperatures for long periods while retaining the integrity of their structures. In order to preserve fertility during cancer treatment, sperm cryopreservation has become a common practice. Polyvinyl alcohol (PVA), a synthetic polymer, has shown to prevent the growth of ice crystals and inhibit ice recrystallization at lower concentrations. In the current investigation, the aim was to assess the PVA as a supplement to improve human sperm cryopreservation. Sperm from 21 normospermic males were collected and divided into five equal groups: Fresh, frozen control, 0.01% PVA, 0.1% PVA, and 0.5% PVA. After being frozen and thawed, the main sperm parameters were examined, such as motility, viability, morphology, malondialdehyde (MDA) level, and DNA fragmentation. Based on the results, human sperm motility and viability were increased by adding 0.01% PVA in comparison with the control group. Moreover, different concentrations of PVA did not affect morphology, MDA, or DNA fragmentation. It is reasonable to declare that using an optimal concentration of impermeable ice inhibitors, such as PVA, in conjunction with a permeable cryoprotectant significantly improves the enhancement of human sperm after thawing.
Keywords
- Cryopreservation,
- Polyvinyl alcohol (PVA),
- Human sperm,
- Cryoprotectant
References
- Alipour H. et al. (2017) Improved sperm kinematics in semen samples collected after 2 h versus 4–7 days of ejaculation abstinence. Human Reproduction (7): 1364–1372.
- Bachtiger F. et al. (2021) The atomistic details of the ice recrystallisation inhibition activity of PVA. Nature Communications 12 (1): 1323.
- Badrzadeh H. et al. (2010) Super cool X-1000 and Super cool Z-1000, two ice blockers, and their effect on vitrification/warming of mouse embryos. Eur J Obstet Gynecol Reprod Biol 151 (1): 70–1.
- Bala R. et al. (2021) Environment, Lifestyle, and Female Infertility. Reproductive Sciences 28 (3): 617–638.
- Beirao J. et al. (2012) Improving sperm cryopreservation with antifreeze proteins: effect on gilthead seabream (Sparus aurata) plasma membrane lipids. Biology of Reproduction 86 (2): 1–9.
- Bleszynski M. (2022) The Modification of Polyvinyl Alcohol for Ice Nucleation Based upon the Structures of Antifreeze Glycoproteins Found in Antarctic Fish. Biophysica 2 (4): 417–427.
- Briard J. G. et al. (2016) Small molecule ice recrystallization inhibitors mitigate red blood cell lysis during freezing, transient warming and thawing. 6 (1): 1–10.
- Budke C., Koop T. (2006) Ice recrystallization inhibition and molecular recognition of ice faces by poly(vinyl alcohol). Chemphyschem 7 (12): 2601–6.
- Castro L. et al. (2016) Sperm cryodamage occurs after rapid freezing phase: flow cytometry approach and antioxidant enzymes activity at different stages of cryopreservation. 7 (1): 1–9.
- Congdon T. R., Notman R., Gibson M. I., et al. (2016) Influence of Block Copolymerization on the Antifreeze Protein Mimetic Ice Recrystallization Inhibition Activity of Poly(vinyl alcohol). Biomacromolecules 17 (9): 3033–3039.
- Cooper T. G. et al. (2010) World Health Organization reference values for human semen characteristics. 16 (3) DOI: https://doi.org/231-245.
- Corcini A. S. C.D.V.J., Schiavon R. S., Gastal G. D. A., Goularte K. L., Ulguim R. da R., Bianchi I., Lucia Junior T. (2010) Effect of the polyvinyl alcohol on the cryopreservation of ram spermatozoa.
- PUBVET 4
- Daneshfar Z. et al. (2023) A Qualitative Study exploring the Impact of Infertility on Iranian Women’s Sexual Health. Journal of Midwifery and Reproductive Health 11 (4): 3937–3945.
- Deller R. C. et al. (2014) Synthetic polymers enable non-vitreous cellular cryopreservation by reducing ice crystal growth during thawing. Nature Communications 5 (1): 3244.
- Di Santo M. et al. (2022) Human Sperm Cryopreservation: Update on Techniques, Effect on DNA Integrity, and Implications for ART. Adv Urol 2012:854837.
- Durairajanayagam D. (2018) Lifestyle causes of male infertility. Arab Journal of Urology 16 (1): 10–20.
- Fayter A. E. R. et al. (2020) Ice recrystallisation inhibiting polymers prevent irreversible protein aggregation during solvent-free cryopreservation as additives and as covalent polymer-protein conjugates. European Polymer Journal 140:110036.
- Gonzalez M. et al. (2022) Restoring Sperm Quality Post-Cryopreservation Using Mitochondrial-Targeted Compounds. Antioxidants (Basel) 11 (9)
- Hezavehei M. et al. (2019) Preconditioning of sperm with sublethal nitrosative stress: a novel approach to improve frozen-thawed sperm function Reprod Biomed Online 38 (3): 413–425.
- Hosseinmardi M. et al. (2022) Protective Effect of Cerium Oxide Nanoparticles on Human Sperm Function During Cryopreservation. Biopreserv Biobank 20 (1): 24–30.
- Jaiswal A. N. et al. (2022) Cryopreservation: A Review Article. Cureus 14 (11)
- Jang T. H. et al. (2017) Cryopreservation and its clinical applications. Integr Med Res 6 (1): 12–18.
- Jeyendran R. S. et al. (2008) Cryopreservation of human sperm in a lecithinsupplemented freezing medium. 90 (4): 1263–1265.
- Li Y.-X. et al. (2019) Vitrification and conventional freezing methods in sperm cryopreservation: A systematic review and meta-analysis Eur J Obstet Gynecol Reprod Biol 233:84–92.
- Lusignan M. F. et al. (2018) Effects of different cryopreservation methods on DNA integrity and sperm chromatin quality in men. Andrology 6 (6): 829–835.
- Mitchell D. E. et al. (2015) Gold Nanoparticle Aggregation as a Probe of Antifreeze (Glyco) Protein-Inspired Ice Recrystallization Inhibition and Identification of New IRI Active Macromolecules. Scientific Reports 5 (1): 15716.
- Mohsennezhad A. et al. (2024) Effect of the Supplementation of L-asparticAcid, L-ascorbic Acid, and Silver Nanoparticles (AgNPs) to the In Vitro Fertilization (IVF) Media on the NMRI Mouse IVF Rates.
- Muino R. et al. (2008) Effect of different thawing rates on post-thaw sperm viability, kinematic parameters and motile sperm subpopulations structure of bull semen. 109 (1-4): 50–64.
- Nabeel A. H. T., Jeon Y., Yu I. J. (2019) Use of polyvinyl alcohol as a chemically defined compound in egg yolk-free extender for dog sperm cryopreservation. Reprod Domest Anim 54 (11): 1449–1458.
- Naullage P. M., Molinero V. (2020) Slow Propagation of Ice Binding Limits the Ice-Recrystallization Inhibition Efficiency of PVA and Other Flexible Polymers. Journal of the American Chemical Society 142 (9): 4356–4366.
- Nishijima K. et al. (2014) Effects of type III antifreeze protein on sperm and embryo cryopreservation in rabbit. Cryobiology 69 (1): 22–25.
- Oldenhof H. et al. (2013) Osmotic stress and membrane phase changes during freezing of stallion sperm: mode of action of cryoprotective agents. 88 (3): 1–11.
- Qadeer S. et al. (2014) Evaluation of antifreeze protein III for cryopreservation of Nili-Ravi (Bubalus bubalis) buffalo bull sperm. Animal Reproduction Science 148 (1): 26–31.
- Sabando C. et al. (2022) Preparation of Poly(vinyl Alcohol) Microparticles for Freeze Protection of Sensitive Fruit Crops. Polymers (Basel) 14 (12)
- Salas-Huetos A., Bull ´o M., Salas-Salvad ´o J. (2017) Dietary patterns, foods and nutrients in male fertility parameters and fecundability: a systematic review of observational studies. Human Reproduction Update 23 (4): 371–389.
- Sanger W. G., Olson J. H., Sherman J. K. (1992) Semen cryobanking for men with cancer–criteria change. Fertil Steril 58 (5): 1024–7.
- Tekin K., Das¸ kın A. (2019) Effect of polyvinyl alcohol on survival and function of angora buck spermatozoa following cryopreservation. Cryobiology 89:60–67.
- Walters E. M. et al. (2009) The history of sperm cryopreservation, in Sperm Banking: Theory and Practice. Cambridge University Press: Cambridge.1–17.
- Wowk B. et al. (2000) Vitrification enhancement by synthetic ice blocking agents. Cryobiology 40 (3): 228–36.
- Zandiyeh S. et al. (2020) A novel approach for human sperm cryopreservation with AFPIII. Reproductive Biology 20 (2): 169–174.

 10.57647/pibm.2024.132416
10.57647/pibm.2024.132416