Development and Characterization of ZnO-Loaded Solid Lipid Nanoparticles for Oral Delivery: Physicochemical, Cytocompatibility, and Antibacterial Evaluation
- Department of Chemistry, SR.C., Islamic Azad University, Tehran, Iran
- Department of Chemistry, TeMS.C., Islamic Azad University, Tehran, Iran

Published in Issue 2024-03-30
Copyright (c) 2025 Zahra Inanloo, Mohammad Yousefi, Sahar Baniyaghoob (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Abstract
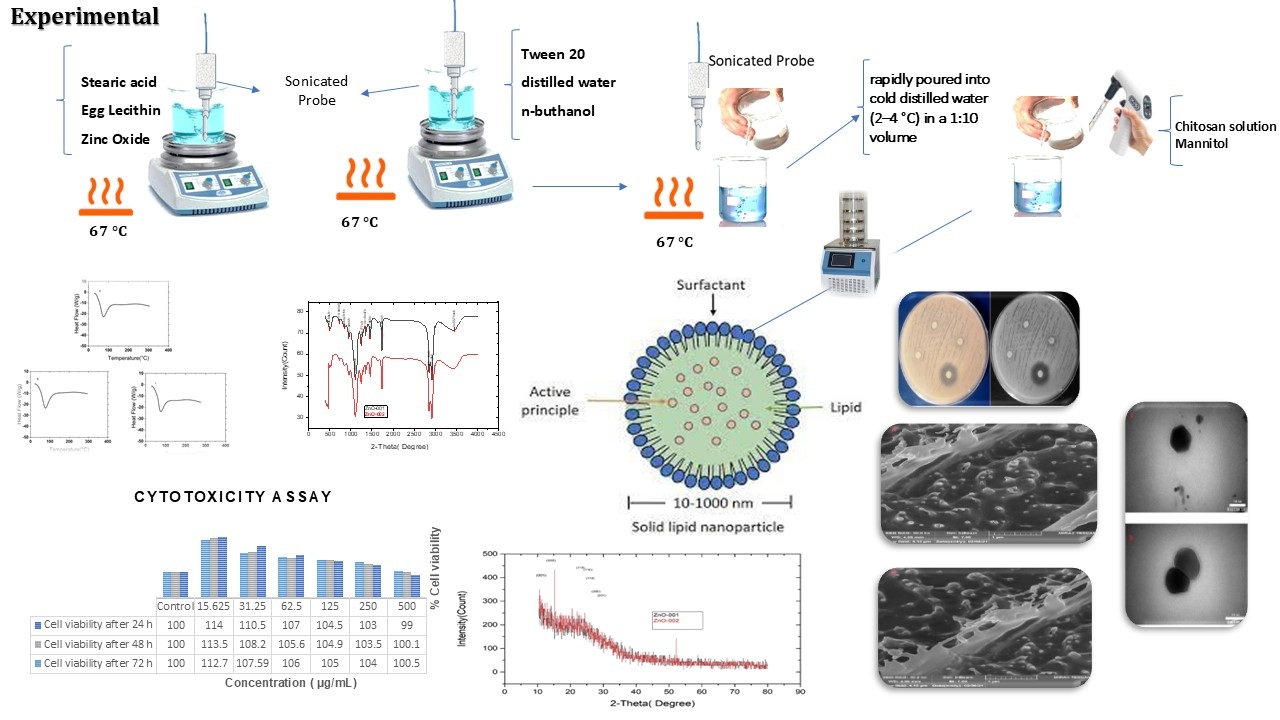
Chitosan-coated solid lipid nanoparticles loaded with zinc oxide (ZnO-CS-SLNs) were developed to enhance oral zinc delivery and improve formulation performance. The nanoparticles were synthesized via a microemulsion technique and exhibited spherical morphology, an average diameter of 91.2 ± 2.8 nm, a positive zeta potential (+25 mV), and high encapsulation efficiency (90 ± 3%), quantified using ICP-OES. FTIR, DSC, and XRD analyses confirmed structural integrity and uniform ZnO distribution within the lipid matrix. FE-SEM and TEM imaging revealed homogeneous particles with no signs of aggregation. In vitro release studies conducted in simulated gastric (pH 1.2) and intestinal (pH 6.8) fluids demonstrated sustained release profiles reaching up to 93% over 24 hours, consistent with Higuchi kinetics. Cytotoxicity assays on AGS cells indicated excellent biocompatibility, with cell viability exceeding 100% across all tested concentrations. Antibacterial activity of ZnO-002 was validated against Gram-positive and Gram-negative strains using disk diffusion, MIC, and MBC assays. Accelerated stability testing over six months (40 ± 2 °C, 75 ± 5% RH) confirmed the physical and colloidal stability of the formulations. Collectively, these results support ZnO-CS-SLNs as a robust oral delivery system with multifunctional attributes and consistent in vitro performance.
Keywords
- Zinc oxide,
- Solid lipid nanoparticles,
- Chitosan,
- Oral delivery,
- Bioavailability,
- Antimicrobial activity,
- Cytocompatibility,
- Sustained release
References
- Prasad AS. Zinc in human health: effect of zinc on immune cells. Curr Opin Clin Nutr Metab Care. 2008;11(6):646-52. doi: 10.1097/MCO.0b013e3283138ab4
- King JC. Zinc: an essential but elusive nutrient. Am J Clin Nutr. 2011;94(2 Suppl):679S-84S. doi: 10.3945/ajcn.110.005744
- Black RE. Trace element undernutrition: biology to interventions. J Nutr. 2003;133(5 Suppl 1):1485S-9S. doi: 10.1093/jn/133.5.1485S
- Brown KH, Wuehler SE, Peerson JM. The importance of zinc in human nutrition and estimation of the global prevalence of zinc deficiency. Food Nutr Bull. 2001;22(2):113-25. doi: 10.1177/156482650102200201
- Trivedi SS, Chudasama RK, Patel N. Effect of zinc supplementation in children with acute diarrhea: randomized double blind controlled trial. Gastroenterol Res. 2009;2(3):137-42. doi: 10.4021/gr2009.06.1297
- Mishra PK, Mishra H, Ekielski A, Talegaonkar S, Vaidya B. Zinc oxide nanoparticles: a promising nanomaterial for biomedical applications. Drug Discov Today. 2017;22(12):1825-34. doi: 10.1016/j.drudis.2017.08.006
- Allen L, de Benoist B, Dary O, Hurrell R, editors. Guidelines on food fortification with micronutrients. Geneva: World Health Organization; 2006
- Salgueiro MJ, Zubillaga MB, Lysionek AE, Caro RA, Weill R, Boccio JR. The role of zinc in the growth and development of children. Nutr Res. 2002;22(7):1005-15. doi: 10.1016/S0271-5317(02)00418-8
- Mehnert W, Mäder K. Solid lipid nanoparticles: production, characterization and applications. Adv Drug Deliv Rev. 2001;47(2-3):165-96. doi: 10.1016/S0169-409X(01)00105-3
- Ekambaram P, Sathali AAH, Priyanka K. Solid lipid nanoparticles: a review. Sci Revs Chem Commun. 2012;2(1):80-102
- Mahajan PS, Mahajan KB, Darekar AB. A review on solid lipid nanoparticle (SLN): an advanced treatment modality. Int J Pharm Sci Res. 2015;6(9):3698-712. doi: 10.13040/IJPSR.0975-8232.6(9).3698-3712
- Müller RH, Radtke M, Wissing SA. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002;54 Suppl 1:S131-55. doi: 10.1016/S0169-409X(02)00118-7
- Bernkop-Schnürch A, Dünnhaupt S. Chitosan-based drug delivery systems. Eur J Pharm Biopharm. 2012;81(3):463-9. doi: 10.1016/j.ejpb.2012.04.007
- Mohammed MA, Syeda JTM, Wasan KM, Wasan EK. An overview of chitosan nanoparticles and its application in non-parenteral drug delivery. Pharmaceutics. 2017;9(4):53. doi: 10.3390/pharmaceutics9040053
- Hosseini SM, Abbasalipourkabir R, Jalili C, Zarei L. Preparation of a nanoliposomal carrier for the oral delivery of zinc and the evaluation of its stability, cytotoxicity, and bioaccessibility. J Food Sci. 2020;85(12):4263-71. doi: 10.1111/1750-3841.15532
- Raghupathi KR, Koodali RT, Manna AC. Size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir. 2011;27(7):4020-8. doi: 10.1021/la104825u
- Pignatello R, Leonardi A, Fuochi V, Petronio GP, Greco AS, Furneri PM. A method for efficient loading of ciprofloxacin hydrochloride in cationic solid lipid nanoparticles: formulation and microbiological evaluation. Nanomaterials (Basel). 2018;8(5):304. doi: 10.3390/nano8050304
- Scioli-Montoto S, Allemann E, Mattos AC, Espina M, Souza J, García ML, et al. Solid lipid nanoparticles for drug delivery: A review of the state of the art. Front Mol Biosci. 2020;7:587997. doi:10.3389/fmolb.2020.587997
- PubChem. 1-Butanol. National Center for Biotechnology Information. PubChem Compound Summary for CID 263. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/1-Butano
- National Toxicology Program. Toxicological profile for 1-Butanol. Toxicol Ind Health. 2008;24(9):597-606. PMID:18830864. doi:10.1177/0748233708098116
- U.S. Food and Drug Administration (FDA). Inactive Ingredient Database (IID). Available from: https://www.fda.gov/drugs/drug-approvals-and-databases/inactive-ingredients-databas
- Date AA, Hanes J, Ensign LM. Nanoparticles for oral delivery: design, evaluation and state-of-the-art. J Control Release. 2016;240:504-26. doi:10.1016/j.jconrel.2016.06.016
- Winter E, Pizzol CD, Locatelli C, Creczynski-Pasa TB. Development and evaluation of lipid nanoparticles for drug delivery: study of toxicity in vitro and in vivo. J Nanosci Nanotechnol. 2016;16(2):1321-30. doi:10.1166/jnn.2016.11676
- Gasco MR. Solid lipid nanospheres from warm microemulsions. Pharm Technol Eur. 1997;9:52-8
- The United States Pharmacopeial Convention. United States Pharmacopeia 42 – National Formulary 37 (USP 42–NF 37). Rockville, MD: USP; 2019
- Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard—tenth edition. CLSI document M07-A10. Wayne, PA: CLSI; 2015
- Müller RH, Eder K, Gohla S. Solid lipid nanoparticles (SLN) for controlled drug delivery: a review of the state of the art. Eur J Pharm Biopharm. 2000;50(1):161-77. doi: 10.1016/S0939-6411(00)00087-4
- Agnihotri SA, Mallikarjuna NN, Aminabhavi TM. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. J Control Release. 2004;100(1):5-28. doi: 10.1016/j.jconrel.2004.08.010
- Luo Y, Teng Z, Li Y, Wang Q. Solid lipid nanoparticles for oral drug delivery: chitosan coating improves stability, controlled delivery, mucoadhesion and cellular uptake. Carbohydr Polym. 2015;122:221-9. doi: 10.1016/j.carbpol.2014.12.084
- Joseph JJ, Sangeetha D, Gomathi T. Sunitinib loaded chitosan nanoparticles formulation and its evaluation. Int J Biol Macromol. 2016;82:952-8. doi: 10.1016/j.ijbiomac.2015.10.077
- Espitia PJP, Soares NFF, dos Reis Coimbra JS, de Andrade NJ, Cruz RS, Medeiros EAA. Zinc oxide nanoparticles: synthesis, antimicrobial activity and food packaging applications. Food Bioprocess Technol. 2012;5(5):1447-64. doi: 10.1007/s11947-012-0797-6
- Ebrahimi HA, Javadzadeh Y, Hamidi M, Jalali MB. Repaglinide-loaded solid lipid nanoparticles: effect of using different surfactants/stabilizers on physicochemical properties of nanoparticles. DARU J Pharm Sci. 2015;23:46. doi:10.1186/s40199-015-0128-3
- Yanti DD, Maryanti E. Green synthesis ZnO nanoparticles using rinds extract of Sapindus rarak DC. J Sci Appl Technol. 2021;5(1):198
- Patel R, Patel M, Patel K. Development and characterization of solid lipid nanoparticles for enhanced oral bioavailability of poorly water-soluble drugs. J Pharm Investig. 2014;44(4):287–302. doi:10.1007/s40005-014-0120-6
- Moezzi A, McDonagh AM, Cortie MB. Zinc oxide particles: synthesis, properties and applications. Chem Eng J. 2012;185–186:1–22. doi:10.1016/j.cej.2012.01.076
- Prokhorov E, Luna-Bárcenas JG, Espinoza-González R, Rentería-Covarrubias N, González-González V, Hernández-Escobar CA, et al. Chitosan-zinc oxide nanocomposites assessed by dielectric, conductivity, mechanical, and piezoelectric properties. Polymers (Basel). 2020;12(9):1991. doi:10.3390/polym12091991
- Selvaraj P, Kalimuthu A, Manjunathan N, Palaniswamy K, Kathirvel D, Rajamani R, et al. Synthesis and characterization of chitosan/zinc oxide nanocomposite for antibacterial activity onto cotton fabrics and dye degradation applications. Int J Biol Macromol. 2020;164:2779–87. doi:10.1016/j.ijbiomac.2020.08.047
- Patel KV, Nath M, Bhatt MD, Dobriyal AK, Bhatt D. Nanofomulation of zinc oxide and chitosan-zinc nano-bioformulation sustain oxidative stress and alter secondary metabolite profile in tobacco. 3 Biotech. 2020;10(11):477. doi:10.1007/s13205-020-02469-x
- Vigani B, Valentino C, Sandri G, Listro R, Fagiani F, Collina S, et al. A composite nanosystem as a potential tool for the local treatment of glioblastoma: chitosan-coated solid lipid nanoparticles embedded in electrospun nanofibers. Polymers (Basel). 2021;13(9):1371. doi:10.3390/polym13091371
- Higuchi T. Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci. 1963;52(12):1145–9. doi:10.1002/jps.2600521210
- Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15(1):25–35. doi:10.1016/0378-5173(83)90064-9
- Mukherjee S, Ray S, Thakur RS. Solid lipid nanoparticles: a modern formulation approach in drug delivery system. Indian J Pharm Sci. 2009;71(4):349–58. doi:10.4103/0250-474X.57282
- Zielińska A, Nowak I, Makuch E, Wójcik M, Carretero C, Souto EB. Optimization of citral-loaded solid lipid nanoparticles (SLN): factorial design and accelerated stability testing. Int J Pharm. 2018;553(1–2):428–40. doi:10.1016/j.ijpharm.2018.10.037
- Zielińska A, Carretero C, Zieliński H, Souto EB. Loading, release profile and accelerated stability assessment of monoterpenes-loaded solid lipid nanoparticles (SLN). J Microencapsul. 2020;37(3):211–21. doi:10.1080/10837450.2020.1744008
- Sastri KT, Radha GV, Pidikiti S, Vajjhala P. Solid lipid nanoparticles: Preparation techniques, their characterization, and an update on recent studies. J Appl Pharm Sci. 2020;10(6):126–41. doi:10.7324/JAPS.2020.10617

 10.57647/pibm.2024.132402
10.57647/pibm.2024.132402