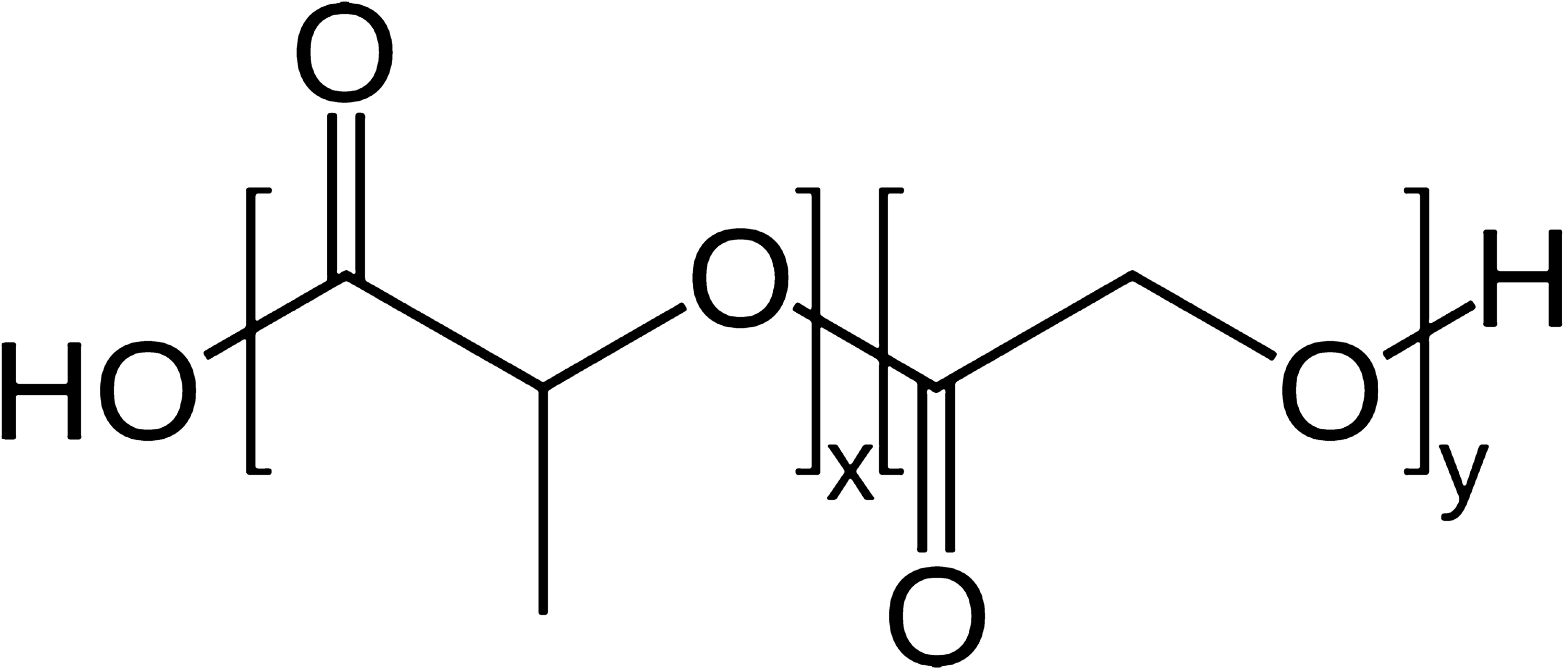
Recent advances in the formulation of PLGA microparticles for controlled drug delivery
- Center for Advanced Biomaterials for HealthCare@CRIB, Istituto Italiano di Tecnologia, Naples, 80125, IT
- Center for Advanced Biomaterials for HealthCare@CRIB, Istituto Italiano di Tecnologia, Naples, 80125, IT Interdisciplinary Research Center of Biomaterials, CRIB, University Federico II, Naples, 80125, IT
- Department of Pharmacy, CIRPEB: Centro Interuniversitario di Ricerca sui Peptidi Bioattivi, University of Naples “Federico II”, Naples, 80134, IT
- Center for Advanced Biomaterials for HealthCare@CRIB, Istituto Italiano di Tecnologia, Naples, 80125, IT Interdisciplinary Research Center of Biomaterials, CRIB, University Federico II, Naples, 80125, IT Department of Chemical, Materials and Industrial Production Engineering (DICMaPI), University of Naples Federico II, Naples, 80125, IT

Published in Issue 2020-10-15
How to Cite
Lagreca, E., Onesto, V., Di Natale, C., La Manna, S., Netti, P. A., & Vecchione, R. (2020). Recent advances in the formulation of PLGA microparticles for controlled drug delivery. Progress in Biomaterials, 9(4 (December 2020). https://doi.org/10.1007/s40204-020-00139-y
Abstract
Abstract Polymeric microparticles (MPs) are recognized as very popular carriers to increase the bioavailability and bio-distribution of both lipophilic and hydrophilic drugs. Among different kinds of polymers, poly-(lactic- co -glycolic acid) (PLGA) is one of the most accepted materials for this purpose, because of its biodegradability (due to the presence of ester linkages that are degraded by hydrolysis in aqueous environments) and safety (PLGA is a Food and Drug Administration (FDA)-approved compound). Moreover, its biodegradability depends on the number of glycolide units present in the structure, indeed, lower glycol content results in an increased degradation time and conversely a higher monomer unit number results in a decreased time. Due to this feature, it is possible to design and fabricate MPs with a programmable and time-controlled drug release. Many approaches and procedures can be used to prepare MPs. The chosen fabrication methodology influences size, stability, entrapment efficiency, and MPs release kinetics. For example, lipophilic drugs as chemotherapeutic agents (doxorubicin), anti-inflammatory non-steroidal (indomethacin), and nutraceuticals (curcumin) were successfully encapsulated in MPs prepared by single emulsion technique, while water-soluble compounds, such as aptamer, peptides and proteins, involved the use of double emulsion systems to provide a hydrophilic compartment and prevent molecular degradation. The purpose of this review is to provide an overview about the preparation and characterization of drug-loaded PLGA MPs obtained by single, double emulsion and microfluidic techniques, and their current applications in the pharmaceutical industry. Graphic abstractKeywords
- PLGA MPs,
- Double emulsion,
- Single emulsion,
- Drug encapsulation,
- Drug release
References
- Ali et al. (2019) Selective anti-ErbB3 aptamer modified sorafenib microparticles: in vitro and in vivo toxicity assessment (pp. 42-53) https://doi.org/10.1016/j.ejpb.2019.10.003
- Alkilani et al. (2015) Transdermal drug delivery: innovative pharmaceutical developments based on disruption of the barrier properties of the stratum corneum 7(4) (pp. 438-470) https://doi.org/10.3390/pharmaceutics7040438
- Allhenn and Lamprecht (2011) Microsphere preparation using the untoxic solvent glycofurol 28(3) (pp. 563-571) https://doi.org/10.1007/s11095-010-0304-6
- Amini-Fazl and Mobedi (2020) Investigation of mathematical models based on diffusion control release for paclitaxel from in-situ forming PLGA microspheres containing HSA microparticles 35(1) (pp. 50-59) https://doi.org/10.1080/10667857.2019.1651549
- Amoyav et al. (2019) Microfluidic based fabrication and characterization of highly porous polymeric microspheres 11(3)
- Angkawinitwong et al. (2020) A novel transdermal protein delivery strategy via electrohydrodynamic coating of PLGA microparticles onto microneedles 12(11) (pp. 12478-12488) https://doi.org/10.1021/acsami.9b22425
- Ansary et al. (2014) Biodegradable poly(d, l-lactic-co-glycolic acid)-based micro/nanoparticles for sustained release of protein drugs—a review 13(7) (pp. 1179-1190)
- Azizi et al. (2020) ChABC-loaded PLGA nanoparticles: a comprehensive study on biocompatibility, functional recovery, and axonal regeneration in animal model of spinal cord injury https://doi.org/10.1016/j.ijpharm.2020.119037
- Bao et al. (2011) Fabrication and characterization of porous poly(lactic-co-glycolic acid) (PLGA) microspheres for use as a drug delivery system 46(8) (pp. 2510-2517)
- Batista et al. (2019) Development and characterization of chitosan microparticles-in-films for buccal delivery of bioactive peptides 12(1)
- Battisti et al. (2019) Non-invasive production of multi-compartmental biodegradable polymer microneedles for controlled intradermal drug release of labile molecules https://doi.org/10.3389/fbioe.2019.00296
- Beck-Broichsitter et al. (2016) Potential of the isolated lung technique for the examination of sildenafil absorption from lung-delivered poly(lactide-co-glycolide) microparticles (pp. 15-20) https://doi.org/10.1016/J.JCONREL.2016.01.057
- Behera and Padhi (2020) Passive and active targeting strategies for the delivery of the camptothecin anticancer drug: a review 18(5) (pp. 1557-1567) https://doi.org/10.1007/s10311-020-01022-9
- Bilati et al. (2005) Nanoprecipitation versus emulsion-based techniques or the encapsulation of proteins into biodegradable nanoparticles and process-related stability issues https://doi.org/10.1208/pt060474
- Biswal et al. (2020) Efficient and prolonged antibacterial activity from porous PLGA microparticles and their application in food preservation https://doi.org/10.1016/J.MSEC.2019.110496
- Brzeziński et al. (2019) Microfluidics for producing polylactide nanoparticles and microparticles and their drug delivery application 68(6) (pp. 997-1014) https://doi.org/10.1002/pi.5753
- Busatto et al. (2018) Effect of particle size, polydispersity and polymer degradation on progesterone release from PLGA microparticles: experimental and mathematical modeling 536(1) (pp. 360-369)
- Calderó et al. (2020) Biomedical perfluorohexane-loaded nanocapsules prepared by low-energy emulsification and selective solvent diffusion
- Capan et al. (2003) Preparation and characterization of poly(d, l-lactide-co-glycolide) micro-spheres for controlled release of human growth hormone 4(2) (pp. 147-156)
- Casalini et al. (2014) Mathematical modeling of PLGA microparticles: from polymer degradation to drug release 11(11) (pp. 4036-4048) https://doi.org/10.1021/mp500078u
- Çetin Altındal and Gümüşderelioğlu (2019) Dual-functional melatonin releasing device loaded with PLGA microparticles and cyclodextrin inclusion complex for osteosarcoma therapy (pp. 586-596) https://doi.org/10.1016/j.jddst.2019.05.027
- Choi and Park (2006) G-CSF loaded biodegradable PLGA nanoparticles prepared by a single oil-in-water emulsion method 311(1–2) (pp. 223-228) https://doi.org/10.1016/j.ijpharm.2005.12.023
- Choi et al. (2011) Development of drug-loaded PLGA microparticles with different release patterns for prolonged drug delivery 32(3) (pp. 867-872)
- Chong et al. (2015) Advances in fabricating double-emulsion droplets and their biomedical applications 19(5) (pp. 1071-1090) https://doi.org/10.1007/s10404-015-1635-8
- Chung et al. (2019) Scalable production of monodisperse functional microspheres by multilayer parallelization of high aspect ratio microfluidic channels 10(9)
- Cocks et al. (2014) Influence of suspension stabilisers on the delivery of protein-loaded porous poly (dl-lactide-co-glycolide) (PLGA) microparticles via pressurised metered dose inhaler (pMDI) 31(8) (pp. 2000-2009) https://doi.org/10.1007/s11095-014-1302-x
- Cocks et al. (2015) Impact of surfactant selection on the formulation and characterization of microparticles for pulmonary drug delivery 41(3) (pp. 522-528) https://doi.org/10.3109/03639045.2014.884117
- Committee for Human Medicinal Products (2018) ICH guideline Q3C (R7) on impurities: guideline for residual solvents
- Cook et al. (2015) A critical evaluation of drug delivery from ligand modified nanoparticles: confounding small molecule distribution and efficacy in the central nervous system (pp. 89-97) https://doi.org/10.1016/j.jconrel.2015.10.013
- Costabile et al. (2018) Physicochemical and in vitro evaluation of drug delivery of an antibacterial synthetic benzophenone in biodegradable PLGA nanoparticles 19(8) (pp. 3561-3570) https://doi.org/10.1208/s12249-018-1187-9
- Cricchio et al. (2017) Novel superparamagnetic microdevices based on magnetized PLGA/PLA microparticles obtained by supercritical fluid emulsion and coating by carboxybetaine-functionalized chitosan allowing the tuneable release of therapeutics 106(8) (pp. 2097-2105)
- da Silva and Pinto (2019) Influence of encapsulated aroma compounds on the formation and morphology of gelatin microparticles 383(1)
- da Silva-Junior et al. (2009) Thermal behavior and stability of biodegradable spray-dried microparticles containing triamcinolone 368(1–2) (pp. 45-55) https://doi.org/10.1016/j.ijpharm.2008.09.054
- Dash et al. (2010) Kinetic modeling on drug release from controlled drug delivery systems 67(3) (pp. 217-223)
- De Alteriis et al. (2015) A method to tune the shape of protein-encapsulated polymeric microspheres 5(1) (pp. 1-9)
- De Rosa et al. (2005) How cyclodextrin incorporation affects the properties of protein-loaded PLGA-based microspheres: the case of insulin/hydroxypropyl-β-cyclodextrin system 102(1) (pp. 71-83) https://doi.org/10.1016/J.JCONREL.2004.09.030
- Di Natale et al. (2018) The level of 24-hydroxycholesteryl esters decreases in plasma of patients with Parkinson’s disease (pp. 108-112) https://doi.org/10.1016/j.neulet.2018.02.041
- Di Natale et al. (2020) Tunable release of curcumin with an in silico-supported approach from mixtures of highly porous PLGA microparticles 13(8) https://doi.org/10.3390/ma13081807
- Ding et al. (2018) Design of mucoadhesive PLGA microparticles for ocular drug delivery 1(3) (pp. 561-571) https://doi.org/10.1021/acsabm.8b00041
- Dong et al. (2019) Development of composite PLGA microspheres containing exenatide-encapsulated lecithin nanoparticles for sustained drug release https://doi.org/10.1016/J.AJPS.2019.01.002
- Dwivedi et al. (2018) Core–shell microencapsulation of curcumin in PLGA microparticles: programmed for application in ovarian cancer therapy 46(sup3) (pp. S481-S491) https://doi.org/10.1080/21691401.2018.1499664
- Esposito et al. (2016) Room temperature consolidation of a porous poly(lactic-co-glycolic acid) matrix by the addition of maltose to the water-in-oil emulsion 9(6) https://doi.org/10.3390/ma9060420
- Fan et al. (2019) Local release of rapamycin by microparticles delays islet rejection within the anterior chamber of the eye 9(1) (pp. 1-9)
- Fenaroli et al. (2018) Enhanced permeability and retention-like extravasation of nanoparticles from the vasculature into tuberculosis granulomas in zebrafish and mouse models 12(8) (pp. 8646-8661) https://doi.org/10.1021/acsnano.8b04433
- Fernández et al. (2012) Controlled release of rasagiline mesylate promotes neuroprotection in a rotenone-induced advanced model of Parkinson’s disease 438(1–2) (pp. 266-278) https://doi.org/10.1016/j.ijpharm.2012.09.024
- Field RW, Bekassy-Molnar E, Lipnizki F, Vatai G (2017) Engineering aspects of membrane separation and application in food processing
- Ford Versypt et al. (2013) Mathematical modeling of drug delivery from autocatalytically degradable PLGA microspheres—a review 165(1) (pp. 29-37) https://doi.org/10.1016/j.jconrel.2012.10.015
- Formiga et al. (2010) Sustained release of VEGF through PLGA microparticles improves vasculogenesis and tissue remodeling in an acute myocardial ischemia–reperfusion model 147(1) (pp. 30-37) https://doi.org/10.1016/j.jconrel.2010.07.097
- Fotticchia et al. (2017) Enhanced drug delivery into cell cytosol via glycoprotein H-derived peptide conjugated nanoemulsions https://doi.org/10.1021/acsnano.7b03058
- Garbayo et al. (2012) Brain drug delivery systems for neurodegenerative disorders 13(12) (pp. 2388-2402) https://doi.org/10.2174/138920112803341761
- Gentile et al. (2016) Localised controlled release of simvastatin from porous chitosan–gelatin scaffolds engrafted with simvastatin loaded PLGA-microparticles for bone tissue engineering application (pp. 249-257) https://doi.org/10.1016/J.MSEC.2015.10.014
- Grizić and Lamprecht (2018) Microparticle preparation by a propylene carbonate emulsification-extraction method 544(1) (pp. 213-221) https://doi.org/10.1016/j.ijpharm.2018.03.062
- Grizić and Lamprecht (2020) Predictability of drug encapsulation and release from propylene carbonate/PLGA microparticles
- Guo et al. (2015) Sustained release donepezil loaded PLGA microspheres for injection: preparation, in vitro and in vivo study 10(5) (pp. 405-414)
- Gupta et al. (2011) PLGA microparticles encapsulating prostaglandin E1-hydroxypropyl-β-cyclodextrin (PGE1-HPβCD) complex for the treatment of pulmonary arterial hypertension (PAH) 28(7) (pp. 1733-1749) https://doi.org/10.1007/s11095-011-0409-6
- Hajavi et al. (2018) Optimization of PLGA formulation containing protein or peptide-based antigen: Recent advances 106(9) (pp. 2540-2551)
- Haji Mansor et al. (2018) Development of a non-toxic and non-denaturing formulation process for encapsulation of SDF-1α into PLGA/PEG-PLGA nanoparticles to achieve sustained release (pp. 38-50) https://doi.org/10.1016/j.ejpb.2017.12.020
- Han et al. (2016) Bioerodable PLGA-based microparticles for producing sustained-release drug formulations and strategies for improving drug loading (pp. 1-11) https://doi.org/10.3389/fphar.2016.00185
- Hasan et al. (2015) Reduction of the in vivo burst release of insulin-loaded microparticles (pp. 486-493) https://doi.org/10.1016/j.jddst.2015.06.020
- He et al. (2019) Designable polymeric microparticles from droplet microfluidics for controlled drug release 4(6) https://doi.org/10.1002/admt.201800687
- Hernández-Giottonini et al. (2020) PLGA nanoparticle preparations by emulsification and nanoprecipitation techniques: Effects of formulation parameters 10(8) (pp. 4218-4231)
- Herrmann (1995) Somatostatin containing biodegradable microspheres prepared by a modified solvent evaporation method based on W/O/W-multiple emulsions 126(1–2) (pp. 129-138) https://doi.org/10.1016/0378-5173(95)04106-0
- Hussain et al. (2017) Regulation of drug release by tuning surface textures of biodegradable polymer microparticles 9(16) (pp. 14391-14400)
- Ito and Makino (2004) Preparation and properties of monodispersed rifampicin-loaded poly(lactide-co-glycolide) microspheres 39(1) (pp. 17-21) https://doi.org/10.1016/j.colsurfb.2004.08.016
- Jafari-Nodoushan et al. (2015) Size and morphology controlling of PLGA microparticles produced by electro hydrodynamic atomization 26(5) (pp. 502-513)
- Jain et al. (2017) Recent advances in lipid-based vesicles and particulate carriers for topical and transdermal application 106(2) (pp. 423-445) https://doi.org/10.1016/J.XPHS.2016.10.001
- Jakhmola et al. (2019) Experimental and theoretical study of biodirected green synthesis of gold nanoflowers https://doi.org/10.1016/J.MTCHEM.2019.100203
- Jamaledin et al. (2020) Progress in microneedle-mediated protein delivery 9(2) https://doi.org/10.3390/jcm9020542
- Jamaledin et al. (2020) Recombinant filamentous bacteriophages encapsulated in biodegradable polymeric microparticles for stimulation of innate and adaptive immune responses 8(5) https://doi.org/10.3390/microorganisms8050650
- Jiang et al. (2014) Targeted oral delivery of BmpB vaccine using porous PLGA microparticles coated with M cell homing peptide-coupled chitosan 35(7) (pp. 2365-2373) https://doi.org/10.1016/j.biomaterials.2013.11.073
- Kamaly et al. (2012) Targeted polymeric therapeutic nanoparticles: design, development and clinical translation 41(7) (pp. 2971-3010) https://doi.org/10.1039/c2cs15344k
- Kang et al. (2014) Applicability of non-halogenated methyl propionate to microencapsulation 31(4) (pp. 323-332) https://doi.org/10.3109/02652048.2013.843729
- Kapoor et al. (2015) PLGA: a unique polymer for drug delivery 6(1) (pp. 41-58)
- Karp et al. (2019) PLGA nano-and microparticles for the controlled release of florfenicol: experimental and theoretical study 136(12)
- Keles et al. (2014) Studying the release of hGH from gamma-irradiated PLGA microparticles using ATR-FTIR imaging (pp. 76-84)
- Keles et al. (2015) Investigation of factors influencing the hydrolytic degradation of single PLGA microparticles (pp. 228-241) https://doi.org/10.1016/j.polymdegradstab.2015.04.025
- Kim et al. (2006) Biodegradable polymeric microspheres with “open/closed” pores for sustained release of human growth hormone 112(2) (pp. 167-174) https://doi.org/10.1016/j.jconrel.2006.02.004
- Kim et al. (2010) Pharmaceutical potential of gelatin as a pH-responsive porogen for manufacturing porous poly(d, l-lactic-co-glycolic acid) microspheres 40(4) (pp. 245-250) https://doi.org/10.4333/kps.2010.40.4.245
- Kim et al. (2011) Highly porous large poly(lactic-co-glycolic acid) microspheres adsorbed with palmityl-acylated exendin-4 as a long-acting inhalation system for treating diabetes 32(6) (pp. 1685-1693) https://doi.org/10.1016/J.BIOMATERIALS.2010.10.045
- Kim et al. (2017) Effect of pore sizes of PLGA scaffolds on mechanical properties and cell behaviour for nucleus pulposus regeneration in vivo 11(1) (pp. 44-57) https://doi.org/10.1002/term.1856
- Kim et al. (2019) Poly(lactic-co-glycolic acid) microparticles in fibrin glue for local, sustained delivery of bupivacaine (pp. 86-92) https://doi.org/10.1016/J.JIEC.2019.02.028
- Kim et al. (2019) Improved drug loading and sustained release of entecavir-loaded PLGA microsphere prepared by spray drying technique 40(4) (pp. 306-312)
- Klose et al. (2006) How porosity and size affect the drug release mechanisms from PLGA-based microparticles 314(2) (pp. 198-206) https://doi.org/10.1016/j.ijpharm.2005.07.031
- Koushik and Kompella (2004) Preparation of large porous deslorelin-PLGA microparticles with reduced residual solvent and cellular uptake using a supercritical carbon dioxide process 21(3) (pp. 524-535) https://doi.org/10.1023/B:PHAM.0000019308.25479.a4
- Larrañeta et al. (2016) Microneedles: a new frontier in nanomedicine delivery 33(5) (pp. 1055-1073) https://doi.org/10.1007/s11095-016-1885-5
- Lee et al. (2007) Protein release behavior from porous microparticle with lysozyme/hyaluronate ionic complex 55(1) (pp. 125-130) https://doi.org/10.1016/j.colsurfb.2006.11.024
- Lee et al. (2010) Facile control of porous structures of polymer microspheres using an osmotic agent for pulmonary delivery 146(1) (pp. 61-67) https://doi.org/10.1016/J.JCONREL.2010.05.026
- Li et al. (2016) Surface-functionalized, pH-responsive poly(lactic-co-glycolic acid)-based microparticles for intranasal vaccine delivery: effect of surface modification with chitosan and mannan (pp. 24-34) https://doi.org/10.1016/j.ejpb.2016.08.012
- Li et al. (2017) Composite core-shell microparticles from microfluidics for synergistic drug delivery 60(6) (pp. 543-553) https://doi.org/10.1007/s40843-016-5151-6
- Li et al. (2018) Preparation of poly(lactide-co-glycolide) microspheres and evaluation of pharmacokinetics and tissue distribution of BDMC-PLGA-MS in rats 13(1) (pp. 82-90) https://doi.org/10.1016/J.AJPS.2017.09.002
- Liang et al. (2011) Improved therapeutic effect of folate-decorated PLGA-PEG nanoparticles for endometrial carcinoma 19(13) (pp. 4057-4066) https://doi.org/10.1016/j.bmc.2011.05.016
- Ma (2014) Microencapsulation of protein drugs for drug delivery: strategy, preparation, and applications (pp. 324-340)
- Maeda and Khatami (2018) Analyses of repeated failures in cancer therapy for solid tumors: poor tumor-selective drug delivery, low therapeutic efficacy and unsustainable costs 7(1) https://doi.org/10.1186/s40169-018-0185-6
- Makadia and Siegel (2011) Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier 3(3) (pp. 1377-1397)
- Mao et al. (2007) Effect of WOW process parameters on morphology and burst release of FITC-dextran loaded PLGA microspheres 334(1) (pp. 137-148) https://doi.org/10.1016/j.ijpharm.2006.10.036
- Martins et al. (2018) Functionalizing PLGA and PLGA derivatives for drug delivery and tissue regeneration applications 7(1)
- Mazzara et al. (2019) Self-healing encapsulation and controlled release of vaccine antigens from PLGA microparticles delivered by microneedle patches 4(1) (pp. 116-128) https://doi.org/10.1002/btm2.10103
- McKiernan et al. (2018) Knockdown of gene expression in macrophages by microRNA mimic-containing poly (lactic-co-glycolic acid) microparticles 5(4)
- Montazeri et al. (2016) Modification of PDMS to fabricate PLGA microparticles by a double emulsion method in a single microfluidic device 16(14) (pp. 2596-2600) https://doi.org/10.1039/C6LC00437G
- Morales-Cruz et al. (2012) Two-step nanoprecipitation for the production of protein-loaded PLGA nanospheres 2(1) (pp. 79-85) https://doi.org/10.1016/j.rinphs.2012.11.001
- Murzin and Heikkilä (2014) Modeling of drug dissolution kinetics with sigmoidal behavior from ordered mesoporous silica 201(5) (pp. 579-592) https://doi.org/10.1080/00986445.2013.782290
- Mylonaki et al. (2018) Imaging the porous structure in the core of degrading PLGA microparticles: the effect of molecular weight (pp. 231-239) https://doi.org/10.1016/j.jconrel.2018.07.044
- Nasr et al. (2013) Hydrophilic versus hydrophobic porogens for engineering of poly(lactide-co-glycolide) microparticles containing risedronate sodium 18(5) (pp. 1078-1088) https://doi.org/10.3109/10837450.2012.693507
- Ni et al. (2017) Exploring polyvinylpyrrolidone in the engineering of large porous PLGA microparticles via single emulsion method with tunable sustained release in the lung: in vitro and in vivo characterization (pp. 11-22) https://doi.org/10.1016/J.JCONREL.2017.01.023
- Nishimura et al. (2017) Porous PLGA microparticles formed by “one-step” emulsification for pulmonary drug delivery: the surface morphology and the aerodynamic properties (pp. 318-326) https://doi.org/10.1016/j.colsurfb.2017.08.004
- Noviendri et al. (2016) Fabrication of fucoxanthin-loaded microsphere(F-LM) by two steps double-emulsion solvent evaporation method and characterization of fucoxanthin before and after microencapsulation 65(8) (pp. 641-653) https://doi.org/10.5650/jos.ess16018
- Nunes and Duarte (2011) Dense CO2 as a solute, co-solute or co-solvent in particle formation processes: a review 4(11) (pp. 2017-2041) https://doi.org/10.3390/ma4112017
- O’Connor et al. (2019) Inhalable poly(lactic-co-glycolic acid) (PLGA) microparticles encapsulating all-trans-retinoic acid (ATRA) as a host-directed, adjunctive treatment for Mycobacterium tuberculosis infection (pp. 153-165) https://doi.org/10.1016/j.ejpb.2018.10.020
- Oh et al. (2011) Preparation of budesonide-loaded porous PLGA microparticles and their therapeutic efficacy in a murine asthma model 150(1) (pp. 56-62) https://doi.org/10.1016/j.jconrel.2010.11.001
- Osman et al. (2013) Inhalable DNase I microparticles engineered with biologically active excipients 26(6) (pp. 700-709) https://doi.org/10.1016/j.pupt.2013.07.010
- Ospina-Villa et al. (2019) Encapsulation of proteins from Leishmania panamensis into PLGA particles by a single emulsion-solvent evaporation method (pp. 1-7)
- Panyam et al. (2003) Polymer degradation and in vitro release of a model protein from poly(d, l-lactide-co-glycolide) nano- and microparticles 92(1–2) (pp. 173-87)
- Park et al. (2019) Biodegradable micro-sized discoidal polymeric particles for lung-targeted delivery system https://doi.org/10.1016/j.biomaterials.2019.119331
- Park et al. (2019) Injectable, long-acting PLGA formulations: analyzing PLGA and understanding microparticle formation (pp. 125-134) https://doi.org/10.1016/j.jconrel.2019.05.003
- Patel et al. (2012) PEG–PLGA based large porous particles for pulmonary delivery of a highly soluble drug, low molecular weight heparin 162(2) (pp. 310-320)
- Patel et al. (2015) Review on PLGA based solvent induced in-situ forming implant
- Peppas and Narasimhan (2014) Mathematical models in drug delivery: how modeling has shaped the way we design new drug delivery systems (pp. 75-81) https://doi.org/10.1016/J.JCONREL.2014.06.041
- Perry et al. (2011) PRINT: a novel platform toward shape and size specific nanoparticle theranostics 44(10) (pp. 990-998)
- Qodratnama et al. (2015) Formulations for modulation of protein release from large-size PLGA microparticles for tissue engineering (pp. 230-236)
- Rezvantalab and Keshavarz Moraveji (2019) Microfluidic assisted synthesis of PLGA drug delivery systems 9(4) (pp. 2055-2072) https://doi.org/10.1039/C8RA08972H
- Ritger and Peppas (1987) A simple equation for description of solute release I. Fickian and non-Fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs 5(1) (pp. 23-36) https://doi.org/10.1016/0168-3659(87)90034-4
- Rodriguez de Anda et al. (2019) Effects of solvent used for fabrication on drug loading and release kinetics of electrosprayed temozolomide-loaded PLGA microparticles for the treatment of glioblastoma 107(7) (pp. 2317-2324) https://doi.org/10.1002/jbm.b.34324
- Ruoslahti (2017) Tumor penetrating peptides for improved drug delivery (pp. 3-12) https://doi.org/10.1016/j.addr.2016.03.008
- Sah (2000) Ethyl formate—alternative dispersed solvent useful in preparing PLGA microspheres 195(1–2) (pp. 103-113) https://doi.org/10.1016/S0378-5173(99)00379-8
- Sah et al. (1996) A novel method of preparing PLGA microcapsules utilizing methylethyl ketone 13(3) (pp. 360-367) https://doi.org/10.1023/A:1016080123176
- Salem (2008) Comparative study of poly (lactic-co-glycolic acid)-poly ethyleneimine-plasmid DNA microparticles prepared using double emulsion methods https://doi.org/10.1080/02652040701659347
- Samadi et al. (2013) The effect of lauryl capping group on protein release and degradation of poly(d, l-lactic-co-glycolic acid) particles 172(2) (pp. 436-443) https://doi.org/10.1016/j.jconrel.2013.05.034
- Shah et al. (2019) Sustained release of a model water-soluble compound via dry powder aerosolizable acetalated dextran microparticles 24(9) (pp. 1133-1143) https://doi.org/10.1080/10837450.2019.1641727
- Shiehzadeh et al. (2019) Preparation and characterization of a dry powder inhaler composed of PLGA large porous particles encapsulating gentamicin sulfate 9(2) (pp. 255-261) https://doi.org/10.15171/apb.2019.029
- Singhvi et al. (2020) Nanocarriers as potential targeted drug delivery for cancer therapy (pp. 51-88) https://doi.org/10.1007/978-3-030-29207-2_2
- Strobel et al. (2020) Stability of fish oil in calcium alginate microcapsules cross-linked by in situ internal gelation during spray drying 13(2) (pp. 275-287)
- Microfluidic Systems (2020) Dolomite Microfluidics.
- https://www.dolomite-microfluidics.com/
- Takami and Murakami (2014) Unexpected and successful “one-step” formation of porous polymeric particles only by mixing organic solvent and water under “low-energy-input” conditions 30(12) (pp. 3329-3336) https://doi.org/10.1021/la500324j
- Takeuchi et al. (2017) Effects of physicochemical properties of poly(lactide-co-glycolide) on drug release behavior of hydrophobic drug-loaded nanoparticles (pp. 771-778)
- Takeuchi et al. (2018) Effects of l-leucine on PLGA microparticles for pulmonary administration prepared using spray drying: fine particle fraction and phagocytotic ratio of alveolar macrophages (pp. 411-417) https://doi.org/10.1016/J.COLSURFA.2017.10.047
- Tan and Danquah (2012) Drug and protein encapsulation by emulsification: technology enhancement using foam formulations 35(4) (pp. 618-626) https://doi.org/10.1002/ceat.201100358
- Tang et al. (2012) Immunosuppressive activity of size-controlled PEG-PLGA nanoparticles containing encapsulated cyclosporine A (pp. 1-9) https://doi.org/10.1155/2012/896141
- Tomic et al. (2016) Setting accelerated dissolution test for PLGA microspheres containing peptide, investigation of critical parameters affecting drug release rate and mechanism 505(1–2) (pp. 42-51) https://doi.org/10.1016/j.ijpharm.2016.03.048
- Tran et al. (2011) Protein-loaded PLGA-PEG-PLGA microspheres: a tool for cell therapy https://doi.org/10.1016/j.ejps.2011.10.030
- Tran et al. (2012) Preparation of polymeric particles in CO2 medium using non-toxic solvents: formulation and comparisons with a phase separation method 82(3) (pp. 498-507) https://doi.org/10.1016/j.ejpb.2012.08.005
- Tran et al. (2013) Lysozyme encapsulation within PLGA and CaCO3 microparticles using supercritical CO2 medium (pp. 159-169) https://doi.org/10.1016/j.supflu.2013.02.024
- Ungaro et al. (2009) Insulin-loaded PLGA/cyclodextrin large porous particles with improved aerosolization properties: in vivo deposition and hypoglycaemic activity after delivery to rat lungs 135(1) (pp. 25-34) https://doi.org/10.1016/j.jconrel.2008.12.011
- Ungaro et al. (2010) Engineering gas-foamed large porous particles for efficient local delivery of macromolecules to the lung 41(1) (pp. 60-70) https://doi.org/10.1016/j.ejps.2010.05.011
- Van De Weert et al. (2000) Protein instability in poly(lactic-co-glycolic acid) microparticles (pp. 1159-1167)
- Vilos et al. (2015) Preclinical development and in vivo efficacy of ceftiofur-PLGA microparticles https://doi.org/10.1371/journal.pone.0123335
- Vladisavljević et al. (2014) Glass capillary microfluidics for production of monodispersed poly (dl-lactic acid) and polycaprolactone microparticles: experiments and numerical simulations (pp. 163-170) https://doi.org/10.1016/j.jcis.2013.12.002
- Vora et al. (2017) Novel bilayer dissolving microneedle arrays with concentrated PLGA nano-microparticles for targeted intradermal delivery: proof of concept (pp. 93-101) https://doi.org/10.1016/J.JCONREL.2017.10.005
- Wan and Yang (2016) Design of PLGA-based depot delivery systems for biopharmaceuticals prepared by spray drying 498(1–2) (pp. 82-95)
- Wang et al. (2015) Comparative studies on the properties of glycyrrhetinic acid-loaded PLGA microparticles prepared by emulsion and template methods 496(2) (pp. 723-731)
- Wang et al. (2017) Enhanced encapsulation and bioavailability of breviscapine in PLGA microparticles by nanocrystal and water-soluble polymer template techniques (pp. 177-185) https://doi.org/10.1016/j.ejpb.2017.02.021
- Wang et al. (2019) Encapsulation and release of doxycycline from electrospray-generated PLGA microspheres: effect of polymer end groups (pp. 1-9) https://doi.org/10.1016/J.IJPHARM.2019.04.023
- Washington et al. (2018) Monomer sequence in PLGA microparticles: effects on acidic microclimates and in vivo inflammatory response (pp. 259-271) https://doi.org/10.1016/j.actbio.2017.10.043
- Webster et al. (2013) Injectable nanomaterials for drug delivery: carriers, targeting moieties, and therapeutics 84(1) (pp. 1-20) https://doi.org/10.1016/j.ejpb.2012.12.009
- Wischke and Schwendeman (2008) Principles of encapsulating hydrophobic drugs in PLA/PLGA microparticles 364(2) (pp. 298-327)
- Wise DL (2000) Handbook of pharmaceutical controlled release technology. CRC Press, Boca Raton.
- https://doi.org/10.1201/9781482289985
- Wu et al. (2017) Hyaluronic acid coated PLGA nanoparticulate docetaxel effectively targets and suppresses orthotopic human lung cancer (pp. 76-82) https://doi.org/10.1016/j.jconrel.2016.12.024
- Xia et al. (2017) Effect of molecular weight of starch on the properties of cassava starch microspheres prepared in aqueous two-phase system (pp. 334-340) https://doi.org/10.1016/j.carbpol.2017.08.074
- Yang et al. (2007) Tumor necrosis factor alpha blocking peptide loaded PEG-PLGA nanoparticles: preparation and in vitro evaluation 331(1) (pp. 123-132) https://doi.org/10.1016/j.ijpharm.2006.09.015
- Yang et al. (2009) Development of highly porous large PLGA microparticles for pulmonary drug delivery 30(10) (pp. 1947-1953) https://doi.org/10.1016/j.biomaterials.2008.12.044
- Yeo and Park (2004) Control of encapsulation efficiency and initial burst in polymeric microparticle systems 27(1)
- Zhang et al. (2020) Engineering large porous microparticles with tailored porosity and sustained drug release behavior for inhalation (pp. 139-146) https://doi.org/10.1016/j.ejpb.2020.08.021
- Zhu et al. (2019) Microencapsulation of thymol in poly(lactide-co-glycolide) (PLGA): physical and antibacterial properties 12(7) https://doi.org/10.3390/ma12071133

 10.1007/s40204-020-00139-y
10.1007/s40204-020-00139-y