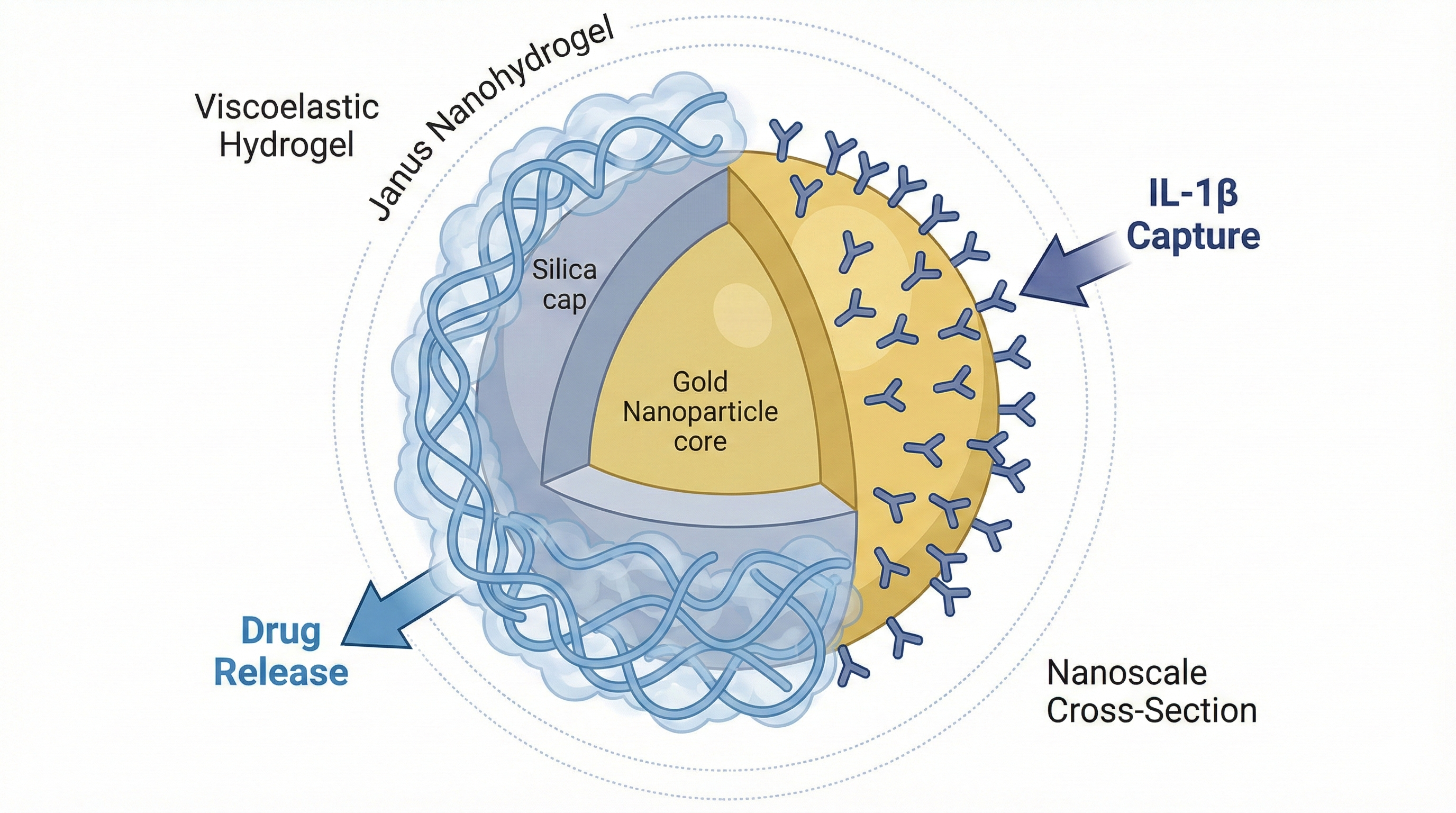
Viscoelastic Janus Nanohydrogels for IL-1β Binding: Synthesis, Structure, and Release Kinetics
- Department of Periodontology, Peking University School and Hospital of Stomatology & National Center for Stomatology & National Clinical Research Center for Oral Diseases & National Engineering Research Center of Oral Biomaterials and Digital Medical Devices, Beijing, 100081, China
- National Research Institute for Family Planning National Human Genetic Resources Center
- School of Medical Technology, Beijing Institute of Technology, Beijing, 100081, China
- First Clinical Division, Peking University School and Hospital of Stomatology & National Center for Stomatology & National Clinical Research Center for Oral Diseases & National Engineering Research Center of Oral Biomaterials and Digital Medical Devices, Beijing, 100034, China

Received: 25-12-2025
Revised: 27-01-2025
Accepted: 14-02-2025
Published in Issue 30-06-2026
Copyright (c) 2026 Lingyan Peng, Guohong Yuan, Guanghao Wu, Xiaohui Yin, Yong Zhang, Xiangying Ouyang, Jing (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 13
Abstract
Interleukin-1β (IL-1β) is a potent pro-inflammatory cytokine central to the pathogenesis of numerous autoinflammatory and chronic inflammatory diseases. Current systemic anti-IL-1β therapies, while effective, are associated with significant side effects, necessitating the development of materials for localized immunomodulation. This study reports the design, synthesis, and comprehensive characterization of a novel, dual-function Viscoelastic Janus Nanohydrogel (VJNH) platform for the simultaneous sequestration of IL-1β and controlled release of an anti-inflammatory therapeutic. VJNHs were synthesized with an asymmetric Au@SiO₂ inorganic core; the gold (Au) face was functionalized with a high-affinity DNA aptamer for IL-1β capture, while the silica (SiO₂) face served as an anchor for a viscoelastic hydrogel shell. This shell was formed via dynamic Schiff base (C=N) chemistry between aldehyde-functionalized hyaluronic acid (oHA) and amine-rich gelatin. Transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS) confirmed the asymmetric Janus morphology and spatially segregated surface chemistry. Rheological analysis demonstrated the ECM-mimetic viscoelastic properties of the network, including high solid-like character (G' > G'') and rapid stress relaxation (relaxation time τ ≈ 120 s), attributed to the dynamic covalent linkages. A comparative study identified an optimal VJNH-2 (1:1 oHA:Gelatin) formulation, which exhibited superior IL-1β binding capacity (≈ 180 ng/mg). Surface plasmon resonance (SPR) analysis revealed high-affinity and specific binding, with an equilibrium dissociation constant (Kₙ) of 1.24 nM. Concurrently, the VJNHs demonstrated sustained, pH-responsive release of a model drug (Dexamethasone), fitting a Fickian diffusion mechanism (Higuchi and Korsmeyer-Peppas models). All VJNH formulations were non-cytotoxic to L929 fibroblasts. These results establish VJNHs as a sophisticated "sense-and-respond" platform, synergizing spatial, mechanical, and chemical functionalities for advanced, localized treatment of inflammatory disorders.
Keywords
- Aptamer,
- Dexamethasone,
- Immunomodulation,
- Periodontitis,
- Schiff Base
References
- Dinarello, C. A. Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol Rev 281, 8–27 (2018).
- Migliorini, P., Italiani, P., Pratesi, F., Puxeddu, I., Boraschi, D. The IL-1 family cytokines and receptors in autoimmune diseases. Autoimmun Rev 19, 102617 (2020).
- Dinarello, C. A. Interleukin-1 in the pathogenesis and treatment of inflammatory diseases. Blood 117, 3720–3732 (2011).
- Scholz, C. C., Cavadas, M. A., Tambuwala, M. M., Hams, E., Rodríguez, J., von Kriegsheim, A., Cotter, P., Bruning, U., Fallon, P. G., Cheong, A., et al. Regulation of IL-1β-induced NF-κB by hydroxylases links key hypoxic and inflammatory signaling pathways. Proc Natl Acad Sci U S A 110, 18490–18495 (2013).
- Jesus, A. A., Goldbach-Mansky, R. IL-1 blockade in autoinflammatory syndromes. Annu Rev Med 65, 223–244 (2014).
- Aggeletopoulou, I., Kalafateli, M., Tsounis, E. P., Triantos, C. Exploring the role of IL-1β in inflammatory bowel disease pathogenesis. Front Med (Lausanne) 11, 1307394 (2024).
- Ren, K., Torres, R. Role of interleukin-1β during pain and inflammation. Brain Res Rev 60, 57–64 (2009).
- Terkeltaub, R., Sundy, J. S., Schumacher, H. R., Murphy, F., Bookbinder, S., Biedermann, S., Wu, R., Mellis, S., Radin, A. The interleukin-1 inhibitor rilonacept in treatment of chronic gouty arthritis. Ann Rheum Dis 68, 1613–1617 (2009).
- Kim, K.-K., Siddiqui, Z., Patel, M., Sarkar, B., Kumar, V. A. A self-assembled peptide hydrogel for cytokine sequestration. J Mater Chem B 8, 945–950 (2020).
- Qin, H., Li, Z., Li, S., Huang, J., Ren, J., Wu, X. Advances in hydrogels for capturing and neutralizing inflammatory cytokines. J Tissue Eng 16, 20417314251342175 (2025).
- Kaur, H., Gogoi, B., Sharma, I., Das, D. K., Azad, M. A., Pramanik, D. D., Pramanik, A. Hydrogels as a potential biomaterial for multimodal therapeutic applications. Mol Pharm 21, 4827–4848 (2024).
- Correa, S., Grosskopf, A. K., Lopez Hernandez, H., Chan, D., Yu, A. C., Stapleton, L. M., Appel, E. A. Translational applications of hydrogels. Chem Rev 121, 11385–11457 (2021).
- Wang, Y., Zhao, P., Zhang, S., Zhu, K., Shangguan, X., Liu, L., Zhang, S. Application of Janus particles in point-of-care testing. Biosensors 12, 689 (2022).
- Le, T. C., Zhai, J., Chiu, W.-H., Tran, P. A., Tran, N. Janus particles: recent advances in biomedical applications. Int J Nanomedicine 14, 6749–6777 (2019).
- Yan, T., Cheng, J., Liu, H., Wang, Y., Zhang, C., Huang, D., Liu, J., Wang, Z. Multifunctional Janus hydrogels: surface design strategies for next-generation clinical solutions. Gels 11, 343 (2025).
- Li, Z.-Y., Li, T.-Y., Yang, H.-C., Ding, M.-H., Chen, L.-J., Yu, S.-Y., Meng, X.-S., Jin, J.-J., Sun, S.-Z., Zhang, J., et al. Design and fabrication of viscoelastic hydrogels as extracellular matrix mimicry for cell engineering. Chem Bio Eng 1, 916–933 (2024).
- Chaudhuri, O., Gu, L., Klumpers, D., Darnell, M., Bencherif, S. A., Weaver, J. C., Huebsch, N., Lee, H., Lippens, E., Duda, G. N. Hydrogels with tunable stress relaxation regulate stem cell fate and activity. Nat Mater 15, 326–334 (2016).
- Liu, B., Chen, K. Advances in hydrogel-based drug delivery systems. Gels 10, 262 (2024).
- Raina, N., Pahwa, R., Bhattacharya, J., Paul, A. K., Nissapatorn, V., Pereira, M. L., Oliveira, S. M. R., Dolma, K. G., Rahmatullah, M., Wilairatana, P., Gupta, M. Drug delivery strategies and biomedical significance of hydrogels. Pharmaceutics 14, 574 (2022).
- Han, Y., Cao, Y., Lei, H. Dynamic covalent hydrogels: strong yet dynamic. Gels 8, 577 (2022).
- Behroozi Kohlan, T., Wen, Y., Mini, C., Finne Wistrand, A. Schiff base crosslinked hyaluronic acid hydrogels with tunable time-dependent mechanical properties. Carbohydr Polym 338, 121122 (2024).
- Carvalho, T., Bártolo, R., Pedro, S. N., Valente, B. F. A., Pinto, R. J. B., Vilela, C., Shahbazi, M.-A., Santos, H. A., Freire, C. S. R. Injectable nanocomposite hydrogels for myocardial regeneration. ACS Appl Mater Interfaces 15, 35612–35628 (2023).
- Hajjar, S., Zhou, X. pH sensing at the intersection of tissue homeostasis and inflammation. Trends Immunol 44, 807–825 (2023).
- Javier, D. J., Nitin, N., Levy, M., Ellington, A., Richards-Kortum, R. Aptamer-targeted gold nanoparticles for reflectance imaging. Bioconjug Chem 19, 1309–1312 (2008).
- Santana Vega, M., Guerrero Martínez, A., Cucinotta, F. Gold@silica hybrid nanoparticles with Janus morphology. Nanomaterials (Basel) 9, 348 (2019).
- Liu, J., Lu, Y. Preparation of aptamer-linked gold nanoparticle aggregates for colorimetric sensing. Nat Protoc 1, 246–252 (2006).
- Weis, M., Shan, J., Kuhlmann, M., Jungst, T., Tessmar, J., Groll, J. Evaluation of oxidized hyaluronic acid hydrogels for bioprinting. Gels 4, 82 (2018).
- Choi, Y., Koh, H. Y., Han, J. Y., Seo, S. Synthesis of hydrogel-based microgels and nanogels. Appl Sci 15, 1368 (2025).
- Kim, J.-T., Lee, D. Y., Kim, Y.-H., Lee, I.-K., Song, Y.-S. Effect of pH on swelling of hyaluronic acid hydrogels. J Sens Sci Technol 21, 256–262 (2012).
- Zhang, F., Zhang, S., Yang, S., Tian, D., Zhang, H., Zhang, Z. Rapid self-healing hyaluronic acid hydrogels via dynamic covalent bonds. ACS Omega 10, 15234–15242 (2025).
- Khatun, M. R., Bhattacharyya, A., Gunbayar, M., Jung, M., Noh, I. Bioresponsive gelatin-hyaluronic acid-genipin hydrogels for bioprinting. Gels 9, 601 (2023).
- Sparks, R. P., Jenkins, J. L., Fratti, R. Surface plasmon resonance for binding kinetics analysis. Methods Mol Biol 1860, 199–210 (2019).
- Bayer, I. S. Controlled drug release from nanoengineered polysaccharides. Pharmaceutics 15, 1364 (2023).
- Wang, J., Huang, D., Ren, H., Shang, L. Biomimetic trained-immunity MSC delivery microcarriers. Small 18, 2200858 (2022).
- Pan, H., Ding, B., Jiang, Z., Wang, J., Li, D., Yu, F., Wang, L., Hu, S., Zhao, Y., Xu, H. Decellularized intestinal submucosa-derived hydrogel for colitis repair. Adv Funct Mater 34, 2405601 (2024).
- Wang, J., Huang, D., Yu, H., Ren, H., Shang, L. Biohybrid response microparticles for liver failure recovery. Adv Healthc Mater 11, 2201085 (2022).
- Lu, Y., Yin, Y., Li, Z.-Y., Xia, Y. Synthesis and self-assembly of Au@SiO₂ core–shell colloids. Nano Lett 2, 785–788 (2002).
- Honciuc, M., Honciuc, A. Scaling amphiphilicity with Janus nanoparticles. Nanomaterials 15, 1079 (2025).
- Jia, X., Xiao, P., Yang, L., Luo, J., He, M., Wang, P., Jiang, B., Xiao, B. Asymmetric Al₂O₃–SiO₂ Janus nanoparticles. Materials (Basel) 17, 1251 (2024).
- Li, J., Wang, L., Benicewicz, B. C. Synthesis of Janus nanoparticles via click chemistry. Langmuir 29, 11547–11553 (2013).
- Kamiya, K., Dohkai, T., Wada, M., Hashimoto, T., Matsuoka, J., Nasu, H. X-ray diffraction of silica gels. J Non-Cryst Solids 240, 202–211 (1998).
- Flores-López, S. L., Dos Santos-Gómez, L., Rey-Raap, N., García-Granda, S., Arenillas, A. High-purity crystalline silicon gels. Acta Crystallogr A 77, C994 (2021).
- Kumar, S., Prasad, L., Bijlwan, P. P., Yadav, A. Thermogravimetric analysis of lignocellulosic composites. Biomass Conv Bioref 14, 12673–12698 (2024).
- Shi, X., Wu, J., Wang, Z., Song, F., Gao, W., Liu, S. Temperature-sensitive PLLA-PDLA hydrogels. RSC Adv 10, 19759–19769 (2020).
- Megeed, Z., Cappello, J., Ghandehari, H. Thermal analysis of silk-elastin hydrogels. Biomacromolecules 5, 793–797 (2004).
- Han, Y., Zhang, L., Yang, W. Mesoporous silica via sol–gel approach. Nanomaterials 14, 903 (2024).
- Dave, P. N., Macwan, P. M., Kamaliya, B. Gum-ghatti-based graphene oxide hydrogels. Mater Adv 4, 2971–2980 (2023).
- Rao, V. S., Srinivas, K., Sujini, G., Kumar, G. S. Protein–protein interaction detection methods. Int J Proteomics 2014, 147648 (2014).
- Van Der Merwe, P. A. Surface plasmon resonance. Protein-Ligand Interactions 1, 137–170 (2001).
- Goh, A. X., Bertin-Maghit, S., Yeo, S. P., Ho, A., Derks, H., Mortellaro, A., Wang, C.-I. Human anti-IL-1β monoclonal antibody. MAbs 6, 764–772 (2014).
- Erikci, S., van den Bergh, N., Boehm, H. Kinetic release from hyaluronan hydrogels. Gels 10, 412 (2024).
- Patroklou, G., Triantafyllopoulou, E., Goula, P.-E., Karali, V., Chountoulesi, M., Valsami, G., Pispas, S., Pippa, N. pH-responsive hydrogels in pharmaceutics. Polymers 17, 1189 (2025).
- Medarametla, R. T., Gopaiah, D. K. V., Shaggir, M., Raghavendra, G., Reddy, D. N., Venkamma, B. Drug release kinetics and mathematical models. J Pharm Anal 27, 102345 (2024).
- Sikora, A., Shard, A. G., Minelli, C. Size and ζ-potential measurement of silica nanoparticles. Langmuir 32, 2216–2224 (2016).
- Pappalardo, R., Boffito, M., Cassino, C., Caccamo, V., Chiono, V., Ciardelli, G. Schiff-base cross-linked hydrogels for drug delivery. ACS Omega 9, 45774–45788 (2024).
- Lin, C.-Y., Yang, C.-M., Lindén, M. Dissolution and morphology evolution of mesoporous silica nanoparticles. J Colloid Interface Sci 608, 995–1004 (2022).

 10.57647/jnsc.2026.1603.14
10.57647/jnsc.2026.1603.14