Cyclodextrin-Linked Corn Starch Nanostructured Microgels as pH-Responsive Nanocarriers for Doxorubicin Delivery in Hepatocellular Carcinoma
- Department of General Medicine, The Fourth Hospital of Hebei Medical University, Shijiazhuang, Hebei, 050000, China
- Department of Oncology, Shijiazhuang First Hospital, Shijiazhuang, Hebei, 050000, China
- Department of Hepatobiliary Surgery, The Fourth Hospital of Hebei Medical University, Shijiazhuang, Hebei, 050000, China

Received: 09-12-2025
Revised: 04-01-2026
Accepted: 03-02-2026
Published in Issue 30-04-2026
Copyright (c) 2026 Shubin Wang, Teng Pan, Jiaqi Zhang, Hao Guo, Wuhan Yang (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 10
Abstract
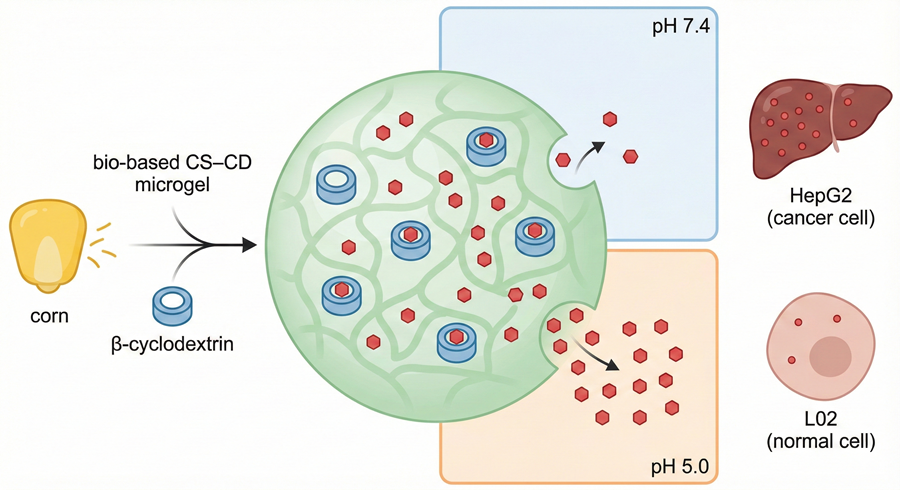
Hepatocellular carcinoma chemotherapy remains constrained by off-target toxicity and multidrug resistance, underscoring the need for biobased nanostructured carriers capable of tumor microenvironment-responsive drug delivery. Here, we report cyclodextrin-linked corn starch nanostructured microgels prepared by inverse water-in-oil emulsion crosslinking with epichlorohydrin as a sustainable platform for doxorubicin delivery. Systematic variation of the β-cyclodextrin content generated colloidally stable microgels with hydrodynamic diameters in the 215–410 nm range, narrow polydispersity indices (~0.22–0.31) and increasingly negative zeta potentials down to −21.5 mV. Nitrogen sorption, FTIR, XRD and thermal analyses confirmed the formation of mesoporous polymer networks, with the lead formulation (CS-CD-3) exhibiting a BET surface area of 12.45 m²/g and an average pore diameter of 9.8 nm. Doxorubicin loading into these microgels afforded high drug payloads with an encapsulation efficiency of 78.4% for CS-CD-3. In vitro release studies demonstrated pronounced pH-dependent behavior, with only ~24% cumulative release at physiological pH 7.4 versus ~72% at pH 5.0 after 24 h, consistent with the intrinsic pH-dependent ionization of doxorubicin and the associated pH-modulated DOX–β-cyclodextrin host–guest interactions within the microgel network. Kinetic fitting of the pH 5.0 release data (Higuchi and Korsmeyer–Peppas models) indicated diffusion through a hydrated matrix with an additional polymer-relaxation contribution under acidic in vitro conditions (n ≈ 0.62). Blank microgels preserved >90% cell viability in both HepG2 and L02 cells, whereas doxorubicin-loaded CS-CD-3 microgels decreased the IC50 in HepG2 cells from 2.15 ± 0.18 to 0.85 ± 0.12 μg/mL and increased the IC50 in L02 cells from 4.50 ± 0.35 to 8.20 ± 0.65 μg/mL, improving the selectivity index from 2.09 to 9.64. Confocal imaging confirmed efficient endocytic uptake, endo-lysosomal release and enhanced apoptotic nuclear damage. These results highlight nanostructured cyclodextrin–starch microgels as a promising bio-derived nanoplatform for safer, more selective hepatocellular carcinoma chemotherapy.
Keywords
- Bio-based Polysaccharide Nanocarriers,
- β-cyclodextrin Crosslinked Microgels,
- Mesoporous Polymer Networks,
- Selective Cancer Chemotherapy Tumor,
- Microenvironment-responsive Release
References
- Reference
- Chan, S.L., Sun, H.-C., Xu, Y., Zeng, H., El-Serag, H.B., Lee, J.M., Schwartz, M.E., Finn, R.S., Seong, J., Wang, X.W., Paradis, V., Abou-Alfa, G.K., Rimassa, L., Kao, J.-H., Zhang, B.-H., Llovet, J.M., Bruix, J., Yip, T.C.-F., Wong, V.W.-S., Wong,G.L.-H., Chan, L.L., Liu, M.-Q., Gao, Q., Shen, F., Kelley, R.K., Cheng, A.-L., Kurosaki, M., Toyoda, H., Chen, W.-X., Murakami, T., Liang, P., Zucman-Rossi, J., Minami, Y., Miyayama, S., Wang, K., Man, K., Hasegawa, K., Li, Q., Tsuchiya, K., Xu, L., Chew, V., Chow, P., Hoshida, Y., Lujambio, A., Ng, I.O.-L., Sakamoto, M., Park, Y.N., Yau, T., Kudo, M., Fan, J., Zhou, J. The Lancet Commission on addressing the global hepatocellular carcinoma burden: comprehensive strategies from prevention to treatment. Lancet 406, 731–778 (2025).
- Pulumati, A., Pulumati, A., Dwarakanath, B.S., Verma, A., Papineni, R.V.L. Technological advancements in cancer diagnostics: improvements and limitations. Cancer Rep. 6, e1764 (2023).
- Anand, U., Dey, A., Chandel, A.K.S., Sanyal, R., Mishra, A., Pandey, D.K., De Falco, V., Upadhyay, A., Kandimalla, R., Chaudhary, A., Dhanjal, J.K., Dewanjee, S., Vallamkondu, J., Pérez de la Lastra, J.M. Cancer chemotherapy and beyond: current status, drug candidates, associated risks and progress in targeted therapeutics. Genes Dis. 10, 1367–1401 (2022).
- Sinha, S.J., Kumar, B., Prasad, C.P., Chauhan, S.S., Kumar, M. Emerging research and future directions on doxorubicin: a snapshot. Asian Pac. J. Cancer Prev. 26, 5–15 (2025).
- Sritharan, S., Sivalingam, N. A comprehensive review on time-tested anticancer drug doxorubicin. Life Sci. 278, 119527 (2021).
- Lim, J.X., Yong, Y.K., Dewi, F.R.P., Chan, S.Y., Lim, V. Nanoscale strategies: doxorubicin resistance challenges and enhancing cancer therapy with advanced nanotechnological approaches. Drug Deliv. Transl. Res. 15, 3835–3863 (2025).
- Kumbhar, D.C.J.R. Stimuli-responsive (smart) drug delivery systems: an in-depth review. (2025).
- Scheffold, F. Pathways and challenges towards a complete characterization of microgels. Nat. Commun. 11, 4315 (2020).
- Aparna, T.N., Kumar, R., Ali, S.R., Patel, D.J., Julekha, K., Begum, T., Bala, J., Kumar, P. Silica nanoparticles: a promising vehicle for anti-cancer drugs delivery. AAPS PharmSciTech 26, 33 (2025).
- Baumann, F., Paul, T., Wassersleben, S., Regenthal, R., Enke, D., Aigner, A. Characterization of drug release from mesoporous SiO₂-based membranes with variable pore structure and geometry. Pharmaceutics 14, 1184 (2022).
- Benalaya, I., Alves, G., Lopes, J., Silva, L.R. A review of natural polysaccharides: sources, characteristics, properties, food, and pharmaceutical applications. Int. J. Mol. Sci. 25, 1322 (2024).
- Jabeen, N., Atif, M. Polysaccharides based biopolymers for biomedical applications: a review. Polym. Adv. Technol. 35, e6203 (2024).
- Behrooznia, Z., Nourmohammadi, J. Polysaccharide-based materials as an eco-friendly alternative in biomedical, environmental, and food packaging. Giant 19, 100301 (2024).
- Yu, J.-K., Moon, Y.-S. Corn starch: quality and quantity improvement for industrial uses. Plants 11, 92 (2022).
- Amaraweera, S.M., Gunathilake, C., Gunawardene, O.H.P., Fernando, N.M.L., Wanninayaka, D.B., Dassanayake, R.S., Rajapaksha, S.M., Manamperi, A., Fernando, C.A.N., Kulatunga, A.K., Manipura, A. Development of starch-based materials using current modification techniques and their applications: a review. Molecules 26, 6880 (2021).
- Li, Y., Zhang, F., Wang, B., Liu, J., Wang, Y., Shi, Z., Du, L., Wang, K., Zhang, W., Wang, Z., Dou, L. Synergistic epichlorohydrin-crosslinked carboxymethyl xylan for enhanced thermal stability and filtration control in water-based drilling fluids. Gels 11, 666 (2025).
- Chen, L., Zhang, D., Wei, L.-F., Zhu, W.-J., Yan, X.-Q., Zhou, R., Din, Z., Ding, W.-P., Ma, T.-Z., Cai, J. Structural and mechanistic insights into starch microgel/anthocyanin complex assembly and controlled release performance. Int. J. Biol. Macromol. 213, 718–727 (2022).
- Xu, W., Li, X., Wang, L., Li, S., Chu, S., Wang, J., Li, Y., Hou, J., Luo, Q., Liu, J. Design of cyclodextrin-based functional systems for biomedical applications. Front. Chem. 9 (2021).
- Zhao, R., Tang, B., Xu, Z., Fang, G. β-Cyclodextrin-based polyelectrolyte complexes for drug delivery. Coord. Chem. Rev. 534, 216581 (2025).
- Nanda, D., Behera, D., Pattnaik, S.S., Behera, A.K. Advances in natural polymer-based hydrogels: synthesis, applications, and future directions in biomedical and environmental fields. Discov. Polym. 2, 6 (2025).
- Nanta, P., Kasemwong, K., Grisdanurak, N. Production and characterization of starch microspheres using supercritical carbon dioxide assisted spray drying. Int. J. Biol. Macromol. 305, 141197 (2025).
- Vagena, I.-A., Malapani, C., Gatou, M.-A., Lagopati, N., Pavlatou, E.A. Enhancement of EPR effect for passive tumor targeting: current status and future perspectives. Appl. Sci. 15, 3189 (2025).
- Hosonuma, M., Yoshimura, K. Association between pH regulation of the tumor microenvironment and immunological state. Front. Oncol. 13, 1175563 (2023).
- Obadi, M., Qi, Y., Xu, B. High-amylose maize starch: structure, properties, modifications and industrial applications. Carbohydr. Polym. 299, 120185 (2023).
- Madrid, F., Rubio-Bellido, M., Morillo, E. Extraction of nonylphenol, pyrene and phenanthrene from sewage sludge and composted biosolids by cyclodextrins and rhamnolipids. Sci. Total Environ. 715, 136986 (2020).
- Krishnan, K.K., Thomas, A.M., Sindhu, K.S., Anilkumar, G. Recent advances and perspectives in the manganese-catalysed epoxidation reactions. Tetrahedron 72, 1–16 (2016).
- Mehryab, F., Ebrahimi, M., Baharvand, H., Haeri, A., Shekari, F. Extracellular vesicle-based formulation of doxorubicin: drug loading optimization, characterization, and cytotoxicity evaluation in tumor spheroids. Pharm. Dev. Technol. 29, 727–737 (2024).
- Lv, Y., He, H., Qi, J., Lu, Y., Zhao, W., Dong, X., Wu, W. Visual validation of the measurement of entrapment efficiency of drug nanocarriers. Int. J. Pharm. 547, 395–403 (2018).
- Oh, Y.J., Hong, J. Application of the MTT-based colorimetric method for evaluating bacterial growth using different solvent systems. LWT 153, 112565 (2022).
- Jonkman, J., Brown, C.M., Wright, G.D., Anderson, K.I., North,A.J. Tutorial: guidance for quantitative confocal microscopy. Nat. Protoc. 15, 1585–1611 (2020).
- Wu, S., Hatahet, T., Bona, B.L., Lodigiani, G., Zhang, M., Bombelli, F.B., Al-Jamal, W.T. Incorporating Span 80 surfactant into lipid nanocapsules improves their biocompatibility and cellular uptake in B16F10 melanoma cells. Int. J. Pharm. 672, 125358 (2025).
- Salleh, K.M., Zakaria, S., Sajab, M.S., Gan, S., Chia, C.H., Jaafar, S.N.S., Amran, U.A. Chemically crosslinked hydrogel and its driving force towards superabsorbent behaviour. Int. J. Biol. Macromol. 118, 1422–1430 (2018).
- Nicu, R., Lisa, G., Darie-Nita, R.N., Avadanei, M.I., Bargan, A., Rusu, D., Ciolacu, D.E. Tailoring the structure and physico-chemical features of cellulose-based hydrogels using multi-epoxy crosslinking agents. Gels 10, 523 (2024).
- Zeng, Y., Zhao, M., Zeng, H., Jiang, Q., Ming, F., Xi, K., Wang, Z., Liang, H. Recent progress in advanced catalysts for electrocatalytic hydrogenation of organics in aqueous conditions. eScience 3, 100156 (2023).
- Vogel, R., Pal, A.K., Jambhrunkar, S., Patel, P., Thakur, S.S., Reátegui, E., Parekh, H.S., Saá, P., Stassinopoulos, A., Broom, M.F. High-resolution single particle zeta potential characterisation of biological nanoparticles using tunable resistive pulse sensing. Sci. Rep. 7, 17479 (2017).
- Greenfield, N.J. Using circular dichroism spectra to estimate protein secondary structure. Nat. Protoc. 1, 2876–2890 (2006).
- Park, S.-H. Cd content dependence of in-plane optical polarization in anisotropically strained c-plane CdZnO/ZnO quantum wells. Physica B 596, 412393 (2020).
- Lim, S.H., Wong, T.W., Tay, W.X. Overcoming colloidal nanoparticle aggregation in biological milieu for cancer therapeutic delivery: perspectives of materials and particle design. Adv. Colloid Interface Sci. 325, 103094 (2024).
- Kypr, J., Kejnovská, I., Renčiuk, D., Vorlíčková, M. Circular dichroism and conformational polymorphism of DNA. Nucleic Acids Res. 37, 1713–1725 (2009).
- Wang, L., Chen, S., Li, C., Gu, Z., Kong, H., Ban, X., Li, Z. Enhancement of β-cyclodextrin production using a glycogen debranching enzyme from Saccharolobus solfataricus STB09. J. Agric. Food Chem. 72, 6491–6499 (2024).
- Yang, X., Yokokura, S., Nagahama, T., Yamaguchi, M., Shimada, T. Molecular dynamics simulation of poly(ether ether ketone) polymer to analyze intermolecular ordering by low wavenumber Raman spectroscopy and X-ray diffraction. Polymers 14, 5406 (2022).
- Li, Z., Pu, L., Hou, D., Yan, J., Zeng, Q., Chen, Y. A macrocycle-assisted platform approach to protein cross-linking via chemically inactive residues. Nano Lett. 25, 3489–3496 (2025).
- Krishnaswamy, S. IR spectrum and characteristic absorption bands. (n.d.). Zhou, J., Zhang, Z., Li, X., Yin, J., Zhou, J., Wu, M. A novel fingerprint recognition strategy driven by convolutional autoencoders and attention mechanisms. in: Proc. 4th Int. Conf. Artificial Intelligence, Robotics, and Communication, 893–900 (2024).
- Glover, J.D., Sudderick, Z.R., Shih, B.B.-J., Batho-Samblas, C., Charlton, L., Krause, A.L., Anderson, C., Riddell, J., Balic, A., Li, J., Klika, V., Woolley, T.E., Gaffney, E.A., Corsinotti, A.,
- Anderson, R.A., Johnston, L.J., Brown, S.J., Wang, S., Chen, Y., Crichton, M.L., Headon, D.J. The developmental basis of fingerprint pattern formation and variation. Cell 186, 940–956.e20 (2023).
- Wang, H., Zhao, P., Ma, R., Jia, J., Fu, Q. Drug–drug co-amorphous systems: an emerging formulation strategy for poorly water-soluble drugs. Drug Discov. Today 29, 103883 (2024).
- Datta, K., van Laar, S.C.W., Taddei, M., Hidalgo, J., Kodalle, T., Aalbers, G.J.W., Lai, B., Li, R., Tamura, N., Frencken, J.T.W., Quiroz Monnens, S.V., Westbrook, R.J.E., Graham, D.J., Sutter-Fella, C.M., Correa-Baena, J.-P., Ginger, D.S., Wienk, M.M., Janssen, R.A.J. Local halide heterogeneity drives surface wrinkling in mixed-halide wide-bandgap perovskites. Nat. Commun. 16, 1967 (2025).
- Rivas-Barbosa, R., Camerin, F., Vialetto, J., Ramakrishna, S.N., Isa, L., Zaccarelli, E. Comparing the structure of microgels at liquid–liquid and solid–liquid interfaces. Langmuir 41, 16674–16684 (2025).
- Gordillo-Galeano, A., Mora-Huertas, C.E. Hydrodynamic diameter and zeta potential of nanostructured lipid carriers: emphasizing some parameters for correct measurements. Colloids Surf. A 620, 126610 (2021).
- Chen, K., Zhang, T., Chen, X., He, Y., Liang, X. Model construction of micro-pores in shale: a case study of Silurian Longmaxi Formation shale in Dianqianbei area, SW China. Pet. Explor. Dev. 45, 412–421 (2018).
- Chai, L., Wang, P., Liu, X., Sun, Y., Li, X., Pan, J. Accurately control the micropore/mesopore ratio to construct a new hierarchical porous carbon with ultrahigh capacitance and rate performance. J. Power Sources 532, 231324 (2022).
- Xu, B., Li, S., Shi, R., Liu, H. Multifunctional mesoporous silica nanoparticles for biomedical applications. Signal Transduct. Target. Ther. 8, 435 (2023).
- He, Y., Zheng, Y., Liu, C., Zhang, H., Shen, J. Citric acid cross-linked β-cyclodextrins: a review of preparation and environmental/biomedical application. Carbohydr. Polym. 323, 121438 (2024).
- Sichina, W.J. Characterization of polymers using TGA. (n.d.). Wu, Z., Chu, C., Jin, Y., Yang, L., Qian, B., Wang, Y., Wang, Y., Wu, J., Jia, Y., Zhang, W., You, Z. Dynamic cross-linked topological network reconciles the longstanding contradictory properties of polymers. Sci. Adv. 11, eadt0825 (2025).
- Mal, A., Prabhuraj, R.S., Malhotra, R., Valvi, S.K., Ingle, A., Srivastava, R., De, A., Bandyopadhyaya, R. pH-responsive sustained delivery of doxorubicin using aminated and PEGylated mesoporous silica nanoparticles leads to enhanced antitumor efficacy in pre-clinical orthotopic breast cancer model. J. Drug Deliv. Sci. Technol. 77, 103800 (2022).
- Tsuchitani, T., Kato, M., Tomaru, A., Aoki, Y., Sugiyama, Y. Trends of in vitro pharmacological potency and in vivo pharmacokinetics parameters of modern drugs: can the therapeutic/subtherapeutic dose be estimated from in vitro Ki and pharmacokinetic parameters? Clin. Transl. Sci. 17, e70034 (2024).
- Kciuk, M., Gielecińska, A., Mujwar, S., Kołat, D., Kałuzińska-Kołat, Ż., Celik, I., Kontek, R. Doxorubicin—an agent with multiple mechanisms of anticancer activity. Cells 12, 659 (2023).
- Zhang, M., Zhang, G., Meng, X., Wang, X., Xie, J., Wang, S., Wang, B., Wang, J., Liu, S., Huang, Q., Yang, X., Li, J., Wang, H. Reduction of the oxidative damage to H₂O₂-induced HepG2 cells via the Nrf2 signalling pathway by plant flavonoids quercetin and hyperoside. Food Sci. Hum. Wellness 13, 1864–1876 (2024).

 10.57647/jnsc.2026.1602.08
10.57647/jnsc.2026.1602.08