Gold Nanoisland Microelectrode Micro-Aptamer Sensor with SWASV Readout for EGFR-Positive Lung Cancer Exosomes
- The First Affiliated Hospital of Nanchang University, Nanchang, Jiangxi, China
- China-Japan Friendship Jiangxi Hospital, National Regional Center for Respiratory Medicine, Nanchang, Jiangxi, China
- Department of Clinical Medicine, South Central MinZu University, Wuhan, Hubei, China
- Department of Pulmonary and Critical Care Medicine, Loudi Central Hospital, Loudi, Hunan, China
- Department of Pathology, Jiangxi Cancer Hospital &Institute, Jiangxi Clinical Research Center for Cancer, The Second Affiliated Hospital of Nanchang Medical College, Nanchang, Jiangxi, China
- Department of Intensive Care Unit, Qingpu District Central Hospital, Shanghai, China Wenxin Yuan and Sicong Jiang contributed equally to this work

Received: 29-10-2025
Revised: 20-11-2025
Accepted: 02-12-2025
Published in Issue 28-02-2026
Copyright (c) 2026 Wenxin Yuan, Sicong Jiang, Chenghong Mao, Jiaru Jiang, Ke Zhang, Dongmei Lin, Jianjun Tang (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 107
Abstract
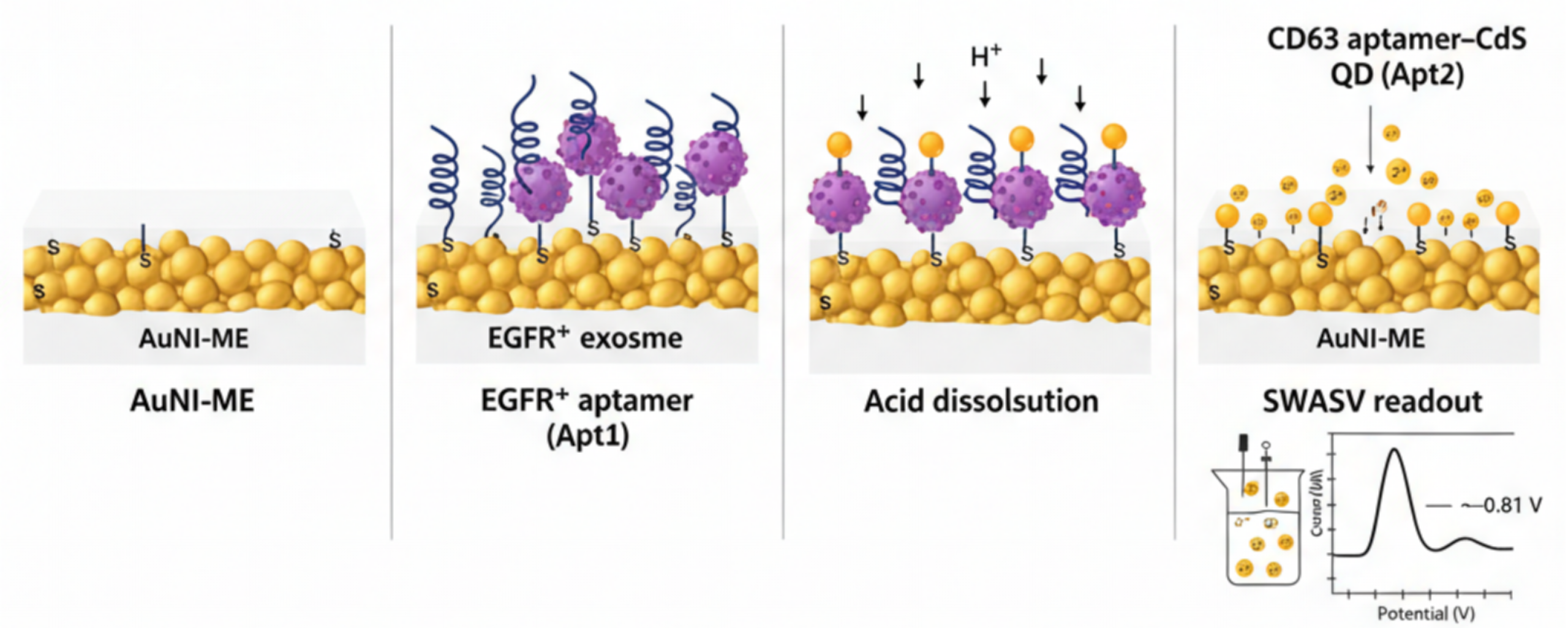
Non-small cell lung cancer (NSCLC) requires highly sensitive liquid biopsy tools capable of detecting EGFR driven disease and resistance mutations such as T790M, which often evade conventional circulating free DNA assays. Exosomes provide a more reliable biomarker due to their abundance, stability, and enrichment of tumor specific EGFR cargo. Here, we report an ultrasensitive electrochemical microaptasensor specifically designed for the quantitative detection of EGFR positive lung cancer exosomes. The innovation of this work lies in the integration of a thermally dewetted gold nanoisland microelectrode (AuNI ME) which offers a large and Au(111) enriched electroactive surface for high density aptamer immobilization. Additionally, the system utilizes a dual aptamer AND gate recognition strategy using EGFR and CD63 aptamers to ensure exceptional specificity along with a cadmium sulfide quantum dot (CdS QD) amplification system quantified via square wave anodic stripping voltammetry (SWASV). This multi tiered amplification architecture combines nanostructured electrode design, orthogonal dual aptamer recognition, and QD based metal ion amplification to result in a remarkably low limit of detection of 150 particles/mL. This performance outperforms ELISA, NTA, and recent electrochemical exosome assays. The sensor exhibits excellent selectivity, reproducibility, and stability, and demonstrates high recovery ranging from 97.8% to 105.0% in spiked human serum samples. These results highlight the translational potential of this platform as a noninvasive tool for early NSCLC diagnosis and real time therapeutic monitoring.
Keywords
- EGFR-positive exosomes,
- Electrochemical aptasensor,
- Gold nanoislands,
- Lung cancer detection,
- Square wave anodic stripping voltammetry
References
- Castellanos-Rizaldos, E., Grimm, D. G., Tadigotla, V., Hurley, J., Healy, J., Neal, P. L., Sher, M., Venkatesan, R., Karlovich, C., Raponi, M. et al. Exosome-based detection of EGFR T790M in plasma from non–small cell lung cancer patients. Clin Cancer Res 24, 2944–2950 (2018).
- Carreca, A. P., Tinnirello, R., Miceli, V., Galvano, A., Gristina, V., Incorvaia, L., Pampalone, M., Taverna, S. and Iannolo, G. Extracellular vesicles in lung cancer: implementation in diagnosis and therapeutic perspectives. Cancers 16, 1967 (2024).
- Zhou, T., Ma, H., Li, Z., Xu, Y. and Zhao, L. Exosomes in lung cancer: a role in early diagnosis. Front Oncol 15, 1599608 (2025).
- Li, X., Corbett, A. L., Taatizadeh, E., Tasnim, N., Little, J. P., Garnis, C., Daugaard, M., Guns, E., Hoorfar, M. and Li, I. T. S. Challenges and opportunities in exosome research: perspectives from biology, engineering, and cancer therapy. APL Bioeng 3, 011503 (2019).
- Imran, M., Shahzadi, I., Haider, A., Hassan, M., Ul-Hamid, A., Safdar, W., Haider, J. and Ikram, M. Catalytic and antibacterial activities with molecular docking analysis of chitosan and polyethylene glycol–NiO₂ nanostructures. J Nanostruct Chem 15, 152520 (2025).
- Xu, Y., Wan, L., Zare, N., Wang, S.-W. and Lei, Z. Cr₂C MXene modification of an electrochemical platform allows for highly selective and sensitive detection of PSMA, a prostate cancer biomarker. J Nanostruct Chem 15, 152517 (2025).
- Stanciu, L. A., Wei, Q., Barui, A. K. and Mohammad, N. Recent advances in aptamer-based biosensors for global health applications. Annu Rev Biomed Eng 23, 433–459 (2021).
- Mutlu, E., Kaya, Ö., Yildirim, A. B. and Çetinkaya, A. Exosome production, isolation and characterization from A549 epithelial carcinoma cells. Hacet J Biol Chem 47, 383–388 (2020).
- Lobb, R. J., Becker, M., Wen Wen, S., Wong, C. S., Wiegmans, A. P., Leimgruber, A. and Möller, A. Optimized exosome isolation protocol for cell culture supernatant and human plasma. J Extracell Vesicles 4, 27031 (2015).
- Chen, F., Mostafiz, B., Suni, J. and Peltola, E. Electrochemical design of gold nanostructures for controllable electrochemical performance and scalable aptamer sensing application. ACS Appl Nano Mater 8, 9812–9823 (2025).
- Švorčík, V., Siegel, J., Šutta, P., Mistrík, J., Janíček, P., Worsch, P. and Kolská, Z. Annealing of gold nanostructures sputtered on glass substrate. Appl Phys A 102, 605–610 (2011).
- Song, R., Liu, Y. and He, L. Synthesis and characterization of mercaptoacetic acid-modified ZnO nanoparticles. Solid State Sci 10, 1563–1567 (2008).
- Hurling, S. Introduction to EDC. New Dir Teach Learn Engl Discuss 1, 2–10 (2012).
- Liu, Y., Canoura, J., Alkhamis, O. and Xiao, Y. Immobilization strategies for enhancing sensitivity of electrochemical aptamer-based sensors. ACS Appl Mater Interfaces 13, 9491–9499 (2021).
- Nooranian, S., Mohammadinejad, A., Mohajeri, T., Aleyaghoob, G. and Kazemi Oskuee, R. Biosensors based on aptamer-conjugated gold nanoparticles: a review. Biotechnol Appl Biochem 69, 1517–1534 (2022).
- Wang, J., Liu, G., Polsky, R. and Merkoçi, A. Electrochemical stripping detection of DNA hybridization based on cadmium sulfide nanoparticle tags. Electrochem Commun 4, 722–726 (2002).
- Fan, F., Dou, J., Ding, A., Zhang, K. and Wang, Y. Determination of lead by square wave anodic stripping voltammetry using an electrochemical sensor. Anal Sci 29, 571–577 (2013).
- Malenica, M., Vukomanović, M., Kurtjak, M., Masciotti, V., Dal Zilio, S., Greco, S., Lazzarino, M., Krušić, V., Perčić, M., Jelovica Badovinac, I. et al. Perspectives of microscopy methods for morphology characterisation of extracellular vesicles from human biofluids. Biomedicines 9, 603 (2021).
- Reclusa, P., Sirera, R., Araujo, A., Giallombardo, M., Valentino, A., Sorber, L., Bazo, I. G., Pauwels, P. and Rolfo, C. Exosomes genetic cargo in lung cancer: a truly Pandora’s box. Transl Lung Cancer Res 5, 483 (2016).
- Bielska, E., Harrison, P. et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles 7, 1535750 (2018).
- Giannopoulos-Dimitriou, A., Saiti, A., Malousi, A., Anagnostopoulos, A. K., Vatsellas, G., Al-Maghrabi, P. M., Müllertz, A., Fatouros, D. G. and Vizirianakis, I. S. Molecular profiling of A549 cell-derived exosomes: proteomic, miRNA, and interactome analysis for identifying potential key regulators in lung cancer. Cancers 16, 4123 (2024).
- Sandua, A., Alegre, E. and Gonzalez, A. Exosomes in lung cancer: actors and heralds of tumor development. Cancers 13, 4330 (2021).
- Liu, W., Howarth, M., Greytak, A. B., Zheng, Y., Nocera, D. G., Ting, A. Y. and Bawendi, M. G. Compact biocompatible quantum dots functionalized for cellular imaging. J Am Chem Soc 130, 1274–1284 (2008).
- Ji, Y., Yang, X., Ji, Z., Zhu, L., Ma, N., Chen, D., Jia, X., Tang, J. and Cao, Y. DFT-calculated IR spectrum amide I, II, and III band contributions of N-methylacetamide fine components. ACS Omega 5, 8572–8578 (2020).
- Badilescu, S., Raju, D., Bathini, S. and Packirisamy, M. Gold nano-island platforms for localized surface plasmon resonance sensing: a short review. Molecules 25, 4661 (2020).
- Potejanasak, P. and Duangchan, S. Gold nanoisland agglomeration upon the substrate assisted chemical etching based on thermal annealing process. Crystals 10, 533 (2020).
- Tesler, A. B., Maoz, B. M., Feldman, Y., Vaskevich, A. and Rubinstein, I. Solid-state thermal dewetting of just-percolated gold films evaporated on glass: development of the morphology and optical properties. J Phys Chem C 117, 11337–11346 (2013).
- Lee, M. R., Lee, H. K., Yang, Y., Koh, C. S. L., Lay, C. L., Lee, Y. H., Phang, I. Y. and Ling, X. Y. Direct metal writing and precise positioning of gold nanoparticles within microfluidic channels for SERS sensing of gaseous analytes. ACS Appl Mater Interfaces 9, 39584–39593 (2017).
- Kim, Y., Park, J., Choi, D., Jang, H., Lee, J., Park, H., Choi, J., Ju, D., Lee, J. and Kim, D. ITO/Au/ITO multilayer thin films for transparent conducting electrode applications. Appl Surf Sci 254, 1524–1527 (2007).
- Yang, G. and Liu, G. New insights for self-assembled monolayers of organothiols on Au(111) revealed by scanning tunneling microscopy. J Phys Chem B 107, 8746–8759 (2003).
- Oberhaus, F. V., Frense, D. and Beckmann, D. Immobilization techniques for aptamers on gold electrodes for the electrochemical detection of proteins: a review. Biosensors 10, 45 (2020).
- Sahoo, S. R. and Ke, S.-C. Spin-orbit coupling effects in Au 4f core-level electronic structures in supported low-dimensional gold nanoparticles. Nanomaterials 11, 554 (2021).
- Mirsaleh-Kohan, N., Bass, A. D. and Sanche, L. X-ray photoelectron spectroscopy analysis of gold surfaces after removal of thiolated DNA oligomers by ultraviolet/ozone treatment. Langmuir 26, 6508–6514 (2010).
- Shon, Y.-S., Aquino, M., Pham, T. V., Rave, D., Ramirez, M., Lin, K., Vaccarello, P., Lopez, G., Gredig, T. and Kwon, C. Stability and morphology of gold nanoisland arrays generated from layer-by-layer assembled nanoparticle multilayer films: effects of heating temperature and particle size. J Phys Chem C 115, 10597–10605 (2011).
- Ma, H., Li, J., Gao, M., Dong, Y., Luo, Y. and Su, S. An electrochemical aptasensor for accurate and sensitive detection of exosomes based on dual-probe recognition and hybridization chain reaction. Biosensors 15, 302 (2025).
- Li, J., Liu, Y., Wang, C., Jia, Q., Zhang, G., Huang, X., Zhou, N. and Zhang, Z. Determination of VEGF165 using impedimetric aptasensor based on cyclohexanehexone-melem covalent-organic framework. Microchim Acta 188, 211 (2021).
- Kayiş, E. Ç., Torul, H., Sazaklıoğlu, S. A., Çelikkan, H., Ensarioğlu, H. K., Gumus, B. H., Vatansever, H. S. and Tamer, U. Electrochemically generated paper SERS substrate for detection of exosome in urine samples. Sens Actuators B Chem 426, 137103 (2025).
- Im, Y., Yoo, H., Ko, R.-E., Lee, J. Y., Park, J. and Jeon, K. Exosomal CD63 in critically ill patients with sepsis. Sci Rep 11, 20300 (2021).
- Wang, C.-K., Tsai, T.-H. and Lee, C.-H. Regulation of exosomes as biologic medicines: regulatory challenges faced in exosome development and manufacturing processes. Clin Transl Sci 17, e13904 (2024).
- Suthar, J., Prieto-Simon, B., Williams, G. R. and Guldin, S. Dual-mode and label-free detection of exosomes from plasma using an electrochemical quartz crystal microbalance with dissipation monitoring. Anal Chem 94, 2465–2475 (2022).

 10.57647/jnsc.2026.1601.04
10.57647/jnsc.2026.1601.04