New forms of Graphitic Carbon Nitride: The Case of S-gC3N4-Au and the Perspectives of a Theragnostic
- Brazilian Nuclear Energy Commission, Nuclear Engineering Institute, Laboratory of Nanoradiopharmacy and Synthesis of New Radiopharmaceuticals, Rio de Janeiro-RJ, 21941906, Brazil
- Group of Chemistry of Advanced Materials (GQMat)- Department of Analytical Chemistry and Physical-Chemistry, Federal University of Ceará, Fortaleza-CE, 451-970, Brazil
- Federal University of Maranhão, Department of Physics, Biophysics Laboratory, Campus Bacanga, São Luís-MA, 65080-805, Brazil
- State University of Rio de Janeiro, Laboratory of Nanoradiopharmacy and Strategic Biomaterials, Rio de Janeiro-RJ, 220000, Brazil

Received: 10-07-2025
Revised: 10-10-2025
Accepted: 27-11-2025
Published in Issue 28-02-2026
Copyright (c) 2026 Jéssica Ingrid Faria De Souza, Natália Cristina Gomes-da-Silva, Natália Mayumi Andrade Yoshihara, Giulia Sigete-Lobo, Francisco Dheyson de Quadro Carvalho, Pedro Hilton Lima Baracho, Leticia Nogueira Xavier, Lillian Maria Uchôa Dutra Fechine, Tiago Melo Freire, Luciana Magalhães Rebelo Alencar, Alan Silva Menezes, Pierre Basílio Almeida Fechine, Ralph Santos-Oliveira (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 147
Abstract
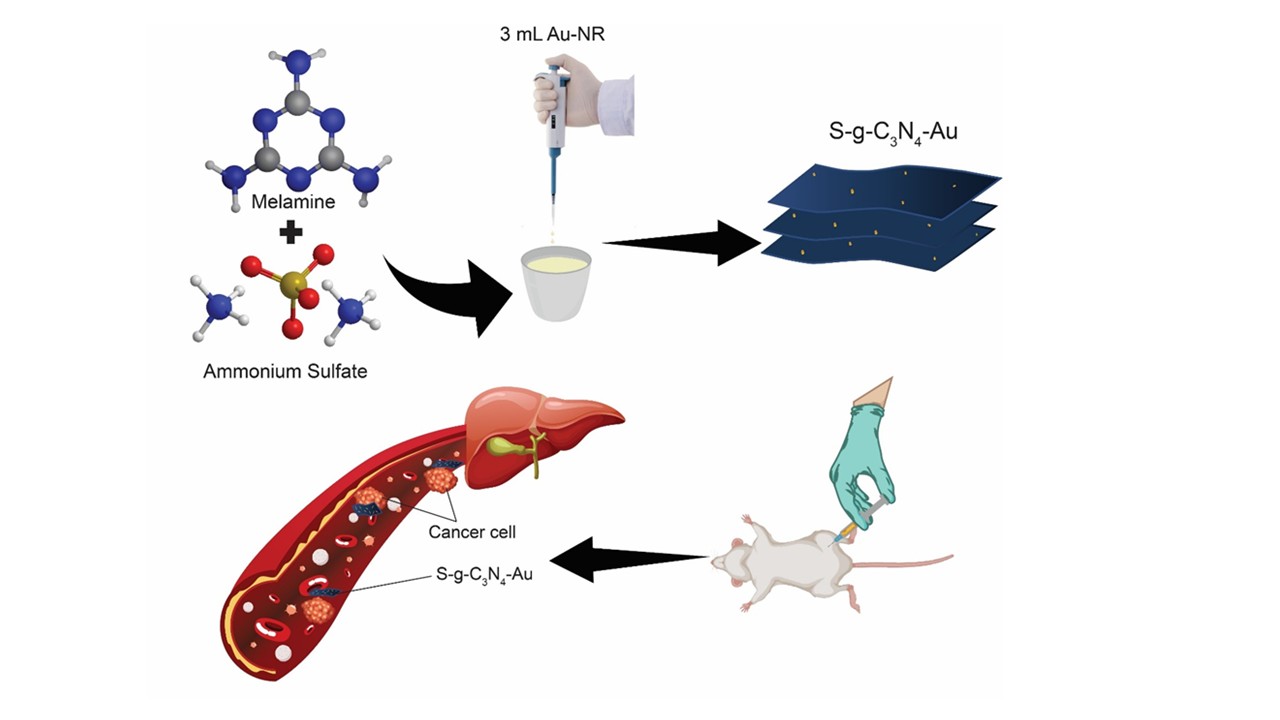
This study aimed to develop and characterize a sulfur-doped graphitic carbon nitride (S-g-C₃N₄) nanosystem functionalized with gold (S-g-C₃N₄-Au) for potential applications in cancer therapy. The focus was to evaluate its physicochemical properties, cytotoxicity against breast and prostate cancer cells, pharmacokinetics, biodistribution, and biochemical impact in vivo. To carry out the study, S-g-C₃N₄-Au was synthesized by thermal polymerization of melamine and ammonium sulfate with incorporation of gold seeds. Characterization techniques included SEM, XRD, FTIR, and UV-Vis DRS. Cytotoxicity was evaluated by MTT assays in MDA-MB-231 (breast cancer) and DU-145 (prostate cancer) cell lines. The nanosystem was radiolabeled with technetium-99m for biodistribution and pharmacokinetic studies in Balb/c mice after intraperitoneal administration. Biochemical analyses were conducted to evaluate systemic effects on the liver, kidneys, and metabolic markers. The results obtained demonstrated that the S-g-C₃N₄-Au nanosystem exhibited a porous, layered morphology with uniformly distributed gold nanoparticles. XRD and FTIR confirmed structural integrity, and SEM, STEM, and EDS confirmed successful gold incorporation. Cytotoxicity assays demonstrated significant, dose-dependent reductions in cell viability in both cancer cell lines, with the effects more pronounced at higher concentrations. In vivo, the nanosystem exhibited predominant accumulation in the liver and small intestine, with low systemic clearance and a plasma elimination half-life of 5.53 hours. Biochemical analysis indicated decreased ALT and glucose levels, but elevated AST, LDH-P, and lipase activities, suggesting stress on liver and digestive tissues. This set of results demonstrates that the S-g-C₃N₄-Au nanosystem exhibits favorable physicochemical properties and cytotoxic potential against cancer cells. Its organ-specific biodistribution and prolonged retention highlight its promise for targeted therapy and metabolic modulation. Notably, its ability to be radiolabeled with technetium-99m (99mTc) allows its application in nuclear imaging, further reinforcing its potential as a theranostic platform that integrates diagnostics and therapy in a single nanostructure. However, the observed biochemical alterations reinforce the need for comprehensive toxicological evaluations to ensure safety and enhance therapeutic efficacy.
Keywords
- Graphite Carbon nitride,
- 2D g-C3N4,
- Cytotoxic effect,
- Cancer cell
References
- De Silva, F., Alcorn, J.: A Tale of Two Cancers: A Current Concise Overview of Breast and Prostate Cancer. Cancers (Basel). 14, 2954 (2022).
- Rebello, R.J., Oing, C., Knudsen, K.E., Loeb, S., Johnson, D.C., Reiter, R.E., Gillessen, S., Van Der Kwast, T., Bristow, R.G.: Prostate cancer. Nat Rev Dis Primers. 7, 9 (2021).
- Arnold, M., Morgan, E., Rumgay, H., Mafra, A., Singh, D., Laversanne, M., Vignat, J., Gralow, J.R., Cardoso, F., Siesling, S., Soerjomataram, I.: Current and future burden of breast cancer: Global statistics for 2020 and 2040. The Breast 66, 15–23 (2022).
- Rawla, P. Epidemiology of Prostate Cancer. World J Oncol 10, 63–89 (2019).
- Debela, D.T., Muzazu, S.G., Heraro, K.D., Ndalama, M.T., Mesele, B.W., Haile, D.C., Kitui, S.K., Manyazewal, T.: New approaches and procedures for cancer treatment: Current perspectives. SAGE Open Med 9, 1–10 (2021).
- Kamal, K.M., Covvey, J.R., Dashputre, A., Ghosh, S., Shah, S., Bhosle, M., Zacker, C.:A Systematic Review of the Effect of Cancer Treatment on Work Productivity of Patients and Caregivers. J Manag Care Spec Pharm 23, 136–162 (2017).
- Zhang, W., Cao, G., Wu, F., Wang, Y., Liu, Z., Hu, H., Xu, K.: Global Burden of Prostate Cancer and Association with Socioeconomic Status, 1990–2019: A Systematic Analysis from the Global Burden of Disease Study. J Epidemiol Glob Health 13, 407–421 (2023).
- Brunckhorst, O., Liszka, J., James, C., Fanshawe, J.B., Hammadeh, M., Thomas, R., Khan, S., Sheriff, M., Ahmed, H.U., Van Hemelrijck, M., Muir, G., Stewart, R., Dasgupta, P., Ahmed, K.: Mental wellbeing and quality of life in prostate cancer (MIND-P): Protocol for a multi-institutional prospective cohort study. PLoS ONE 18, e0284727 (2023).
- Houédé, N., Rébillard, X., Bouvet, S., Kabani, S., Fabbro-Peray, P., Trétarre, B., Ménégaux, F.: Impact on quality of life 3 years after diagnosis of prostate cancer patients below 75 at diagnosis: an observational case-control study. BMC Cancer 20, 757 (2020).
- Taylor, J.M., Chen, V.E., Miller, R.C., Greenberger, B.A.: The Impact of Prostate Cancer Treatment on Quality of Life: A Narrative Review with a Focus on Randomized Data. Res Rep Urol 12, 533–546 (2020).
- Bhati, D., Deogade, M.S., Kanyal, D. Improving Patient Outcomes Through Effective Hospital Administration: A Comprehensive Review. Cureus 15, e47731. (2023).
- Elmore, L.W., Greer, S.F., Daniels, E.C., Saxe, C.C., Melner, M.H., Krawiec, G.M., Cance, W.G., Phelps, W.C.: Blueprint for cancer research: Critical gaps and opportunities. CA Cancer J Clin 71, 107–139 (2021).
- Lei, Z.-N., Teng, Q.-X., Tian, Q., Chen, W., Xie, Y., Wu, K., Zeng, Q., Zeng, L., Pan, Y., Chen, Z.-S., He, Y.: Signaling pathways and therapeutic interventions in gastric cancer. Sig Transduct Target Ther 7, 358 (2022).
- Barrios, C., De Lima Lopes, G., Yusof, M.M., Rubagumya, F., Rutkowski, P., Sengar, M.: Barriers in access to oncology drugs — a global crisis. Nat Rev Clin Oncol 20, 7–15 (2023).
- Sunilkumar, M.M., Finni, C.G., Lijimol, A.S., Rajagopal, M.R.: Health-Related Suffering and Palliative Care in Breast Cancer. Curr Breast Cancer Rep 13, 241–246 (2021).
- Schaft, N., Dörrie, J., Schuler, G., Schuler-Thurner, B., Sallam, H., Klein, S., Eisenberg, G., Frankenburg, S., Lotem, M., Khatib, A.: The future of affordable cancer immunotherapy. Front Immunol 14, 1248867 (2023).
- Mohammadpour, R., Dobrovolskaia, M.A., Cheney, D.L., Greish, K.F., Ghandehari, H.: Subchronic and chronic toxicity evaluation of inorganic nanoparticles for delivery applications. Adv Drug Deliv Rev 144, 112–132 (2019).
- Nawaz, A., Taj, M.B., Carabineiro, S.A.C. Graphitic carbon nitride as an efficient carrier for anti-cancer drug systems: A review. Next Nanotechnology 6, 100074 (2024).
- Shamim, M., Perveen, M., Nazir, S., Hussnain, M., Mehmood, R., Khan, M.I., Iqbal, J.: DFT study of therapeutic potential of graphitic carbon nitride (g-C3N4) as a new drug delivery system for carboplatin to treat cancer. J Mol Liq 331, 115607 (2021).
- Alipournazari, P., Pourmadadi, M., Abdouss, M., Rahdar, A., Pandey, S.: Enhanced delivery of doxorubicin for breast cancer treatment using pH-sensitive starch/PVA/g-C3N4 hydrogel. Int J Biol Macromol 265, 130901 (2024).
- Dash, P., Thirumurugan, S., Nataraj, N., Lin, Y.-C., Liu, X., Dhawan, U., Chung, R.-J.: Near-Infrared Driven Gold Nanoparticles-Decorated g-C3N4/SnS2 Heterostructure through Photodynamic and Photothermal Therapy for Cancer Treatment. Int J Nanomedicine 19, 10537–10550 (2024).
- S Singh, R., Kesharwani, P., Mehra, N.K., Singh, S., Banerjee, S., Jain, N.K.: Development and characterization of folate anchored Saquinavir entrapped PLGA nanoparticles for anti-tumor activity. Drug Dev Ind Pharm 41, 1888–1901 (2015).
- Carvalho, F.D.D.Q., Gomes-da-Silva, N.C., De Souza, J.I.F., Baracho, P.H.L., Freire, T.M., Xavier, L.N., Fechine, L.M.U.D., Batista, B.D.S., Alencar, L.M.R., De Menezes, A.S., Antunes, R.A., Santos-Oliveira, R., Fechine, P.B.A.: Pharmacokinetic analysis of ultrathin g-C₃N₄ nanosheets via acid exfoliation. Surf Interfaces 72, 107223 (2025).
- Lee, M.-K., Han, K.-D., Lee, J.-H., Sohn, S.-Y., Jeong, J.-S., Kim, M.-K., Baek, K.-H., Song, K.-H., Kwon, H.-S.:High hemoglobin levels are associated with decreased risk of diabetic retinopathy in Korean type 2 diabetes. Sci Rep 8, 5538 (2018).
- Freire, T.M., Sant’Anna, C., Yoshihara, N., Hu, R., Qu, J., Alencar, L.M.R., Oliveira Da Silva De Barros, A., Helal-Neto, E., Fernandes, L.R., Simoes, R.L., Barja-Fidalgo, C., Fechine, P.B.A., Santos-Oliveira, R.: Biomedical application of graphitic carbon nitrides: tissue deposition in vivo, induction of reactive oxygen species (ROS) and cell viability in tumor cells. Nanotechnology 32, 435301 (2021).
- Hong, L., Xu, H., Zhu, Y., Li, Z., Bai, B., Ding, G.: Surface Plasmon Resonance Enhanced Hydrogen Evolution from Water with Graphitic Carbon Nitride Photocatalyst. Catal Lett 153, 2296–2307 (2023).
- Arushi, Sharma, A., Arora, A., Mehta, N., Kataria, R., Mehta, S.K.: Au nanoparticles decorated graphitic carbon nitride nanosheets as a sensitive and selective fluorescence probe for Fe3+ and dichromate ions in aqueous medium. Chemosphere 363, 142834 (2024).
- Chebanenko, M.I., Zakharova, N.V., Lobinsky, A.A., Popkov, V.I.: Ultrasonic-Assisted Exfoliation of Graphitic Carbon Nitride and its Electrocatalytic Performance in Process of Ethanol Reforming. Semiconductors 53, 2072–2077 (2019).
- Guo, H., Shu, Z., Chen, D., Tan, Y., Zhou, J., Meng, F., Li, T.: One-step synthesis of S-doped g-C3N4 nanosheets for improved visible-light photocatalytic hydrogen evolution. Chemical Physics 533, 110714 (2020).
- Yang, Y., Yan, J., Zhang, Y., Xing, S., Ran, J., Ma, Y., Li, X.: S/P co-doped g-C3N4 with secondary calcination for excellent photocatalytic performance. Int J Hydrogen Energy 51, 962–974 (2024).
- Raziq, F., Humayun, M., Ali, A., Wang, T., Khan, A., Fu, Q., Luo, W., Zeng, H., Zheng, Z., Khan, B., Shen, H., Zu, X., Li, S., Qiao, L.: Synthesis of S-Doped porous g-C3N4 by using ionic liquids and subsequently coupled with Au-TiO2 for exceptional cocatalyst-free visible-light catalytic activities. Appl Catal B 237, 1082–1090 (2018).
- Holzwarth, U., Gibson, N.: The Scherrer equation versus the “Debye-Scherrer equation.” Nature Nanotech 6, 534–534 (2011).
- Mohtasham, H., Gholipour, B., Rostamnia, S., Ghiasi-Moaser, A., Farajzadeh, M., Nouruzi, N., Jang, H.W., Varma, R.S., Shokouhimehr, M.:Hydrothermally exfoliated P-doped g-C3N4 decorated with gold nanorods for highly efficient reduction of 4-nitrophenol. Colloids Surf A Physicochem Eng Asp 614, 126187 (2021).
- Liang, S., Xia, Y., Zhu, S., Zheng, S., He, Y., Bi, J., Liu, M., Wu, L.: Au and Pt co-loaded g-C3N4 nanosheets for enhanced photocatalytic hydrogen production under visible light irradiation. Appl Surf Sci 358, 304–312 (2015).
- Zhu, B., Xia, P., Li, Y., Ho, W., Yu, J.: Fabrication and photocatalytic activity enhanced mechanism of direct Z-scheme g-C 3 N 4 /Ag 2 WO 4 photocatalyst. Appl Surf Sci 391, 175–183 (2017).
- Pérez-Torres, A.F., Hernández-Barreto, D.F., Bernal, V., Giraldo, L., Moreno-Piraján, J.C., Da Silva, E.A., Alves, M.D.C.M., Morais, J., Hernandez, Y., Cortés, M.T., Macías, M.A.:Sulfur-Doped g-C3 N4 Heterojunctions for Efficient Visible Light Degradation of Methylene Blue. ACS Omega 8, 47821–47834 (2023).
- iu, G., Niu, P., Sun, C., Smith, S.C., Chen, Z., Lu, G.Q. (Max), Cheng, H.-M.: Unique Electronic Structure Induced High Photoreactivity of Sulfur-Doped Graphitic C3 N4. J. Am. Chem. Soc 132, 11642–11648 (2010).
- Lin, Y.-R., Dizon, G.V.C., Yamada, K., Liu, C.-Y., Venault, A., Lin, H.-Y., Yoshida, M., Hu, C.: Sulfur-doped g-C3N4 nanosheets for photocatalysis: Z-scheme water splitting and decreased biofouling. J Colloid Interface Sci 567, 202–212 (2020).
- Kadam, A.N., Moniruzzaman, Md., Lee, S.-W. Dual Functional S-Doped g-C3N4 Pinhole Porous Nanosheets for Selective Fluorescence Sensing of Ag+ and Visible-Light Photocatalysis of Dyes. Molecules 24, 450 (2019).
- Xiao, X., Wang, Y., Bo, Q., Xu, X., Zhang, D.: One-step preparation of sulfur-doped porous g-C3 N4 for enhanced visible light photocatalytic performance. Dalton Trans 49, 8041–8050 (2020).
- Guan, K., Li, J., Lei, W., Wang, H., Tong, Z., Jia, Q., Zhang, H., Zhang, S.: Synthesis of sulfur doped g-C3N4 with enhanced photocatalytic activity in molten salt. J Mater 7, 1131–1142 (2021).
- Almeida, A.S., Faleiros, A.C.G., Teixeira, D.N.S., Cota, U.A., Chica, J.E.L.: Valores de referência de parâmetros bioquímicos no sangue de duas linhagens de camundongos. J Bras Patol Med Lab 44, 429–432 (2008).
- Xu, M., Yang, G., Bi, H., Xu, J., Feng, L., Yang, D., Sun, Q., Gai, S., He, F., Dai, Y., Zhong, C., Yang, P.: Combination of CuS and g-C3N4 QDs on upconversion nanoparticles for targeted photothermal and photodynamic cancer therapy. Chem Eng J 360, 866–878 (2019).
- Godwin, M.A., Mahithashri, K., Shiney, O.J., Bhagat, M., Praseetha, P.K.: Metal Incorporated g-C3 N4 Nanosheets as Potential Cytotoxic Agents for Promoting Free Radical Scavenging in Cancer Cell Lines. J Nanosci Nanotechnol 19, 5448–5455 (2019).
- P Padmanabhan, V.P., Sivashanmugam, P., S. M., M., Sagadevan, S., Kulandaivelu, R.: The development of ZnO nanoparticle-embedded graphitic-carbon nitride towards triple-negative breast cancer therapy. RSC Adv 13, 24333–24342 (2023).
- Muhammad, N., Guo, Z. Metal-based anticancer chemotherapeutic agents. Curr Opin Chem Biol 19, 144–153 (2014).
- Khan, H.Y., Ansari, M.F., Tabassum, S., Arjmand, F.: A review on the recent advances of interaction studies of anticancer metal-based drugs with therapeutic targets, DNA and RNAs. Drug Discov Today 29, 104055 (2024).
- Hannon, M.J. Metal-based anticancer drugs: From a past anchored in platinum chemistry to a post-genomic future of diverse chemistry and biology. Pure Appl Chem 79, 2243–2261 (2007).
- Bufarwa, S.M., El-Sefait, R.M., Thbayh, D.K., Belaidi, M., Al-Shemary, R.K., Abdusamea, Rema.M., El-Ajaily, M.M., Fiser, B., Bader, H.A., Saleh, A.A., Bufarwa, M.M.: Antituberculosis, antimicrobial, antioxidant, cytotoxicity and anti-inflammatory activity of Schiff base derived from 2,3-diaminophenazine moiety and its metal(II) complexes: structural elucidation, computational aspects, and biological evaluation. Rev Inorg Chem 45, 105–124 (2025).
- Naseem, K., Aziz, A., Khan, M.E., Ali, S., Khalid, A.: Bioinorganic metal nanoparticles and their potential applications as antimicrobial, antioxidant and catalytic agents: a review. Rev Inorg Chem 45, 207–236 (2025).
- Duan, X., Li, Y. Physicochemical Characteristics of Nanoparticles Affect Circulation, Biodistribution, Cellular Internalization, and Trafficking. Small 9, 1521–1532 (2013).
- Owens, D.E., Peppas, N.A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int J Pharm 307, 93–102 (2006).
- Åslund, A.K.O., Vandebriel, R.J., Caputo, F., De Jong, W.H., Delmaar, C., Hyldbakk, A., Rustique, E., Schmid, R., Snipstad, S., Texier, I., Vernstad, K., Borgos, S.E.F.: A comparative biodistribution study of polymeric and lipid-based nanoparticles. Drug Deliv and Transl Res 12, 2114–2131 (2022).
- Kumar, M., Kulkarni, P., Liu, S., Chemuturi, N., Shah, D.K.: Nanoparticle biodistribution coefficients: A quantitative approach for understanding the tissue distribution of nanoparticles. Adv Drug Deliv Rev 194, 114708 (2023).
- Liu, T., Choi, H., Zhou, R., Chen, I.-W.: Quantitative Evaluation of the Reticuloendothelial System Function with Dynamic MRI. PLoS ONE 9, e103576 (2014).
- Li, S.-D., Huang, L. Nanoparticles evading the reticuloendothelial system: Role of the supported bilayer. Biochim Biophys Acta Biomembr 1788, 2259–2266 (2009).
- Tang, Y., Wang, X., Li, J., Nie, Y., Liao, G., Yu, Y., Li, C.: Overcoming the Reticuloendothelial System Barrier to Drug Delivery with a “Don’t-Eat-Us” Strategy. ACS Nano 13, 13015–13026 (2019).
- Zhang, M., Gao, S., Yang, D., Fang, Y., Lin, X., Jin, X., Liu, Y., Liu, X., Su, K., Shi, K.: Influencing factors and strategies of enhancing nanoparticles into tumors in vivo. Acta Pharm Sin B 11, 2265–2285 (2021).
- Zhang, X.-D., Wu, D., Shen, X., Liu, P.-X., Fan, F.-Y., Fan, S.-J.: In vivo renal clearance, biodistribution, toxicity of gold nanoclusters. Biomaterials 33, 4628–4638 (2012).
- Daems, N., Verlinden, B., Van Hoecke, K., Cardinaels, T., Baatout, S., Michiels, C., Lucas, S., Aerts, A.: In Vivo Pharmacokinetics, Biodistribution and Toxicity of Antibody-Conjugated Gold Nanoparticles in Healthy Mice. J Biomed Nanotechnol 16, 985–996 (2020).
- Escudero-Francos, M.A., Cepas, V., González-Menédez, P., Badía-Laíño, R., Díaz-García, M.E., Sainz, R.M., Mayo, J.C., Hevia, D.: Cellular Uptake and Tissue Biodistribution of Functionalized Gold Nanoparticles and Nanoclusters. J Biomed Nanotechnol 13, 167–179 (2017)
- Li, N., Chen, L., Zeng, C., Yang, H., He, S., Wei, Q.: Comparative Toxicity, Biodistribution and Excretion of Ultra-Small Gold Nanoclusters with Different Emission Wavelengths. J Biomed Nanotechnol 17, 1778–1787 (2021).
- Colino, C.I., Lanao, J.M., Gutierrez-Millan, C. Targeting of Hepatic Macrophages by Therapeutic Nanoparticles. Front. Immunol 11, 218 (2020).
- De Matteis, V. Exposure to Inorganic Nanoparticles: Routes of Entry, Immune Response, Biodistribution and In Vitro/In Vivo Toxicity Evaluation. Toxics 5, 29 (2017).
- Zelepukin, I.V., Shevchenko, K.G., Deyev, S.M. Rediscovery of mononuclear phagocyte system blockade for nanoparticle drug delivery. Nat Commun 15, 4366 (2024).
- Gustafson, H.H., Holt-Casper, D., Grainger, D.W., Ghandehari, H.: Nanoparticle uptake: The phagocyte problem. Nano Today 10, 487–510 (2015).
- Albanese, A., Tang, P.S., Chan, W.C.W. The Effect of Nanoparticle Size, Shape, and Surface Chemistry on Biological Systems. Annu Rev Biomed Eng 14, 1–16 (2012).
- Wei, Y., Quan, L., Zhou, C., Zhan, Q.: Factors Relating to the Biodistribution & Clearance of Nanoparticles & Their Effects on In Vivo Application. Nanomedicine (Lond) 13, 1495–1512 (2018).
- Tavares, A.J., Poon, W., Zhang, Y.-N., Dai, Q., Besla, R., Ding, D., Ouyang, B., Li, A., Chen, J., Zheng, G., Robbins, C., Chan, W.C.W.:Effect of removing Kupffer cells on nanoparticle tumor delivery. Proc Natl Acad Sci U S A 114, (2017).
- Pandey, P., Patel, J., Kumar, S., Pathak, Y.: Pharmacokinetics and Pharmacodynamics of Liposomal Nanoparticles. In: Patel, J.K. and Pathak, Y.V. (eds.) Pharmacokinetics and Pharmacodynamics of Nanoparticulate Drug Delivery Systems. pp. 143–158. Springer International Publishing, Cham (2022).
- Verma, A.K., Kumar, A. Pharmacokinetics and biodistribution of negatively charged pectin nanoparticles encapsulating paclitaxel. Cancer Nano 4, 99–102 (2013).
- Haripriyaa, M., Suthindhiran, K. Pharmacokinetics of nanoparticles: current knowledge, future directions and its implications in drug delivery. Futur J Pharm Sci 9, 113 (2023).
- Li, J., Wang, H. Selective organ targeting nanoparticles: from design to clinical translation. Nanoscale Horiz 8, 1155–1173 (2023).
- Y Yoshioka, Y., Higashisaka, K., Tsunoda, S., Tsutsumi, Y.: The Absorption, Distribution, Metabolism, and Excretion Profile of Nanoparticles. In: Akashi, M., Akagi, T., and Matsusaki, M. (eds.) Engineered Cell Manipulation for Biomedical Application. pp. 259–271. Springer Japan, Tokyo. (2014)
- Alexis, F., Pridgen, E., Molnar, L.K., Farokhzad, O.C.: Factors Affecting the Clearance and Biodistribution of Polymeric Nanoparticles. Mol Pharm 5, 505–515 (2008).
- Wang, L., Quine, S., Frickenstein, A.N., Lee, M., Yang, W., Sheth, V.M., Bourlon, M.D., He, Y., Lyu, S., Garcia‐Contreras, L., Zhao, Y.D., Wilhelm, S.: Exploring and Analyzing the Systemic Delivery Barriers for Nanoparticles. Adv Funct Materials 34, 2308446 (2024).
- Hu, J., Sheng, Y., Shi, J., Yu, B., Yu, Z., Liao, G.:Long Circulating Polymeric Nanoparticles for Gene/Drug Delivery. Curr Drug Metab 19, 723–738 (2018).
- Guyton, A.C., Hall, J.E. Textbook of medical physiology. Elsevier Saunders, Philadelphia (2006)
- Huang, X.-J., Choi, Y.-K., Im, H.-S., Yarimaga, O., Yoon, E., Kim, H.-S.: Aspartate Aminotransferase (AST/GOT) and Alanine Aminotransferase (ALT/GPT) Detection Techniques. Sensors 6, 756–782 (2006).
- Das, S.K., Sen, K., Ghosh, B., Ghosh, N., Sinha, K., Sil, P.C.: Molecular mechanism of nanomaterials induced liver injury: A review. World J Hepatol 16, 566–600 (2024).
- Zhang, H., Kang, K., Chen, S., Su, Q., Zhang, W., Zeng, L., Lin, X., Peng, F., Lin, J., Chai, D.: High serum lactate dehydrogenase as a predictor of cardiac insufficiency at follow-up in elderly patients with acute myocardial infarction. Arch Gerontol Geriatr 117, 105253 (2024).
- Khan, A.A., Allemailem, K.S., Alhumaydhi, F.A., Gowder, S.J.T., Rahmani, A.H.: The Biochemical and Clinical Perspectives of Lactate Dehydrogenase: An Enzyme of Active Metabolism. Endocr Metab Immune Disord Drug Targets 20, 855–868 (2020).
- Rafaqat, S., Radoman Vujacic, I., Behnoush, A.H., Sharif, S., Klisic, A.: Role of Cardiac Biomarkers in Hepatic Disorders: A Literature Review. Metab Syndr Relat Disord 22, 251–262 (2024).
- Grundy, S.M., Stone, N.J., Bailey, A.L., Beam, C., Birtcher, K.K., Blumenthal, R.S., Braun, L.T., De Ferranti, S., Faiella-Tommasino, J., Forman, D.E., Goldberg, R., Heidenreich, P.A., Hlatky, M.A., Jones, D.W., Lloyd-Jones, D., Lopez-Pajares, N., Ndumele, C.E., Orringer, C.E., Peralta, C.A., Saseen, J.J., Smith, S.C., Sperling, L., Virani, S.S., Yeboah, J.: 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: Executive Summary. J Am Coll Cardiol 73, 3168–3209 (2019).
- Tong, S., Luo, S., Yang, Q., Song, B., Chang, R., Wu, J.: Preparation and biological evaluation of a glucose-responsive block copolymer nanoparticle with the ability to ameliorate diabetic kidney damage. Eur Polym J 220, 113472 (2024).
- Mohammadparast, V., Mallard, B.L. The effect and underlying mechanisms of titanium dioxide nanoparticles on glucose homeostasis: A literature review. J Appl Toxicol 43, 22–31 (2023).
- Lin, Q., Qiu, C., Li, X., Sang, S., McClements, D.J., Chen, L., Long, J., Jiao, A., Tian, Y., Jin, Z.:The inhibitory mechanism of amylase inhibitors and research progress in nanoparticle-based inhibitors. Crit Rev Food Sci Nutr 63, 12126–12135 (2023)
- Jalal, M., Gbadegesin, S.A., Tehami, N., Nakajima, K.: What is the clinical significance of low serum amylase? Systematic review of the conditions associated with low serum amylase. Frontline Gastroenterol 15, 154–161 (2024).
- Ko, J., Cho, J., Petrov, M.S. Low serum amylase, lipase, and trypsin as biomarkers of metabolic disorders: A systematic review and meta-analysis. Diabetes Res Clin Pract 159, 107974 (2020).

 10.57647/jnsc.2026.1601.02
10.57647/jnsc.2026.1601.02