A New Heterogeneous PMO@MXene Catalyst for One-Pot Synthesis of Substituted 5-amino-3-(aryl)-1-phenyl-1H-pyrazole-4-carbonitrile Derivatives
- Department of Organic Chemistry, Faculty of Chemistry, University of Guilan, Rasht, Iran
- Department of Surface Coating and Corrosion, Institute for Color Science and Technology, Tehran, Iran

Received: 02-07-2025
Revised: 12-08-2025
Accepted: 04-10-2025
Published in Issue 17-10-2025
Copyright (c) 2025 Safa Hnaif, Farhad Shirini, Bahram Ramezanzadeh, Hassan Tajik (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 186
Abstract
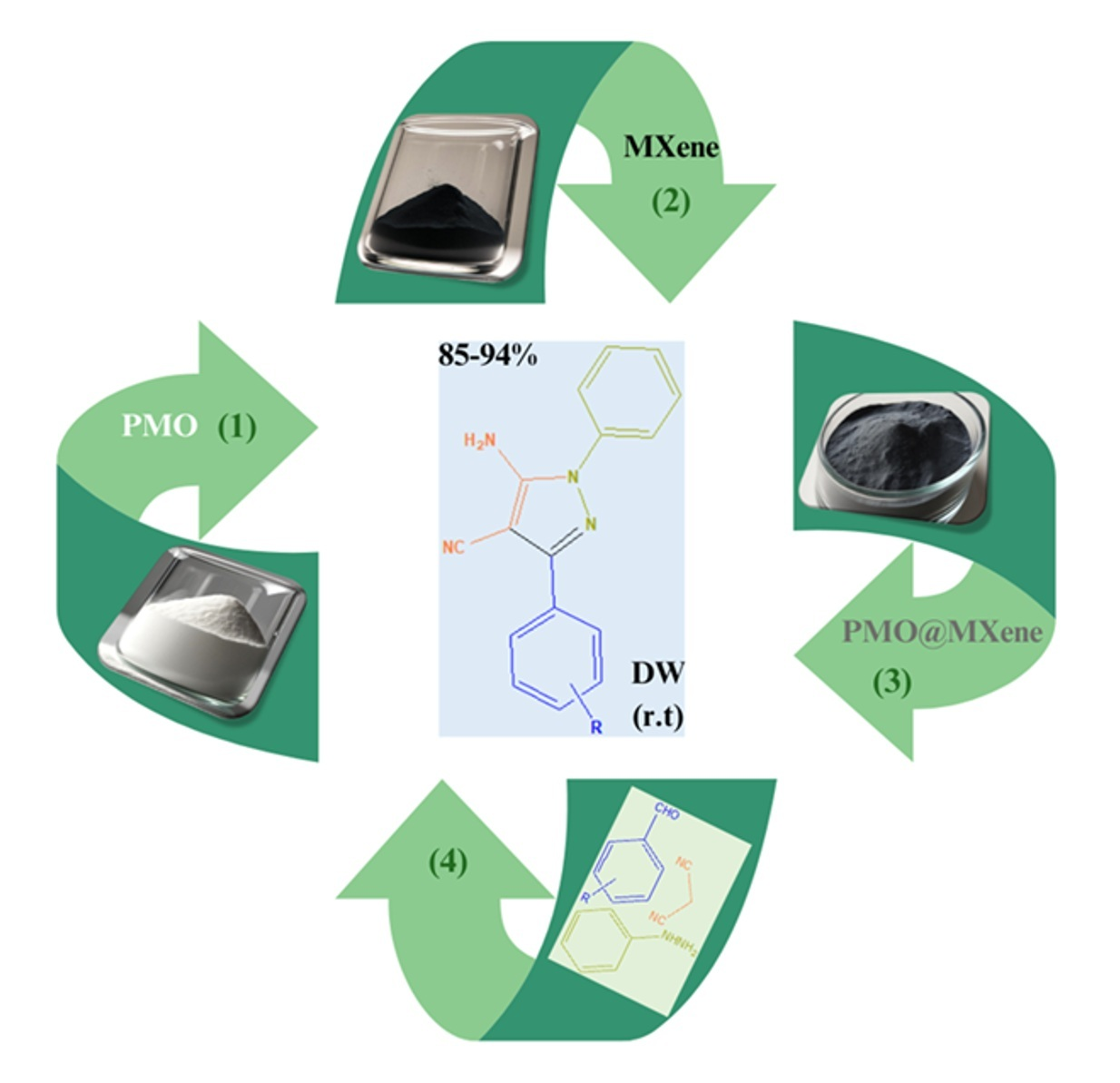
In this study, we report the development of a novel and efficient MXene-based heterogeneous catalyst, PMO@MXene, for the green synthesis of 5-amino-3-(aryl)-1-phenyl-1H-pyrazole-4-carbonitrile derivatives. By integrating periodic mesoporous organosilica (PMO) into multilayered MXene, a hybrid nanocomposite was achieved that benefits from the high surface area and electrical conductivity of MXene along with the welldefined mesoporosity of PMO. This unique synergy enabled highly efficient catalytic performance under mild, eco-friendly conditions, requiring only a small amount of catalyst and allowing for easy recovery and reuse with minimal loss of activity. The pyrazole derivatives, known for their potential pharmaceutical relevance, were obtained in excellent yields and characterized using standard spectroscopic techniques. Importantly, this work highlights the potential of the PMO@MXene as a sustainable and recyclable heterogeneous catalyst platform for the synthesis of biologically significant heterocyclic compounds.
Keywords
- MXene,
- Periodic mesoporous organosillica,
- Pyrazole,
- Ti3AlC2,
- PMO,
- Heterogeneous catalyst,
- Nanocomposite
References
- Melchionna M, Fornasiero P. What Is to Be Expected from Heterogeneous Catalysis in the Pipeline to Circular Economy? Chem Sus Chem. 18(5), e202402064 (2025).
- Wacławek S. Greener Catalysis for Environmental Applications: MDPI-Multidisciplinary Digital Publishing Institute; (2022).
- Haghighat M, Shirini F, Golshekan M. Periodic mesoporous organosilica containing bridges with N-sulfonic acid groups: A new catalyst for the efficient formylation of amines and alcohols. Silicon. ;12(9), 2087-98 (2020).
- Zou Y, Sun Z, Wang Q, Ju Y, Sun N, Yue Q, et al. Core–Shell Magnetic Particles: Tailored Synthesis and Applications. Chem Rev. 125(2), 972-1048 (2024).
- Bordet A, Leitner W, Chaudret B. Magnetically Induced Catalysis: Definition, Advances, and Potential. Angewandte Chemie International Edition. 64(24), e202424151 (2025).
- Mohammadi M, Khodamorady M, Tahmasbi B, Bahrami K, Ghorbani-Choghamarani A. Boehmite nanoparticles as versatile support for organic–inorganic hybrid materials: Synthesis, functionalization, and applications in eco-friendly catalysis. J Ind Eng Chem. 97, 1-78 (2021).
- Reina A, Carmona‐Chávez R, Pulido‐Díaz IT, Martínez D, Salas‐Martin KP, Guerrero‐Ríos I. Silica‐Supported 1st Row Transition Metal (Nano) Catalysts: Synthetic and Catalytic Insight. Chem Cat Chem. 15(11), e202300285 (2023).
- Yu X, Williams CT. Recent advances in the applications of mesoporous silica in heterogeneous catalysis. Cat Sci Technol. 12(19), 5765-94 (2022).
- Erigoni A, Diaz U. Porous silica-based organic-inorganic hybrid catalysts: A review. Catalysts. 11(1), 79 (2021).
- Amirsoleimani, A.; Bahrami, Z.; Kafshdouzan, K. Synthesis of a novel antibacterial nanocomposite based on periodic mesoporous organosilica, zinc oxide, and medical leech saliva as a nanocarrier for streptomycin release. Surf Interfaces, 64, 106445 (2025).
- Soni, A. B.; Satishkumar, G.; Man, M. W. C. Palladium Nanoparticles Stabilized in Ionic Liquid‐Based Periodic Mesoporous Organosilica: An Efficient Heterogeneous Catalyst for Oxidative Acylation of Ketones. Chemistry Select, 9 (44), e202403464 (2024).
- Melde BJ, Holland BT, Blanford CF, Stein A. Mesoporous sieves with unified hybrid inorganic/organic frameworks. Chem Mater. 11(11), 3302-8 (1999).
- Asefa T, MacLachlan MJ, Coombs N, Ozin GA. Periodic mesoporous organosilicas with organic groups inside the channel walls. Nature. 402(6764), 867-71 (1999).
- Moghaddam SMH, Haghighat M, Shirini F. Ionic liquid-based periodic mesoporous organosilicas: Metal/solvent-free synthesis of formamides and formamidines. J Mol Struct. 1274, 134473 (2023).
- Karimi B, Ganji N, Pourshiani O, Thiel WR. Periodic mesoporous organosilicas (PMOs): From synthesis strategies to applications. Prog Mater Sci. 125, 100896 (2022).
- Mizoshita N, Tani T, Inagaki S. Syntheses, properties and applications of periodic mesoporous organosilicas prepared from bridged organosilane precursors. Chem Soc Rev. 40(2), 789-800 (2011).
- Yang Q, Liu J, Zhang L, Li C. Functionalized periodic mesoporous organosilicas for catalysis. J Mater Chem. 19(14), 1945-55 (2009).
- Haghighat M, Golshekan M, Shirini F. Periodic mesoporous organosilica containing bridged N‐Sulfonic acid groups: promotion of the synthesis of N, N’‐diarylformamidines, benzoxazoles, benzothiazoles and benzimidazoles. Chem Select. 4(27), 7968-75 (2019).
- Handoko AD, Fredrickson KD, Anasori B, Convey KW, Johnson LR, Gogotsi Y, et al. Tuning the basal plane functionalization of two-dimensional metal carbides (MXenes) to control hydrogen evolution activity. ACS Appl Energ Mater. 1(1), 173-80 (2017).
- Xu, S.; Zhang, X.; Zheng, T.; Zhao, Z.; Shang, C.; Hu, Z.; Dong, M.; Qiao, Y.; Bai, C.; Zhang, X. Advancements in high-performance MXene composite fibers integrated with various functional materials: Fabrication, functionalization, property enhancement, and applications. J Mater Sci Technol. 252 (1) (2025).
- Cai, Z.; Kim, H. Recent advances in MXene gas sensors: synthesis, composites, and mechanisms. NPJ 2D Mater Appl. 9 (1), 66 (2025).
- Shao H, Luo S, Descamps‐Mandine A, Ge K, Lin Z, Taberna Pl, et al. Synthesis of MAX phase nanofibers and nanoflakes and the resulting MXenes. Adv Sci. 10(1), 2205509 (2023).
- Khabisi MA, Shirini F, Shirini K, Khorsand H, Marian M, Rosenkranz A. Additively manufactured MAX-and MXene-composite scaffolds for bone regeneration-recent advances and future perspectives. Colloid Surface B. 225, 113282 (2023).
- Riazi H, Nemani SK, Grady MC, Anasori B, Soroush M. Ti3C2 MXene–polymer nanocomposites and their applications. J Mater Chem A. 9(13), 8051-98 (2019).
- Alhabeb M, Maleski K, Anasori B, Lelyukh P, Clark L, Sin S, et al. Guidelines for synthesis and processing of two-dimensional titanium carbide (Ti3C2Tx MXene). Chem Mater. 29(18), 7633-44 (2017).
- Naguib M, Mochalin VN, Barsoum MW, Gogotsi Y. Two‐dimensional materials: 25th anniversary article: MXenes: a new family of two‐dimensional materials. Adv Mater. 26(7), 982-982 (2014).
- Barsoum MW. MAX phases: properties of machinable ternary carbides and nitrides: John Wiley & Sons; (2013).
- Alhabeb M, Maleski K, Mathis TS, Sarycheva A, Hatter CB, Uzun S, et al. Selective etching of silicon from Ti3SiC2 (MAX) to obtain 2D titanium carbide (MXene). Mxenes: Jenny Stanford Publishing. 4, p. 451-62 (2023).
- Shuck CE, Han M, Maleski K, Hantanasirisakul K, Kim SJ, Choi J, et al. Effect of Ti3AlC2 MAX phase on structure and properties of resultant Ti3C2Tx MXene. ACS Appl Nano Mater. 2(6), 3368-76 (2019).
- Singh N, Pandey J. DABCO catalyzed, green and efficient, one-pot multicomponent synthesis of 5-aminopyrazole-4-carbonitrile. CRGSC. 4, 100134 (2021).
- Rehman, M. U., He, F., Shu, X., Guo, J., Liu, Z., Cao, S., & Long, S.. Antibacterial and antifungal pyrazoles based on different construction strategies. Eur J Med Chem, 282, 117081 (2025).
- Hanifi S, Dekamin MG, Eslami M. Magnetic BiFeO3 nanoparticles: a robust and efficient nanocatalyst for the green one-pot three-component synthesis of highly substituted 3, 4-dihydropyrimidine-2 (1H)-one/thione derivatives. Sci Rep. 14(1), 22201 (2024).
- Li G, Cheng Y, Han C, Song C, Huang N, Du Y. Pyrazole-containing pharmaceuticals: target, pharmacological activity, and their SAR studies. RSC Med Chem. 13(11), 1300-21 (2022).
- Krygowski TM, Anulewicz R, Cyrański MK, Puchala A, Rasala D. Separation of the energetic and geometric contribution to the aromaticity. Part IX. Aromaticity of pyrazoles in dependence on the kind of substitution. Tetrahedron. 54(40), 12295-300 (1998).
- Rue K, Raptis RG. Low-temperature crystal structure of 4-chloro-1H-pyrazole. Structure Reports. 77(9), 955-7 (2021).
- Thore S, Gupta SV, Baheti KG. Novel ethyl-5-amino-3-methylthio-1H-pyrazole-4-carboxylates: Synthesis and pharmacological activity. J Saudi Chem Soc. 20(3), 259-64 (2016).
- Liu M, Li J, Chai H, Zhang K, Yang D, Zhang Q, et al. A convenient four-component one-pot strategy toward the synthesis of pyrazolo [3,4-d] pyrimidines. Beilstein J Org Chem. 11(1), 2125-31 (2015).
- Nemati F, Nikkhah SH, Elhampour A. An environmental friendly approach for the catalyst-free synthesis of highly substituted pyrazoles promoted by ultrasonic radiation. Chinese Chem Lett. 26(11), 1397-9 (2015).
- Gupta S, Rodrigues LM, Esteves AP, Oliveira-Campos AM, Nascimento MSJ, Nazareth N, et al. Synthesis of N-aryl-5-amino-4-cyanopyrazole derivatives as potent xanthine oxidase inhibitors. Euro J Med Chem. 43(4), 771-80 (2008).
- Alizadeh A, Roosta A. A convenient approach for the synthesis of 1, 3-diphenyl-1H-pyrazole-5-carbonitrile. Synlett. 27(17), 2455-8 (2016).
- Elnagdy HM, Sarma D. FeCl3/PVP as Green Homogeneous Catalyst to Synthesize 5‐Amino‐1H‐Pyrazole‐4‐Carbonitriles from Malononitrile Derivatives. Chem Select. 4(3), 783-7 (2019).
- Mashhadinezhad M, Shirini F, Mamaghani M. Nanoporous Na+-montmorillonite perchloric acid as an efficient heterogeneous catalyst for synthesis of merocyanine dyes based on isoxazolone and barbituric acid. Micropor Mesopor Mat. 262, 269-82 (2018).
- Seddighi M, Shirini F, Mamaghani M. Sulfonated rice husk ash (RHA-SO3H) as a highly efficient and reusable catalyst for the synthesis of some bis-heterocyclic compounds. RSC Adv. 3(46), 24046-53 (2013).
- Surchani MM, Shirini F, Tajik H, Jolodar OG. 2, 2â-Bipyridine: An efficient organo-catalyst for the synthesis of pyrano [2, 3-d] pyrimidine and pyrido [2, 3-d] pyrimidine derivatives. Iranian J Cat. 14(3) (2024).
- Hideshima S, Kawasaki Y, Takimoto D, Gogotsi Y, Sugimoto W. Size-dependent electrochemical properties of vertically aligned MXene electrodes for fast Li-ion storage. Electrochimica Acta. 519, 145849 (2025).
- Arora P, Rajput JK. One-pot multicomponent click synthesis of pyrazole derivatives using cyclodextrin-supported capsaicin nanoparticles as catalyst. J Mater Sci. 52(19), 11413-27 (2017).
- Kiyani H, Bamdad M. Sodium ascorbate as an expedient catalyst for green synthesis of polysubstituted 5-aminopyrazole-4-carbonitriles and 6-amino-1, 4-dihydropyrano [2, 3-c] pyrazole-5-carbonitriles. Res Chem Intermediate. 44(4) (2018), 2761-78.
- Sapkal A, Kamble S. Sodium toluene‐4‐sulfonate as a reusable and eco-friendly catalyst for greener synthesis of 5‐aminopyrazole‐4‐carbonitrile in aqueous medium. J Heterocyclic Chem. 57(10),3597-604 (2020).
- Soleimani M, Akbarpour T, Khazaei A. Fabrication of Copper (II)-coated Magnetic Core-shell Nanoparticles an Engineered Nano-magnetic Catalyst for the Synthesis of Pyrano Pyrazole and Pyrazole Derivatives. Polycycl Aromati Comp. 44(1), 90-116 (2024).
- Gorji A, Akbarpour T, Khazaei A. Synthesis of hexahydroquinolines, 5-amino-1, 3-diphenyl-1H-pyrazole-4-carbonitrile and 1-aminoalkyl-2-naphthols derivatives using an engineered copper-based nano-magnetic catalyst (Fe3O4@ CQD@ Si (OEt)(CH2)3NH@CC@Ad@Cu (OAc)2). Polycycl Aromat Comp. 43(6), 5041-73 (2023).
- Singh N, Pandey J. DABCO catalyzed, green and efficient, one-pot multicomponent synthesis of 5-aminopyrazole-4-carbonitrile. Current Research in Green and Sustainable Chemistry. 4, 100134 (2021).
- Obaid AR, Shiri L. Synthesis, Characterization, and Application of CoFe2O4@ SiO2@ CPTES@ Melamine Nanoparticles as a Magnetic Catalyst for the Synthesis of Pyrazole Derivatives. J Appl Organometall Chem. 4, 178-89 (2024).
- Moosavi-Zare AR, Rezaei-Gohar M, Tavasoli M, Goudarziafshar H. The synthesis of 5-amino-1H-pyrazole-4-carbonitriles by anomeric-based oxidative aromatization over [CSPy] ZnCl3 as a new catalyst. Res on Chem Intermediate. 47(7), 2689-700 (2021).

 10.57647/jnsc.2025.1505.19
10.57647/jnsc.2025.1505.19