Published in Issue 31-05-2013
How to Cite
Atghia, S. V., & Beigbaghlou, S. S. (2013). Nanocrystalline titania-based sulfonic acid (TiO2-Pr-SO3H) as a new, highly efficient, and recyclable solid acid catalyst for preparation of quinoxaline derivatives. Journal of Nanostructure in Chemistry, 3(1 (December 2013). https://doi.org/10.1186/2193-8865-3-38
HTML views: 34
PDF views: 112
Abstract
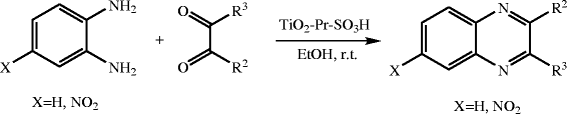
Abstract Nanocrystalline titania-based sulfonic acid (TiO 2 -Pr-SO 3 H) was used as an efficient and reusable catalyst for the synthesis of quinoxalines. The clean and mild acidity condition, quantitative yields of products, short reaction time, and low reaction temperature are attractive features of this method, making it suitable for heat- or acid-sensitive substrates, particularly in drug synthesis. In practice, this method affords an advantageous combination of satisfactory yields, easy product isolation, and purification.Keywords
- Nanocrystalline titania,
- 1,2-Dicarbonyls,
- Quinoxaline,
- Solid acid catalyst
References
- Sarges et al. (1990) 4-Amino[1,2,4]triazolo[4,3-a]quinoxalines. A novel class of potent adenosine receptor antagonists and potential rapid-onset antidepressants (pp. 2240-2254) https://doi.org/10.1021/jm00170a031
- Brown (2005) Chemistry and enzymology of vitamin B12 (pp. 2075-2150) https://doi.org/10.1021/cr030720z
- Sakata et al. (1998) Recent progress in the quinoline chemistry: synthesis and biological activity (pp. 2481-2515)
- Ali et al. (2000) Synthesis and antimicrobial activities of some novel quinoxalinone derivatives (pp. 864-873) https://doi.org/10.3390/50600864
- Sonawane and Rangnekar (2002) Synthesis and application of 2-styryl-6,7-dichlorothiazolo[4,5-b]-quinoxaline based fluorescent dyes: part 3 (pp. 303-308) https://doi.org/10.1002/jhet.5570390210
- Dailey et al. (2001) Synthesis and device characterisation of side-chain polymer electron transport materials for organic semiconductor applications (pp. 2238-2243) https://doi.org/10.1039/b104674h
- Elwahy (2000) Synthesis of new benzo-substituted macrocyclic ligands containing quinoxaline subunits (pp. 897-907) https://doi.org/10.1016/S0040-4020(99)01072-8
- Crossley and Johnston (2002) Laterally-extended porphyrin systems incorporating a switchable unit
- Jafarpour et al. (2011) Easy access to quinoxaline derivatives using alumina as an effective and reusable catalyst under solvent-free conditions (pp. 48-51) https://doi.org/10.1016/j.apcata.2010.12.022
- Huang et al. (2008) Montmorillonite K-10: an efficient and reusable catalyst for the synthesis of quinoxaline derivatives in water (pp. 1143-1147) https://doi.org/10.1016/j.catcom.2007.10.024
- Krishnakumar and Swaminathan (2011) Solvent free synthesis of quinoxalines, dipyridophenazines and chalcones under microwave irradiation with sulfated Degussa titania as a novel solid acid catalyst (pp. 16-25) https://doi.org/10.1016/j.molcata.2011.08.026
- Krishnakumar and Swaminathan (2010) A recyclable and highly effective sulfated TiO2-P25 for the synthesis of quinoxaline and dipyridophenazine derivatives at room temperature (pp. 2572-2577) https://doi.org/10.1016/j.jorganchem.2010.08.055
- Krishnakumar et al. (2010) An efficient protocol for the green synthesis of quinoxaline and dipyridophenazine derivatives at room temperature using sulfated titania (pp. 997-1002) https://doi.org/10.1016/j.catcom.2010.04.021
- Dhakshinamoorthy et al. (2011) Zn2+-K10-clay (clayzic) as an efficient water-tolerant, solid acid catalyst for the synthesis of benzimidazoles and quinoxalines at room temperature (pp. 69-73) https://doi.org/10.1016/j.tetlet.2010.10.146
- Sharma and Sharma (2011) Zirconium(IV)-modified silica gel: preparation, characterization and catalytic activity in the synthesis of some biologically important molecules (pp. 327-331) https://doi.org/10.1016/j.catcom.2010.10.011
- Zhang et al. (2010) Synthesis of quinoxaline derivatives catalyzed by PEG-400 (pp. 395-398) https://doi.org/10.1016/j.cclet.2009.12.015
- Heravi et al. (2007) Wells-Dawson type heteropolyacid catalyzed synthesis of quinoxaline derivatives at room temperature (pp. 465-467) https://doi.org/10.1007/s00706-007-0594-5
- Ajaikumar and Pandurangan (2009) Efficient synthesis of quinoxaline derivatives over ZrO2/MxOy (M = Al, Ga, In and La) mixed metal oxides supported on MCM-41 mesoporous molecular sieves (pp. 184-192) https://doi.org/10.1016/j.apcata.2009.01.021
- Shaabani et al. (2009) Green chemistry approaches for the synthesis of quinoxaline derivatives: comparison of ethanol and water in the presence of the reusable catalyst cellulose sulfuric acid (pp. 1249-1252) https://doi.org/10.1016/j.crci.2009.01.006
- Cai et al. (2008) Gallium(III) triflate-catalyzed synthesis of quinoxaline derivatives (pp. 7386-7390) https://doi.org/10.1016/j.tetlet.2008.10.058

 10.1186/2193-8865-3-38
10.1186/2193-8865-3-38