Protective effect of melanin nanoparticles created from squid ink against irradiation on human keratinocytes
- Department of Biochemistry and Molecular Biology, Faculty of Biology, VNU University of Science, Vietnam National University, Hanoi, VN
- Center of Applied Sciences, Regenerative Medicine and Advance Technologies (CARA)–Vinmec Healthcare System, Hanoi, VN
- Department of Practical and Experimental Surgery, Vietnam Military Medical University, Hanoi, VN
- Department of Biochemistry and Molecular Biology, Faculty of Biology, VNU University of Science, Vietnam National University, Hanoi, VN National Key Laboratory of Enzyme and Protein Technology, VNU University of Science, Vietnam National University, Hanoi, VN

Published in Issue 20-09-2022

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Nguyen Thi, L. N., Le Duc, S., Bui Thi, V. K., Dinh Thi, T. T., Do Xuan, H., Hoang Thi, M. N., & Nguyen Dinh, T. (2022). Protective effect of melanin nanoparticles created from squid ink against irradiation on human keratinocytes. Journal of Nanostructure in Chemistry, 14(3 (June 2024). https://doi.org/10.1007/s40097-022-00513-1
Abstract
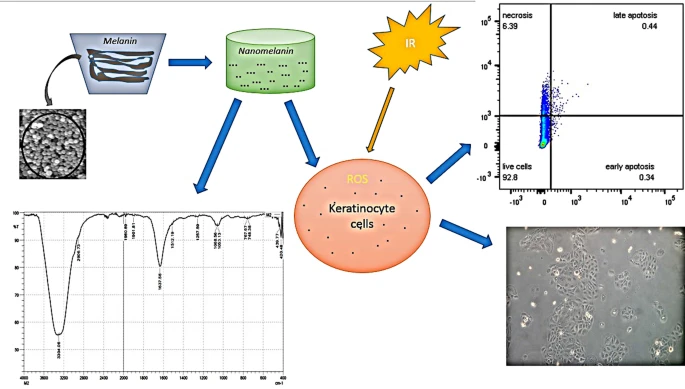
Abstract Because the skin is the first organ to be impacted by radiation, keratinocytes (HaCaT cells) are frequently used as an in vitro model for testing potential radioprotective agents. Melanin extracted from squid ink was considered an antioxidant compound and a possible radioprotector. In this study, nanomelanin was prepared at a size of 100–200 nm from isolated squid melanin. The zeta potential of melanin nanoparticles and the functional groups on their surface were investigated. The antioxidant activity of nanomelanin was examined by the DPPH method. Keratinocyte cells were incubated with or without melanin particles for 24 h before being irradiated in various doses. The radioprotective properties of nanomelanin after 3 days post-radiation were examined by cell survival rates. Treatment with nanomelanin enhances the percentage of cell viability by 10%, depending on the dose of radiation. At 2 days post-radiation, cell groups treated with nanomelanin were reduced to the transcription levels of gene coding BAX, TNF-α, and Caspase 3 for the cellular apoptosis process at various doses (3–5 Gy) of radiotherapy in comparison with control and eliminated the apoptotic cell rate as analyzed by flow cytometry methods. Furthermore, at 3 Gy, melanin nanoparticles promoted the transcription level gene coding SOD1 enzyme by 3.4 folds compared to control. Graphical abstractKeywords
- Nanomelanin,
- Keratinocyte cells,
- X-ray exposure,
- Squid ink
References
- Sung et al. (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries (pp. 209-249) https://doi.org/10.3322/caac.21660
- Sample and He (2018) Mechanisms and prevention of UV-induced melanoma (pp. 13-24) https://doi.org/10.1111/phpp.12329
- Tuieng et al. (2021) The Effects of ionising and non-ionising electromagnetic radiation on extracellular matrix proteins https://doi.org/10.3390/cells10113041
- Wang et al. (2018) Molecular mechanism of bystander effects and related abscopal/cohort effects in cancer therapy (pp. 18637-18647) https://doi.org/10.18632/oncotarget.24746
- Suzuki and Yamashita (2014) Radiation-induced bystander response: mechanism and clinical implications (pp. 16-24) https://doi.org/10.1089/wound.2013.0468
- Rosen et al. (2014) New approaches to radiation protection
- Mun et al. (2018) Pharmacology of natural radioprotectors (pp. 1033-1050) https://doi.org/10.1007/s12272-018-1083-6
- Solano (2017) Melanin and melanin-related polymers as materials with biomedical and biotechnological applications-cuttlefish ink and mussel foot proteins as inspired biomolecules https://doi.org/10.3390/ijms18071561
- Tran-Ly et al. (2020) Microbial production of melanin and its various applications https://doi.org/10.1007/s11274-020-02941-z
- Guo et al. (2014) Preparation of water-soluble melanin from squid ink using ultrasound-assisted degradation and its anti-oxidant activity (pp. 3680-3690) https://doi.org/10.1007/s13197-013-0937-7
- Vasileiou and Summerer (2020) A biomimetic approach to shielding from ionizing radiation: The case of melanized fungi https://doi.org/10.1371/journal.pone.0229921
- Savjani et al. (2012) Drug solubility: importance and enhancement techniques https://doi.org/10.5402/2012/195727
- Le Na, N.T., Van Khanh, B.T., Xuan Huy, P., Nhung, H.T.M., Thi Van Anh, N., Thang, N.D.: Melanin biomaterial effectively eliminates bacteria from water and synergistically induces melanoma-cell death during X-ray irradiation. Mater. Technol.
- 36
- , 261–269 (2021)
- Ryu and Chung (2016) Fucoidan reduces oxidative stress by regulating the gene expression of HO-1 and SOD-1 through the Nrf2/ERK signaling pathway in HaCaT cells (pp. 3255-3260) https://doi.org/10.3892/mmr.2016.5623
- Wang et al. (2020) Anti-Photoaging and anti-melanogenesis effects of fucoidan isolated from hizikia fusiforme and its underlying mechanisms https://doi.org/10.3390/md18080427
- Tang et al. (2022) Genistein protects against ultraviolet B-induced wrinkling and photoinflammation in in vitro and in vivo models https://doi.org/10.1186/s12263-022-00706-x
- Le Na, N.T., Duc Loc, S., Minh Tri, N.L., Bich Loan, N.T., Anh Son, H., Linh Toan, N., Phuong Thu, H., My Nhung, H.T., Lai Thanh, N., Van Anh, N.T., Dinh Thang, N.: Nanomelanin Potentially Protects the Spleen from Radiotherapy-Associated Damage and Enhances Immunoactivity in Tumor-Bearing Mice. Materials (Basel).
- 12
- , 1725 (2019)
- Gabriele, V.R., Mazhabi, R.M., Alexander, N., Mukherjee, P., Seyfried, T.N., Nwaji, N., Akinoglu, E.M., Mackiewicz. A., Zhou, G., Giersig, M., Naughton, M.J., Kempa, K.: Light- and Melanin Nanoparticle-Induced Cytotoxicity in Metastatic Cancer Cells. Pharmaceutics.
- 13
- , 965 (2021)
- Orellana and Kasinski (2016) Sulforhodamine B (SRB) Assay in Cell Culture to Investigate Cell Proliferation https://doi.org/10.21769/BioProtoc.1984
- Cuong et al. (2018) Melanin-embedded materials effectively remove hexavalent chromium (Cr(VI)) from aqueous solution https://doi.org/10.1186/s12199-018-0699-y
- Liu, H., Yang, Y., Liu, Y., Pan, J., Wang, J., Man, F., Zhang, W., Liu, G.: Melanin-Like Nanomaterials for Advanced Biomedical Applications: A Versatile Platform with Extraordinary Promise. Adv. Sci. (Weinh).
- 7
- , 1903129 (2020)
- Polapally et al. (2022) Melanin pigment of Streptomyces puniceus RHPR9 exhibits antibacterial, antioxidant and anticancer activities https://doi.org/10.1371/journal.pone.0266676
- Kumar et al. (2011) Physicochemical characterization and antioxidant activity of melanin from a novel strain of Aspergillus bridgeri ICTF-201 (pp. 350-358) https://doi.org/10.1111/j.1472-765X.2011.03116.x
- Cecchi et al. (2020) On the antioxidant activity of eumelanin biopigments: a quantitative comparison between free radical scavenging and redox properties (pp. 2465-2473) https://doi.org/10.1080/14786419.2018.1542391
- Wang et al. (2014) Effectiveness of treatment of iron deficiency anemia in rats with squid ink melanin-Fe (pp. 123-128) https://doi.org/10.1039/C3FO60383K
- Derby (2014) Cephalopod ink: production, chemistry, functions and applications (pp. 2700-2730) https://doi.org/10.3390/md12052700
- Amparyup et al. (2013) Prophenoloxidase system and its role in shrimp immune responses against major pathogens (pp. 990-1001) https://doi.org/10.1016/j.fsi.2012.08.019
- El-Naggar and El-Ewasy (2017) Bioproduction, characterization, anticancer and antioxidant activities of extracellular melanin pigment produced by newly isolated microbial cell factories Streptomyces glaucescens NEAE-H https://doi.org/10.1038/srep42129
- Oh et al. (2021) Fungal melanin as a biocompatible broad-spectrum sunscreen with high antioxidant activity (pp. 19682-19689) https://doi.org/10.1039/D1RA02583J
- Wu et al. (2021) Melanin-loaded CpG DNA hydrogel for modulation of tumor immune microenvironment (pp. 540-553) https://doi.org/10.1016/j.jconrel.2020.12.040
- Solano (2020) Photoprotection and Skin Pigmentation: Melanin-Related Molecules and Some Other New Agents Obtained from Natural Sources https://doi.org/10.3390/molecules25071537
- Schweitzer et al. (2010) Melanin-covered nanoparticles for protection of bone marrow during radiation therapy of cancer (pp. 1494-1502) https://doi.org/10.1016/j.ijrobp.2010.02.020
- Solano, F.: Melanins: Skin Pigments and Much More—Types, Structural Models, Biological Functions, and Formation Routes. New J. Sci. 498276, (2014)
- Vijay, K., SinghDivision, Thomas, M. Seed: The efficacy and safety of amifostine for the acute radiation syndrome. Expert Opin Drug Saf.
- 18
- , 1077–1090 (2019)
- Crook et al. (2021) Radiation exposure induces cross-species temporal metabolic changes that are mitigated in mice by amifostine https://doi.org/10.1038/s41598-021-93401-7
- Park et al. (2019) Recent advances in melanin-like nanomaterials in biomedical applications: a mini review https://doi.org/10.1186/s40824-019-0175-9
- Hein et al. (2014) Radiation-induced signaling pathways that promote cancer cell survival (review) (pp. 1813-1819) https://doi.org/10.3892/ijo.2014.2614
- Baskar et al. (2012) Cancer and radiation therapy: current advances and future directions (pp. 193-199) https://doi.org/10.7150/ijms.3635
- Hanahan and Weinberg (2011) Hallmarks of cancer: the next generation (pp. 646-674) https://doi.org/10.1016/j.cell.2011.02.013
- Golden et al. (2012) The convergence of radiation and immunogenic cell death signaling pathways https://doi.org/10.3389/fonc.2012.00088
- Wang et al. (2008) TNF-alpha induces two distinct caspase-8 activation pathways (pp. 693-703) https://doi.org/10.1016/j.cell.2008.03.036
- Qin et al. (2017) Apoptosis and injuries of heavy ion beam and x-ray radiation on malignant melanoma cell (pp. 953-960) https://doi.org/10.1177/1535370216689827
- Yahyapour et al. (2018) Radiation Protection and Mitigation by Natural Antioxidants and Flavonoids: Implications to Radiotherapy and Radiation Disasters (pp. 285-304) https://doi.org/10.2174/1874467211666180619125653
- Kma and Baruah (2022) The interplay of ROS and the PI3K/Akt pathway in autophagy regulation (pp. 248-264) https://doi.org/10.1002/bab.2104
- Zou et al. (2012) Extracellular superoxide dismutase is important for hippocampal neurogenesis and preservation of cognitive functions after irradiation (pp. 21522-21527) https://doi.org/10.1073/pnas.1216913110
- Sonis (2021) Superoxide Dismutase as an Intervention for Radiation Therapy-Associated Toxicities: Review and Profile of Avasopasem Manganese as a Treatment Option for Radiation-Induced Mucositis (pp. 1021-1029) https://doi.org/10.2147/DDDT.S267400
- Greenberger et al. (2021) Gene Therapy for Systemic or Organ Specific Delivery of Manganese Superoxide Dismutase https://doi.org/10.3390/antiox10071057
- Liu et al. (2021) Multifaceted roles of a bioengineered nanoreactor in repressing radiation-induced lung injury https://doi.org/10.1016/j.biomaterials.2021.121103

 10.1007/s40097-022-00513-1
10.1007/s40097-022-00513-1