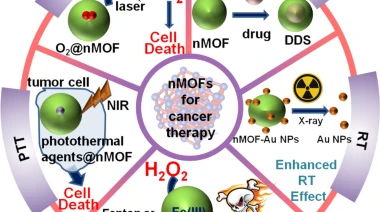
Nanoscale metal–organic frameworks as smart nanocarriers for cancer therapy
- School of Environment, Nanjing Normal University, Nanjing, 210023, CN
- Department of Orthopedic Surgery, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, CN
- Analytical and Testing Center, Nanjing Normal University, Nanjing, 210023, CN
- College of Chemistry and Materials Science, Nanjing Normal University, Nanjing, 210023, CN

Published in Issue 01-06-2022

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Liu, Y., Lei, P., Liao, X., & Wang, C. (2022). Nanoscale metal–organic frameworks as smart nanocarriers for cancer therapy. Journal of Nanostructure in Chemistry, 14(1 (February 2024). https://doi.org/10.1007/s40097-022-00493-2
Abstract
Abstract Cancer is one of the serious diseases to human life. Early and precise cancer diagnosis and timely therapy are in urgent need nowadays. Due to the advantages of porous structures and tunable properties, metal–organic frameworks (MOFs) are becoming type of rapidly developing and attractive supports used in biomedicine, which have been widely applied in the fields of chemistry, biology, materials science, etc. Particularly, nanoscale MOFs (nMOFs) with more accessible active sites and improved stability are ideal platforms for biological and clinical applications in vitro and in vivo. This review article summarizes the recent progresses in nMOFs based nanoplatforms for drug delivery and cancer therapy. Different techniques using nMOFs are systematically summarized including chemotherapy, photodynamic therapy (PDT), photothermal therapy (PTT), chemodynamic therapy (CDT), radiotherapy (RT), and the combined therapy methods. Finally, a brief conclusion and outlook for biomedical applications of this special field is provided. We expect this review could be helpful for future designing and fabrication of multi-functional nMOFs platforms for drug delivery, disease therapy, and other biomedical applications.Keywords
- Nanoscale metal–organic frameworks (nMOFs),
- Nanocarrier,
- Drug delivery,
- Cancer therapy,
- Biomedicine
References
- Coussens and Werb (2002) Inflammation and cancer (pp. 860-867) https://doi.org/10.1038/nature01322
- Zhang et al. (2020) Cancer diagnosis with DNA molecular computation (pp. 709-715) https://doi.org/10.1038/s41565-020-0699-0
- Li et al. (1999) Design and synthesis of an exceptionally stable and highly porous metal-organic framework (pp. 276-279) https://doi.org/10.1038/46248
- Jin et al. (2021) Redox-responsive micelles integrating catalytic nanomedicine and selective chemotherapy for effective tumor treatment (pp. 3076-3082) https://doi.org/10.1016/j.cclet.2021.03.084
- Yang et al. (2021) Molecular-scale drug delivery systems loaded with oxaliplatin for supramolecular chemotherapy (pp. 729-734) https://doi.org/10.1016/j.cclet.2020.08.035
- Anbia and Faryadras (2015) In situ Na center dot Cu-3(BTC)(2) and Li center dot Cu-3(BTC)(2) nanoporous MOFs synthesis for enhancing H-2 storage at ambient temperature (pp. 357-364) https://doi.org/10.1007/s40097-015-0167-9
- Yang et al. (2019) Boosting the photodynamic therapy efficiency with a mitochondria-targeted nanophotosensitizer (pp. 1293-1296) https://doi.org/10.1016/j.cclet.2019.03.032
- Ghiamaty et al. (2016) Synthesis of palladium-carbon nanotube-metal organic framework composite and its application as electrocatalyst for hydrogen production (pp. 299-308) https://doi.org/10.1007/s40097-016-0203-4
- Wu et al. (2020) A photosensitizer-loaded zinc oxide-polydopamine core-shell nanotherapeutic agent for photodynamic and photothermal synergistic therapy of cancer cells (pp. 189-192) https://doi.org/10.1016/j.cclet.2019.05.004
- Rodriguez et al. (2020) Synthesis of highly stable κ/ι-hybrid carrageenan micro- and nanogels via a sonication-assisted microemulsion route (pp. 69-82)
- Makvandi et al. (2021) Endocytosis of abiotic nanomaterials and nanobiovectors: inhibition of membrane trafficking https://doi.org/10.1016/j.nantod.2021.101279
- Gulla et al. (2021) Titanium dioxide nanotubes conjugated with quercetin function as an effective anticancer agent by inducing apoptosis in melanoma cells (pp. 721-734) https://doi.org/10.1007/s40097-021-00396-8
- Tabasi et al. (2021) Metal–polymer-coordinated complexes as potential nanovehicles for drug delivery https://doi.org/10.1007/s40097-021-00432-7
- Eivazzadeh-Keihan and Maleki (2021) Design and synthesis of a new magnetic aromatic organo-silane star polymer with unique nanoplate morphology and hyperthermia application https://doi.org/10.1007/s40097-021-00401-0
- Nasri et al. (2020) Thymoquinone-loaded ethosome with breast cancer potential: optimization, in vitro and biological assessment (pp. 19-31) https://doi.org/10.1007/s40097-019-00325-w
- Kong et al. (2020) A green-emission metal-organic framework-based nanoprobe for imaging dual tumor biomarkers in living cells (pp. 35375-35384) https://doi.org/10.1021/acsami.0c10038
- Ma et al. (2019) Functional metal organic framework/SiO2 nanocomposites: from versatile synthesis to advanced applications https://doi.org/10.3390/polym11111823
- Liu et al. (2020) Core-shell nanosystems for self-activated drug-gene combinations against triple-negative breast cancer (pp. 53654-53664) https://doi.org/10.1021/acsami.0c15089
- Shu et al. (2020) Stretchable electrochemical biosensing platform based on Ni-MOF composite/Au nanoparticle-coated carbon nanotubes for real-time monitoring of dopamine released from living cells (pp. 49480-49488) https://doi.org/10.1021/acsami.0c16060
- Hu et al. (2021) Cytotoxicity of aptamer-conjugated chitosan encapsulated mycogenic gold nanoparticles in human lung cancer cells https://doi.org/10.1007/s40097-021-00437-2
- Rehman et al. (2020) Investigation of ROS scavenging properties and in vitro cytotoxicity of oxygen-deficient La2O3−x nanostructure synthesized by spray pyrolysis method (pp. 347-361) https://doi.org/10.1007/s40097-020-00356-8
- Yang et al. (2012) Functionalized mesoporous silica materials for controlled drug delivery (pp. 3679-3698) https://doi.org/10.1039/c2cs15308d
- Tabasi et al. (2021) Metal-polymer-coordinated complexes as potential nanovehicles for drug delivery (pp. 501-526) https://doi.org/10.1007/s40097-021-00432-7
- Xu et al. (2021) Recent advances in nanoscale metal-organic frameworks biosensors for detection of biomarkers https://doi.org/10.1016/j.cclet.2021.06.015
- Duman and Forgan (2021) Applications of nanoscale metal-organic frameworks as imaging agents in biology and medicine (pp. 3423-3449) https://doi.org/10.1039/D1TB00358E
- Zhang et al. (2020) Metal-organic framework-based nanomaterials for biomedical applications (pp. 1060-1070) https://doi.org/10.1016/j.cclet.2019.11.036
- Rojas et al. (2017) Metal organic frameworks based on bioactive components (pp. 2560-2573) https://doi.org/10.1039/C6TB03217F
- Castillo-Blas et al. (2020) The role of defects in the properties of functional coordination polymers (pp. 73-119) https://doi.org/10.1016/bs.adioch.2020.03.002
- Peng et al. (2012) Surfactant-directed assembly of mesoporous metal-organic framework nanoplates in ionic liquids (pp. 8688-8690) https://doi.org/10.1039/c2cc34416e
- Zheng et al. (2017) Controllable preparation of nanoscale metal-organic frameworks by ionic liquid microemulsions (pp. 5899-5905) https://doi.org/10.1021/acs.iecr.7b00694
- Zhang et al. (2020) pH-Responsive metal-organic framework encapsulated gold nanoclusters with modulated release to enhance photodynamic therapy/chemotherapy in breast cancer (pp. 1739-1747) https://doi.org/10.1039/C9TB02621E
- Zhang et al. (2019) A non-enzymatic glucose sensor with enhanced anti-interference ability based on a MIL-53(NiFe) metal-organic framework (pp. 7006-7013) https://doi.org/10.1039/C9TB01832H
- Huang et al. (2021) Nanoscale metal-organic frameworks for tumor phototherapy (pp. 3756-3777) https://doi.org/10.1039/D1TB00349F
- Lei et al. (2018) Constructing redox-responsive metal–organic framework nanocarriers for anticancer drug delivery (pp. 16698-16706) https://doi.org/10.1021/acsami.7b19693
- He et al. (2019) A catalase-like metal-organic framework nanohybrid for O2-evolving synergistic chemoradiotherapy (pp. 8752-8756) https://doi.org/10.1002/anie.201902612
- Su et al. (2021) Determination of acetamiprid by fluorescence monitoring of a glycine-l-histidine copper-organic framework aptasensor https://doi.org/10.1080/00032719.2021.1946555
- Cai et al. (2019) Biological metal-organic frameworks: Structures, host-guest chemistry and bio-applications (pp. 207-221) https://doi.org/10.1016/j.ccr.2017.12.003
- Zhu and Liu (2019) The synthetic strategies of metal-organic framework membranes, films and 2D MOFs and their applications in devices (pp. 21004-21035) https://doi.org/10.1039/C9TA05383B
- He et al. (2019) Recent progress in nanoscale metal-organic frameworks for drug release and cancer therapy (pp. 1343-1365) https://doi.org/10.2217/nnm-2018-0347
- Zhou et al. (2018) Nanoscaled metal-organic frameworks for biosensing, imaging, and cancer therapy https://doi.org/10.1002/adhm.201800022
- Zhang et al. (2016) Centromere and kinetochore gene misexpression predicts cancer patient survival and response to radiotherapy and chemotherapy https://doi.org/10.1038/ncomms12619
- Ren et al. (2014) Polyacrylic acid@zeolitic imidazolate framework-8 nanoparticles with ultrahigh drug loading capability for pH-sensitive drug release (pp. 1000-1002) https://doi.org/10.1039/C3CC47666A
- Zhu et al. (2021) Synergistic effects of rapamycin and fluorouracil to treat a gastric tumor in a PTEN conditional deletion mouse model https://doi.org/10.1007/s10120-021-01229-x
- Kiss et al. (2021) Targeting DNA damage response and repair to enhance therapeutic index in cisplatin-based cancer treatment https://doi.org/10.3390/ijms22158199
- Zheng et al. (2021) Self-assembled pH-sensitive nanoparticles based on ganoderma lucidum polysaccharide-methotrexate conjugates for the co-delivery of anti-tumor drugs (pp. 3764-3773) https://doi.org/10.1021/acsbiomaterials.1c00663
- Allen and Cullis (2004) Drug delivery systems: entering the mainstream (pp. 1818-1822) https://doi.org/10.1126/science.1095833
- Sosnik and Menaker Raskin (2015) Polymeric micelles in mucosal drug delivery: challenges towards clinical translation (pp. 1380-1392) https://doi.org/10.1016/j.biotechadv.2015.01.003
- Du et al. (2015) Regulating the surface poly(ethylene glycol) density of polymeric nanoparticles and evaluating its role in drug delivery in vivo (pp. 1-11) https://doi.org/10.1016/j.biomaterials.2015.07.048
- Gong et al. (2019) Highly fluorescent N-doped carbon dots with two-photon emission for ultrasensitive detection of tumor marker and visual monitor anticancer drug loading and delivery (pp. 994-1002) https://doi.org/10.1016/j.cej.2018.09.100
- Wang et al. (2015) Mesoporous silica nanoparticles in drug delivery and biomedical applications (pp. 313-327) https://doi.org/10.1016/j.nano.2014.09.014
- Horcajada et al. (2008) Flexible porous metal-organic frameworks for a controlled drug delivery (pp. 6774-6780) https://doi.org/10.1021/ja710973k
- Horcajada et al. (2010) Porous metal-organic-framework nanoscale carriers as a potential platform for drug delivery and imaging (pp. 172-178) https://doi.org/10.1038/nmat2608
- Cai and Jiang (2017) A modulator-induced defect-formation strategy to hierarchically porous metal-organic frameworks with high stability (pp. 563-567) https://doi.org/10.1002/anie.201610914
- Wang et al. (2020) Metal-organic frameworks for stimuli-responsive drug delivery https://doi.org/10.1016/j.biomaterials.2019.119619
- Orellana-Tavra et al. (2015) Amorphous metal-organic frameworks for drug delivery (pp. 13878-13881) https://doi.org/10.1039/C5CC05237H
- Teplensky et al. (2017) Temperature treatment of highly porous zirconium-containing metal–organic frameworks extends drug delivery release (pp. 7522-7532) https://doi.org/10.1021/jacs.7b01451
- Wang et al. (2019) MOFBOTS: metal–organic-framework-based biomedical microrobots https://doi.org/10.1002/adma.201901592
- Liang et al. (2018) A protein@metal-organic framework nanocomposite for pH-triggered anticancer drug delivery (pp. 10223-10228) https://doi.org/10.1039/C8DT01789A
- Jia et al. (2019) A γ-cyclodextrin-based metal-organic framework embedded with graphene quantum dots and modified with PEGMA via SI-ATRP for anticancer drug delivery and therapy (pp. 20956-20967) https://doi.org/10.1039/C9NR06195A
- Kahn et al. (2017) Stimuli-responsive DNA-functionalized metal-organic frameworks (MOFs) https://doi.org/10.1002/adma.201602782
- Mura et al. (2013) Stimuli-responsive nanocarriers for drug delivery (pp. 991-1003) https://doi.org/10.1038/nmat3776
- Liu et al. (2019) In situ polymerization on nanoscale metal-organic frameworks for enhanced physiological stability and stimulus-responsive intracellular drug delivery https://doi.org/10.1016/j.biomaterials.2019.119365
- Min et al. (2019) Biomimetic metal–organic framework nanoparticles for cooperative combination of antiangiogenesis and photodynamic therapy for enhanced efficacy https://doi.org/10.1002/adma.201808200
- Ranji-Burachaloo et al. (2019) Combined fenton and starvation therapies using hemoglobin and glucose oxidase (pp. 5705-5716) https://doi.org/10.1039/C8NR09107B
- Kim et al. (2019) MOF × biopolymer: collaborative combination of metal-organic framework and biopolymer for advanced anticancer therapy (pp. 27512-27520) https://doi.org/10.1021/acsami.9b05736
- Yang et al. (2019) Nanoscale ATP-responsive zeolitic imidazole framework-90 as a general platform for cytosolic protein delivery and genome editing (pp. 3782-3786) https://doi.org/10.1021/jacs.8b11996
- Chen et al. (2017) ATP-responsive aptamer-based metal-organic framework nanoparticles (NMOFs) for the controlled release of loads and drugs https://doi.org/10.1002/adfm.201702102
- Ma et al. (2017) H2S-activable MOF nanoparticle photosensitizer for effective photodynamic therapy against cancer with controllable singlet-oxygen release (pp. 13752-13756) https://doi.org/10.1002/anie.201708005
- Cheung and Li (2021) Development of self-care textile wearables with thermally stimulated drug delivery function via biological and physical investigations (pp. 820-827) https://doi.org/10.1177/0040517520960758
- Fernandez and Orozco (2021) Advances in functionalized photosensitive polymeric nanocarriers https://doi.org/10.3390/polym13152464
- Wu et al. (2021) Pressure-driven spreadable deferoxamine-laden hydrogels for vascularized skin flaps dagger (pp. 3162-3170) https://doi.org/10.1039/D1BM00053E
- Jiang et al. (2017) Thermal stimuli-triggered drug release from a biocompatible porous metal–organic framework (pp. 10215-10221) https://doi.org/10.1002/chem.201701904
- Roth Stefaniak et al. (2018) Photo-triggered release of 5-fluorouracil from a MOF drug delivery vehicle (pp. 7617-7620) https://doi.org/10.1039/C8CC01601A
- Lin et al. (2020) Acetaldehyde-modified-cystine functionalized Zr-MOFs for pH/GSH dual-responsive drug delivery and selective visualization of GSH in living cells (pp. 3084-3091) https://doi.org/10.1039/C9RA05741B
- Li et al. (2018) Coordination-responsive drug release inside gold nanorod@metal-organic framework core–shell nanostructures for near-infrared-induced synergistic chemo-photothermal therapy (pp. 3294-3305) https://doi.org/10.1007/s12274-017-1874-y
- Ng and Zheng (2015) Molecular interactions in organic nanoparticles for phototheranostic applications (pp. 11012-11042) https://doi.org/10.1021/acs.chemrev.5b00140
- Yoon et al. (2013) Advance in photosensitizers and light delivery for photodynamic therapy (pp. 7-23) https://doi.org/10.5946/ce.2013.46.1.7
- Lovell et al. (2010) Activatable photosensitizers for imaging and therapy (pp. 2839-2857) https://doi.org/10.1021/cr900236h
- Agostinis et al. (2011) Photodynamic therapy of cancer: an update (pp. 250-281) https://doi.org/10.3322/caac.20114
- Chatterjee et al. (2008) Nanoparticles in photodynamic therapy: an emerging paradigm (pp. 1627-1637) https://doi.org/10.1016/j.addr.2008.08.003
- Dolmans et al. (2003) Photodynamic therapy for cancer (pp. 380-387) https://doi.org/10.1038/nrc1071
- Lismont et al. (2017) Metal-organic framework nanoparticles in photodynamic therapy: current status and perspectives https://doi.org/10.1002/adfm.201606314
- Secret et al. (2014) Two-photon excitation of porphyrin-functionalized porous silicon nanoparticles for photodynamic therapy (pp. 7643-7648) https://doi.org/10.1002/adma.201403415
- Konan et al. (2002) State of the art in the delivery of photosensitizers for photodynamic therapy (pp. 89-106) https://doi.org/10.1016/S1011-1344(01)00267-6
- Chouikrat et al. (2012) Non-polymeric nanoparticles for photodynamic therapy applications: recent developments (pp. 781-792) https://doi.org/10.2174/092986712799034897
- Lu et al. (2014) Nanoscale metal–organic framework for highly effective photodynamic therapy of resistant head and neck cancer (pp. 16712-16715) https://doi.org/10.1021/ja508679h
- Lu et al. (2015) A chlorin-based nanoscale metal–organic framework for photodynamic therapy of colon cancers (pp. 7600-7603) https://doi.org/10.1021/jacs.5b04069
- Carvalho et al. (2013) Tetrabenzoporphyrins: synthetic developments and applications (pp. 3302-3317) https://doi.org/10.1039/c3cs35500d
- Liu et al. (2017) Dual-stage light amplified photodynamic therapy against hypoxic tumor based on an O2 self-sufficient nanoplatform https://doi.org/10.1002/smll.201701621
- Gao et al. (2018) Biomimetic O2-evolving metal-organic framework nanoplatform for highly efficient photodynamic therapy against hypoxic tumor (pp. 83-94) https://doi.org/10.1016/j.biomaterials.2018.06.007
- Liu et al. (2019) Metal–organic framework (MOF) hybrid as a tandem catalyst for enhanced therapy against hypoxic tumor cells (pp. 7808-7812) https://doi.org/10.1002/anie.201903475
- Zhang et al. (2018) Nanozyme decorated metal–organic frameworks for enhanced photodynamic therapy (pp. 651-661) https://doi.org/10.1021/acsnano.7b07746
- Zheng et al. (2016) Carbon-dot-decorated carbon nitride nanoparticles for enhanced photodynamic therapy against hypoxic tumor via water splitting (pp. 8715-8722) https://doi.org/10.1021/acsnano.6b04156
- Cairns et al. (2011) Regulation of cancer cell metabolism (pp. 85-95) https://doi.org/10.1038/nrc2981
- Chen et al. (2019) Extracellular vesicle-packaged HIF-1α-stabilizing lncRNA from tumour-associated macrophages regulates aerobic glycolysis of breast cancer cells (pp. 498-510) https://doi.org/10.1038/s41556-019-0299-0
- Li et al. (2017) Cancer cell membrane camouflaged cascade bioreactor for cancer targeted starvation and photodynamic therapy (pp. 7006-7018) https://doi.org/10.1021/acsnano.7b02533
- Chen et al. (2016) Lactate metabolism is associated with mammalian mitochondria (pp. 937-943) https://doi.org/10.1038/nchembio.2172
- Chen et al. (2018) Interfering with lactate-fueled respiration for enhanced photodynamic tumor therapy by a porphyrinic MOF nanoplatform https://doi.org/10.1002/adfm.201803498
- He et al. (2020) Solvent-assisted self-assembly of a metal–organic framework based biocatalyst for cascade reaction driven photodynamic therapy (pp. 6822-6832) https://doi.org/10.1021/jacs.0c02497
- Cramer et al. (2017) Systemic depletion of l-cyst(e)ine with cyst(e)inase increases reactive oxygen species and suppresses tumor growth (pp. 120-127) https://doi.org/10.1038/nm.4232
- Cheng et al. (2019) Nanotherapeutics interfere with cellular redox homeostasis for highly improved photodynamic therapy https://doi.org/10.1016/j.biomaterials.2019.119500
- Zhang et al. (2018) Enhanced photodynamic therapy by reduced levels of intracellular glutathione obtained by employing a nano-MOF with CuII as the active center (pp. 4891-4896) https://doi.org/10.1002/anie.201710800
- Wang et al. (2019) Specific generation of singlet oxygen through the Russell mechanism in hypoxic tumors and GSH depletion by Cu-TCPP nanosheets for cancer therapy (pp. 9846-9850) https://doi.org/10.1002/anie.201903981
- Miyamoto et al. (2014) Singlet molecular oxygen generated by biological hydroperoxides (pp. 24-33) https://doi.org/10.1016/j.jphotobiol.2014.03.028
- Melancon et al. (2011) Cancer theranostics with near-infrared light-activatable multimodal nanoparticles (pp. 947-956) https://doi.org/10.1021/ar200022e
- Dykman and Khlebtsov (2012) Gold nanoparticles in biomedical applications: recent advances and perspectives (pp. 2256-2282) https://doi.org/10.1039/C1CS15166E
- Tian et al. (2011) Hydrophilic flower-like CuS superstructures as an efficient 980 nm laser-driven photothermal agent for ablation of cancer cells (pp. 3542-3547) https://doi.org/10.1002/adma.201101295
- Yang et al. (2013) Nano-graphene in biomedicine: theranostic applications (pp. 530-547) https://doi.org/10.1039/C2CS35342C
- Zhu et al. (2016) PPy@MIL-100 nanoparticles as a pH- and near-IR-irradiation-responsive drug carrier for simultaneous photothermal therapy and chemotherapy of cancer cells (pp. 34209-34217) https://doi.org/10.1021/acsami.6b11378
- Jiang et al. (2018) CuS@MOF-based well-designed quercetin delivery system for chemo–photothermal therapy (pp. 34513-34523) https://doi.org/10.1021/acsami.8b13487
- Keerthiga et al. (2020) Photodynamic nanophotosensitizers: promising materials for tumor theranostics (pp. 5474-5485) https://doi.org/10.1021/acsbiomaterials.0c01058
- Wang et al. (2018) In situ one-pot synthesis of MOF–polydopamine hybrid nanogels with enhanced photothermal effect for targeted cancer therapy https://doi.org/10.1002/advs.201800287
- Li et al. (2018) Ultrathin Cu-TCPP MOF nanosheets: a new theragnostic nanoplatform with magnetic resonance/near-infrared thermal imaging for synergistic phototherapy of cancers (pp. 4086-4096) https://doi.org/10.7150/thno.25433
- Zhang et al. (2016) Synthesis of iron nanometallic glasses and their application in cancer therapy by a localized fenton reaction (pp. 2101-2106) https://doi.org/10.1002/anie.201510031
- Tang et al. (2019) Chemodynamic therapy: tumour microenvironment-mediated fenton and fenton-like reactions (pp. 946-956) https://doi.org/10.1002/anie.201805664
- Zhang et al. (2018) An adenosine triphosphate-responsive autocatalytic fenton nanoparticle for tumor ablation with self-supplied H2O2 and acceleration of Fe(III)/Fe(II) conversion (pp. 7609-7618) https://doi.org/10.1021/acs.nanolett.8b03178
- Yu et al. (2021) Hydrogen peroxide-generating nanomedicine for enhanced chemodynamic therapy (pp. 2127-2138) https://doi.org/10.1016/j.cclet.2021.02.015
- Liou and Storz (2010) Reactive oxygen species in cancer (pp. 479-496) https://doi.org/10.3109/10715761003667554
- Zhang et al. (2019) Positive feedback nanoamplifier responded to tumor microenvironments for self-enhanced tumor imaging and therapy https://doi.org/10.1016/j.biomaterials.2019.119255
- Wang et al. (2016) Magnetically guided delivery of DHA and Fe ions for enhanced cancer therapy based on pH-responsive degradation of DHA-loaded Fe3O4@C@MIL-100(Fe) nanoparticles (pp. 88-101) https://doi.org/10.1016/j.biomaterials.2016.08.039
- Xue et al. (2019) Doxorubicin-loaded nanoscale metal–organic framework for tumor-targeting combined chemotherapy and chemodynamic therapy (pp. 4615-4623) https://doi.org/10.1039/C9BM01044K
- Wang et al. (2014) Ferrocene-catalyzed heterogeneous fenton-like degradation of methylene blue: influence of initial solution pH (pp. 6334-6340) https://doi.org/10.1021/ie500115j
- Ornelas (2011) Application of ferrocene and its derivatives in cancer research (pp. 1973-1985) https://doi.org/10.1039/c1nj20172g
- Jaouen et al. (2015) Ferrocifen type anticancer drugs (pp. 8802-8817) https://doi.org/10.1039/C5CS00486A
- Fang et al. (2020) Co–ferrocene MOF/glucose oxidase as cascade nanozyme for effective tumor therapy https://doi.org/10.1002/adfm.201910085
- Ranji-Burachaloo et al. (2017) MOF-mediated destruction of cancer using the cell’s own hydrogen peroxide (pp. 33599-33608) https://doi.org/10.1021/acsami.7b07981
- Brenner and Ward (1992) Constraints on energy deposition and target size of multiply damaged sites associated with DNA double-strand breaks (pp. 737-748) https://doi.org/10.1080/09553009214551591
- Song et al. (2017) Emerging nanotechnology and advanced materials for cancer radiation therapy https://doi.org/10.1002/adma.201700996
- Pallares and Abergel (2020) Nanoparticles for targeted cancer radiotherapy (pp. 2887-2897) https://doi.org/10.1007/s12274-020-2957-8
- Li et al. (2020) Nanoscale metal-organic frameworks: synthesis, biocompatibility, imaging applications, and thermal and dynamic therapy of tumors https://doi.org/10.1002/adfm.201908924
- Ma et al. (2019) Quercetin-modified metal–organic frameworks for dual sensitization of radiotherapy in tumor tissues by inhibiting the carbonic anhydrase IX (pp. 4209-4219) https://doi.org/10.1021/acsnano.8b09221
- Wang et al. (2014) Synergistic assembly of heavy metal clusters and luminescent organic bridging ligands in metal–organic frameworks for highly efficient X-ray scintillation (pp. 6171-6174) https://doi.org/10.1021/ja500671h
- Misawa and Takahashi (2011) Generation of reactive oxygen species induced by gold nanoparticles under X-ray and UV irradiations (pp. 604-614) https://doi.org/10.1016/j.nano.2011.01.014
- Song et al. (2017) TaOx decorated perfluorocarbon nanodroplets as oxygen reservoirs to overcome tumor hypoxia and enhance cancer radiotherapy (pp. 257-263) https://doi.org/10.1016/j.biomaterials.2016.10.020
- Zhu et al. (2018) Recent progress in the development of near-infrared organic photothermal and photodynamic nanotherapeutics (pp. 746-765) https://doi.org/10.1039/C7BM01210A
- Gong et al. (2014) Engineering of multifunctional nano-micelles for combined photothermal and photodynamic therapy under the guidance of multimodal imaging (pp. 6492-6502) https://doi.org/10.1002/adfm.201401451
- Gai et al. (2018) Recent advances in functional nanomaterials for light–triggered cancer therapy (pp. 146-187) https://doi.org/10.1016/j.nantod.2018.02.010
- Wang et al. (2019) Exploiting single atom iron centers in a porphyrin-like MOF for efficient cancer phototherapy (pp. 35228-35237) https://doi.org/10.1021/acsami.9b11238
- Zhang et al. (2018) Metal–organic framework nanoshuttle for synergistic photodynamic and low-temperature photothermal therapy https://doi.org/10.1002/adfm.201804634
- Liu et al. (2017) Multifunctional metal–organic framework nanoprobe for cathepsin B-activated cancer cell imaging and chemo-photodynamic therapy (pp. 2150-2158) https://doi.org/10.1021/acsami.6b14446
- Zhu et al. (2019) Two-dimensional metal-organic-framework as a unique theranostic nano-platform for nuclear imaging and chemo-photodynamic cancer therapy (pp. 1307-1312) https://doi.org/10.1007/s12274-018-2242-2
- Luo et al. (2019) Light-induced redox-responsive smart drug delivery system by using selenium-containing polymer@MOF shell/core nanocomposite https://doi.org/10.1002/adhm.201900406
- Feng et al. (2019) Stimuli-responsive multifunctional metal-organic framework nanoparticles for enhanced chemo-photothermal therapy (pp. 994-1004) https://doi.org/10.1039/C8TB02815J
- Zeng et al. (2018) Porphyrinic metal–organic frameworks coated gold nanorods as a versatile nanoplatform for combined photodynamic/photothermal/chemotherapy of tumor https://doi.org/10.1002/adfm.201705451
- Ni et al. (2018) Nanoscale metal-organic frameworks for mitochondria-targeted radiotherapy-radiodynamic therapy https://doi.org/10.1038/s41467-018-06655-7
- Cai et al. (2020) Photodynamic therapy combined with antihypoxic signaling and CpG adjuvant as an in situ tumor vaccine based on metal–organic framework nanoparticles to boost cancer immunotherapy https://doi.org/10.1002/adhm.201900996
- Shao et al. (2020) Engineering of upconverted metal–organic frameworks for near-infrared light-triggered combinational photodynamic/chemo-/immunotherapy against hypoxic tumors (pp. 3939-3946) https://doi.org/10.1021/jacs.9b12788
- Gautam et al. (2022) Drug delivery of paracetamol by metal-organic frameworks (HKUST-1): improvised synthesis and investigations https://doi.org/10.1016/j.mtchem.2021.100647
- Wen et al. (2021) Versatile nanoscale metal-organic frameworks (nMOFs): an emerging 3D nanoplatform for drug delivery and therapeutic applications https://doi.org/10.1002/smll.202005064
- Li et al. (2020) Nanoscale metal - organic frameworks: synthesis, biocompatibility, imaging applications, thermal and dynamic therapy of tumors https://doi.org/10.1002/adfm.201908924
- Liu et al. (2021) Multifunctional metal-organic framework heterostructures for enhanced cancer therapy (pp. 1188-1218) https://doi.org/10.1039/D0CS00178C
- Liu et al. (2021) A multifunctional aminated UiO-67 metal-organic framework for enhancing antitumor cytotoxicity through bimodal drug delivery https://doi.org/10.1016/j.cej.2020.127899
- Liu et al. (2021) Recent advances in cell membrane coated metal-organic frameworks (MOFs) for tumor therapy (pp. 4459-4474) https://doi.org/10.1039/D1TB00453K
- Sepehrpour et al. (2019) Biomedically relevant self-assembled metallacycles and metallacages (pp. 14005-14020) https://doi.org/10.1021/jacs.9b06222
- Sun et al. (2020) Recent developments in the construction and applications of platinum-based metallacycles and metallacages via coordination (pp. 3889-3919) https://doi.org/10.1039/D0CS00038H

 10.1007/s40097-022-00493-2
10.1007/s40097-022-00493-2