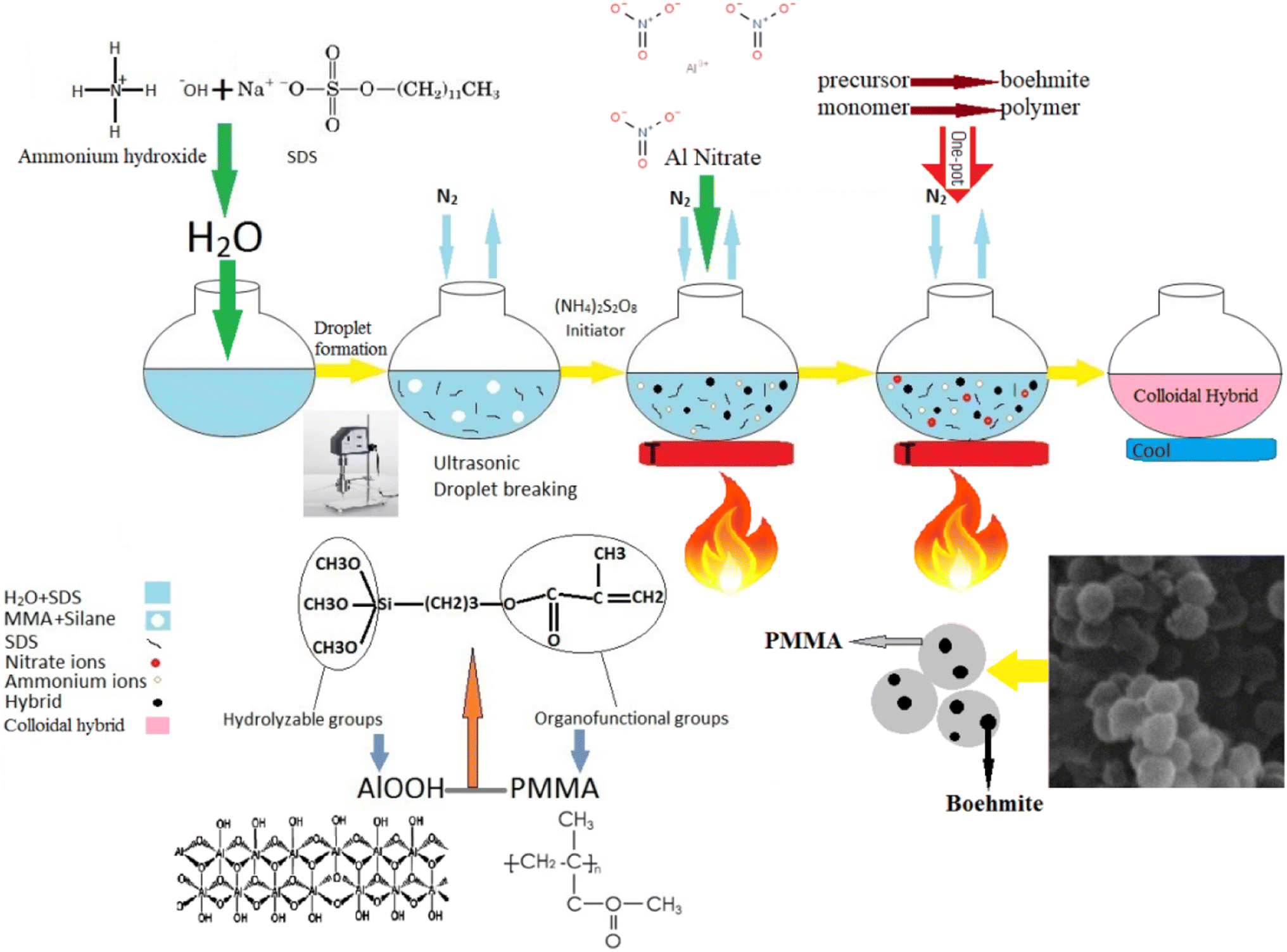
Aluminum oxyhydroxide-doped PMMA hybrids powder prepared via facile one-pot method towards copper ion removal from aqueous solution
- Nanotechnology Lab, Esfarayen University of Technology, Esfarayen, IR
- Department of Materials Engineering, The University of British Columbia, Vancouver, BC, V6T 1Z4, CA
- Department of Physics, The University of Western Australia, Perth, WA, 6009, AU
- Nanotechnology Lab, Esfarayen University of Technology, Esfarayen, IR Department of Physics, The University of Western Australia, Perth, WA, 6009, AU

Published in Issue 2019-11-07
How to Cite
Ghamari, M., Imani, A., Williams, J. F., & Ghasemifard, M. (2019). Aluminum oxyhydroxide-doped PMMA hybrids powder prepared via facile one-pot method towards copper ion removal from aqueous solution. International Nano Letters, 9(4 (December 2019). https://doi.org/10.1007/s40089-019-00286-6
HTML views: 98
PDF views: 101
Abstract
Highlights PMMA/aluminum oxyhydroxide hybrids act as a good Cu adsorbent The absorption behavior depends on hybrid morphology The higher surface area of hybrids provides a great capability to absorb Cu ions in aqueous mediaKeywords
- PMMA,
- Boehmite,
- Copper ion,
- Hybrid,
- Morphology
References
- Wang and Yang (2016) Industrial water pollution, water environment treatment, and health risks in China (pp. 358-365) https://doi.org/10.1016/j.envpol.2016.07.011
- Asgari Lajayer et al. (2018) Removal of heavy metals (Cu2+ and Cd2+) from effluent using gamma irradiation, titanium dioxide nanoparticles and methanol (pp. 483-496) https://doi.org/10.1007/s40097-018-0292-3
- Asgari Lajayer et al. (2017) Heavy metals in contaminated environment: destiny of secondary metabolite biosynthesis, oxidative status and phytoextraction in medicinal plants (pp. 377-390) https://doi.org/10.1016/j.ecoenv.2017.07.035
- Sadegh et al. (2017) The role of nanomaterials as effective adsorbents and their applications in wastewater treatment (pp. 1-14) https://doi.org/10.1007/s40097-017-0219-4
- Ali Sarhan (2015) Adsorption characteristics of copper(II) ions from aqueous solution onto ionic cross-linked Pva/P(Aam-Nipaam) core-shell nanogels (pp. 1-9) https://doi.org/10.4172/2157-7439.S7-001
- Tisato et al. (2009) Copper in diseases and treatments, and copper-based anticancer strategies https://doi.org/10.1002/med.20174
- Muhammad Ekramul Mahmud et al. (2016) The removal of heavy metal ions from wastewater/aqueous solution using polypyrrole-based adsorbents: a review 6(18) (pp. 14778-14791) https://doi.org/10.1039/C5RA24358K
- Ali et al. (2016) Potential of using green adsorbent of heavy metal removal from aqueous solutions: adsorption kinetics, isotherm, thermodynamic, mechanism and economic analysis (pp. 317-332) https://doi.org/10.1016/j.ecoleng.2016.03.015
- Khoshsang et al. (2018) One-pot synthesis of S-doped Fe2O3/C magnetic nanocomposite as an adsorbent for anionic dye removal: equilibrium and kinetic studies (pp. 23-32) https://doi.org/10.1007/s40097-017-0251-4
- Khandel and Shahi (2018) Mycogenic nanoparticles and their bio-prospective applications: current status and future challenges (pp. 369-391) https://doi.org/10.1007/s40097-018-0285-2
- Ghaemi and Daraei (2016) Enhancement in copper ion removal by PPy@Al2O3 polymeric nanocomposite membrane (pp. 26-33) https://doi.org/10.1016/j.jiec.2016.05.027
- Fouladgar et al. (2015) Single and binary adsorption of nickel and copper from aqueous solutions by γ-alumina nanoparticles: equilibrium and kinetic modeling (pp. 1060-1073) https://doi.org/10.1016/j.molliq.2015.08.029
- Xu et al. (2015) Efficient catalytic removal of formaldehyde at room temperature using AlOOH nanoflakes with deposited Pt (pp. 306-312) https://doi.org/10.1016/j.apcatb.2014.08.017
- Sun et al. (2016) Hierarchical aminated PAN/γ-AlOOH electrospun composite nanofibers and their heavy metal ion adsorption performance (pp. 219-227) https://doi.org/10.1016/j.jtice.2016.02.008
- Dinari et al. (2016) Fabrication of poly(methyl methacrylate)/silica KIT-6 nanocomposites via in situ polymerization approach and their application for removal of Cu2+ from aqueous solution (pp. 11419-11429) https://doi.org/10.1039/C5RA23500F
- Ghamari and Farzi (2017) Effect of morphology control on optical properties of PMMA/boehmite nano-hybrid prepared through facile one-pot process (pp. 16570-16574) https://doi.org/10.1007/s10854-017-7570-6
- Mohammadnezhad et al. (2016) The preparation of modified boehmite/PMMA nanocomposites by: in situ polymerization and the assessment of their capability for Cu2+ ion removal (pp. 3612-3621) https://doi.org/10.1039/c5nj03109e
- Sun et al. (2015) Dynamics of poly(methyl methacrylate)–montmorillonite nanocomposites: a dielectric study (pp. 43-50) https://doi.org/10.1016/j.jnoncrysol.2014.11.030
- Trung et al. (2015) Facile synthesis of three-dimensional graphene/nickel oxide nanoparticles composites for high performance supercapacitor electrodes (pp. 603-609) https://doi.org/10.1016/j.cej.2014.11.140
- Ghamari and Farzi (2017) The impact of morphology control on the microhardness of PMMA/boehmite hybrid nanoparticles prepared via facile aqueous one-pot process (pp. 135-144) https://doi.org/10.1007/s10971-017-4487-8
- Ghamari and Farzi (2017) Frequency and composition dependency of optical and dielectric properties of PMMA/boehmite nano-hybrid prepared via facile aqueous one-pot process https://doi.org/10.1142/S0217984917501202
- Park et al. (2008) Preparation of Al(OH)3/PMMA nanocomposites by emulsion polymerization (pp. 1803-1808) https://doi.org/10.1002/pat.1197
- Imani et al. (2016) Effect of in situ oxidative preparation on electrical properties of epoxy/PANi/MWCNTs nanocomposites (pp. 10364-10370) https://doi.org/10.1007/s10854-016-5122-0
- Grohens et al. (1997) PMMA conformational changes on γ-alumina powder: influence of the polymer tacticity on the configuration of the adsorbed layer (pp. 163-167) https://doi.org/10.1016/s0143-7496(96)00035-8
- Pantoja et al. (2009) Analysis of hydrolysis process of γ-methacryloxypropyltrimethoxysilane and its influence on the formation of silane coatings on 6063 aluminum alloy (pp. 6386-6390) https://doi.org/10.1016/j.apsusc.2009.02.022
- Naskar and Chatterjee (2005) Boehmite nanoparticles by the two-reverse emulsion technique (pp. 3322-3326) https://doi.org/10.1111/j.1551-2916.2005.00600.x
- Laachachi et al. (2009) A comparison of the role of boehmite (AlOOH) and alumina (Al2O3) in the thermal stability and flammability of poly(methyl methacrylate) (pp. 1373-1378) https://doi.org/10.1016/j.polymdegradstab.2009.05.014
- Miao et al. (2012) Electrospun self-standing membrane of hierarchical SiO2 at γ-AlOOH (Boehmite) core/sheath fibers for water remediation (pp. 5353-5359) https://doi.org/10.1021/am3012998
- Rajamani and Rajendrakumar (2019) Chitosan-boehmite desiccant composite as a promising adsorbent towards heavy metal removal (pp. 257-264) https://doi.org/10.1016/j.jenvman.2019.05.056
- Imani and Oveisi (2019) Self-assembly assisted fabrication of nanoporous nickel(II) phosphate octahydrate microspheres catalyst with orange peel surface toward urea oxidation (pp. 7338-7342) https://doi.org/10.1002/slct.201901107

 10.1007/s40089-019-00286-6
10.1007/s40089-019-00286-6