Formulation and In Vitro Evaluation of Metformin-Loaded Chitosan Nanoparticles Using Ionic Gelation Technique
- Faculty of Pharmacy, Cyprus International University, Nicosia, Cyprus
- Department of Pharmaceutical Sciences, Faculty of Pharmacy, Isra University, Queen Alya Airport Street, Amman 11622, Jordan
- College of Pharmacy, Igbinedion University, Okada, Edo State, Nigeria
- Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, University of Benin, Benin-City, Nigeria

Received: 2025-01-02
Revised: 2025-02-25
Accepted: 2025-03-18
Published in Issue 2025-03-31
Copyright (c) 2025 Airemwen Collins Ovenseri, Ahmad Saleh Malkawi, Obarisiagbon A. Johnbull, Uhumwangho U. Michael (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 135
Abstract
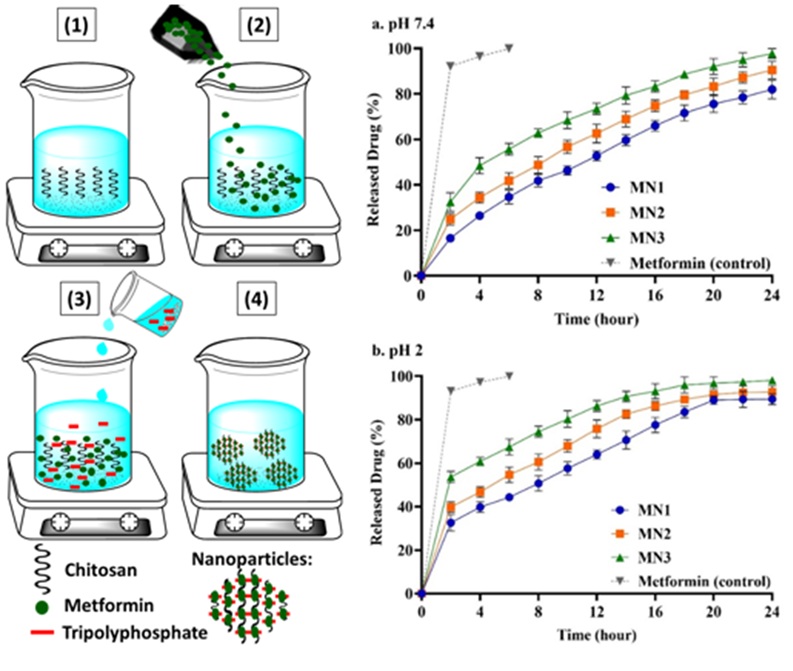
This study formulated and characterized metformin-loaded chitosan nanoparticles (NPs) using the ionic gelation technique and evaluated their drug release kinetics. Characterization confirmed successful drug encapsulation, with FTIR indicating compatibility, XRD showing reduced crystallinity, and particle sizes ranging from 184.28 to 246.82 nm. The NPs exhibited stable zeta potentials (+42.38 to +49.06 mV) and high entrapment efficiencies (68.42% − 81.26%). In vitro drug release studies at pH 7.4 and pH 2 demonstrated an initial burst release, followed by sustained release over 24 hours. The cumulative drug release ranged from 81.92% to 97.72% at pH 7.4 and 89.4% to 98.1% at pH 2, with a faster release at pH 2. Drug release kinetics followed First-order for MN1, while MN2 and MN3 best fitted the Higuchi model, indicating diffusion-controlled release through the chitosan polymeric network. These findings highlight the potential
of metformin-loaded chitosan NPs for sustained drug delivery, which may enhance patient compliance by reducing dosing frequency. Future studies should further explore their clinical applications.
Keywords
- Metformin,
- Chitosan nanoparticles,
- Ionic gelation,
- Drug release kinetics,
- Controlled drug delivery
References
- S.A. Patil et al. Nanopharmaceuticals for Drug Delivery. In: Advances in Novel Formulations for Drug Delivery. . pages 29–43, (2023).
- A.C. Ovenseri and E.M. Halilu. Formulation and in vitro evaluation of polymeric metronidazole nanoparticles. Pak J Pharm Sci, 35(5):1333–1338, (2022).
- A. Pereira et al. In vitro-in vivo availability of metformin hydrochloride-PLGA nanoparticles in diabetic rats in a periodontal disease experimental model. Pharm Biol, 59(1):1576–1584, (2021).
- L. Alhajj, C. Ovenseri Airemwen, and L.B. Pozharani. Formulation of aspirin nanoparticles using solvent evaporation method and in vivo evaluation of its antithrombotic effect. Pak J Pharm Sci, 36(5(Special)):1583–1589, (2023).
- A.C. Ovenseri and O.A. Johnbull. Formulation of silver nanoparticles from the leaves extract of Vernonia amygdalina. . Nigerian Journal of Pharmacy, 57(1):459–466, (2023). DOI: https://doi.org/10.51412/psnnjp.2023.7.
- E. Rajaei et al. HLAs in Autoimmune Diseases: Dependable Diagnostic Biomarkers? . Curr Rheumatol Rev, 15(4):269–276, (2019).
- R. Klein et al. The incidence of hypertension in insulin-dependent diabetes. Arch Intern Med, 156(6):622–7, (1996).
- S.S. Fajans, G.I. Bell, and K.S. Polonsky. Molecular mechanisms and clinical pathophysiology of maturity-onset diabetes of the young. . N Engl J Med, 345(13):971–80, (2001).
- B.M. Shields et al. Maturity-onset diabetes of the young (MODY): How many cases are we missing? . Diabetologia, 53(12):2504–8, (2010).
- S.I. Othman et al. Chitosan for biomedical applications, promising antidiabetic drug delivery system, and new diabetes mellitus treatment based on stem cell. Int J Biol Macromol, 190:417–432, (2021).
- D.N. Priyanka, K.V.H. Prashanth, and R.N. Tharanathan. A review on potential anti-diabetic mechanisms of chitosan and its derivatives. Carbohydrate Polymer Technologies and Applications, 3:100188, (2022).
- N.C. Ngwuluka, D.J. Kotak, and P.V. Devarajan. Design and Characterization of Metformin-Loaded Solid Lipid Nanoparticles for Colon Cancer. AAPS Pharm Sci Tech, 18(2):358–368, (2017).
- A.C.O.a.U.U. Michael. Formulation and Evaluation of Effervescent Floating Matrix Tablets of a Biguanide Using Grewia mollis Gum. . Asian Journal of Applied Sciences, 12(2):91–98, (2019).
- G. Corti et al. Sustained-release matrix tablets of metformin hydrochloride in combination with triacetyl-beta-cyclodextrin. Eur J Pharm Biopharm, 68(2):303–9, (2008). DOI: https://doi.org/10.1016/j.photonics.2018.08.005.
- S. Bayda et al. The History of Nanoscience and Nanotechnology: From Chemical-Physical Applications to Nanomedicine. Molecules, 25(1), (2019).
- L.D. Hu et al. Preparation and in vitro/in vivo evaluation of sustained-release metformin hydrochloride pellets.”. Eur J Pharm Biopharm, 64(2):185–92, (2006).
- R. Dumitrescu et al. Metformin-clinical pharmacology in PCOs. J Med Life, 8(2):187–92, (2015).
- S. Kumar et al. Metformin-loaded alginate nanoparticles as an effective antidiabetic agent for controlled drug release. . J Pharm Pharmacol, 69(2): 143–150, (2017).
- H. Bangun, S. Tandiono, and A. Arianto. Preparation and Evaluation of Chitosan ˆ A–Tripolyphosphate Nanoparticles Suspension as an Antibacterial Agent. Journal of Applied Pharmaceutical Science:147–156, (2018).
- S. Sarkar et al. Chitosan: A promising therapeutic agent and effective drug delivery system in managing diabetes mellitus. Carbohydr Polym, 247: 116594, (2020).
- A. Elkomy et al. Preparation and characterization of metformin-loaded chitosan nanoparticles for biomedical applications. (2022).
- B.A. Sabbagh et al. Determination of metformin in fixed-dose combination tablets by ATR-FTIR spectroscopy. Chemical Data Collections, 39: 100868, (2022).
- A.A. Ozturk, E. Yenilmez, and Y. Yazan. Dexketoprofen trometamol-loaded eudragitA® rl 100 nanoparticle formulation, characterization and release kinetics. ACTA Pharmaceutica Sciencia, (2019).
- G. Mubeen and K. Noor. Spectrophotometric method for analysis of metformin hydrochloride. Indian J Pharm Sci, 71(1):100–2, (2009).
- Q. Bao et al. In vitro release testing method development for long-acting injectable suspensions. . Int J Pharm, 622:121840, (2022).
- N.C.f.B. Information. PubChem Compound Summary for CID 4091, Metformin. (2025).
- V. Mokale et al. Formulation of metformin hydrochloride nanoparticles by using spray drying technique and in vitro evaluation of sustained release with 32-level factorial design approach. Drying Technology(12):1455–1461, (2016).
- T. Higuchi. Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. . Journal of Pharmaceutical Sciences, 52(12):1145–1149, (1963).
- R. Censi and P. Di Martino. Polymorph Impact on the Bioavailability and Stability of Poorly Soluble Drugs. . Molecules, 20(10):18759–76, (2015). DOI: https://doi.org/10.3390/molecules26195832.
- S. Jagdale et al. Preparation and Characterization of Metformin Hydrochloride-Compritol 888 ATO Solid Dispersion. . J Young Pharm, 3(3): 197–204, (2011).
- A. Malkawi et al. Development of Self-Emulsifying Drug Delivery Systems (SEDDSs) Displaying Enhanced Permeation of the Intestinal Mucus Following Sustained Release of Prototype Thiol-Based Mucolytic Agent Load. . Molecules, 27(14):4611, (2022).
- M. Yanat and K. Schro¨en. Preparation methods and applications of chitosan nanoparticles; with an outlook toward reinforcement of biodegradable packaging. Reactive and Functional Polymers, 161:104849, (2021).
- W.F. Huang et al. Surface charge switchable and pH-responsive chitosan/polymer core-shell composite nanoparticles for drug delivery application. Composites Part B: Engineering, 121:83–91, (2017).
- A. Malkawi, N. Alrabadi, and R.A. Kennedy. Dual-Acting Zeta-Potential-Changing Micelles for Optimal Mucus Diffusion and Enhanced Cellular Uptake after Oral Delivery. . Pharmaceutics, 13(7):974, (2021).
- A.S. Malkawi, C.O. Airemwen, and A. Malkawi. Formulation of Mixed Micelles of Various Model Drug Ionic Complexes in an Optimized Micellar Base Composed of Surfactants and Cosolvent Mixture: In Vitro Analysis of Drug Delivery Efficiency. Journal of Pharmaceutical Innovation, 20(4): 132, (2025).
- S. Salatin et al. Development of a nanoprecipitation method for the entrapment of a very water soluble drug into Eudragit RL nanoparticles. ”. Res Pharm Sci, 12(1):1–14, (2017).
- S.A. Agnihotri, N.N. Mallikarjuna, and T.M. Aminabhavi. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. Journal of Controlled Release, 100(1):5–28, (2004).
- J.M.d.S. Leite et al. Nanoparticle design for hydrophilic drugs: Isoniazid biopolymeric nanostructure. . Journal of Drug Delivery Science and Technology, 87:104754, (2023).
- T.Y.A. Essel et al. Chitosan Composites Synthesized Using Acetic Acid and Tetraethylorthosilicate Respond Differently to Methylene Blue Adsorption. . Polymers, 10(5):466, (2018).
- A. Saleh. Chitosan-Polyphosphate nanoparticles for a targeted drug release at the absorption membrane. Heliyon, 8(9):e10577, (2022).
- N. Bhattarai, J. Gunn, and M. Zhang. Chitosan-based hydrogels for controlled, localized drug delivery. . Advanced Drug Delivery Reviews, 62(1): 83–99, (2010).
- M. Warsito and F. Agustiani. A review on factors affecting chitosan nanoparticles formation. . IOP Conference Series: Materials Science and Engineering, 1011:012027, (2021).
- A. Ari and B.R. Alhamad. Evaluating dry powder inhalers: From in vitro studies to mobile health technologies. Respiratory Medicine, 215:107281, (2023).
- Y. Lu, S. Kim, and K. Park. In vitro-in vivo correlation: perspectives on model development. . Int J Pharm, 418(1):142–8, (2011).
- A.S. Malkawi et al. Development of Fluorescently Labeled Self-Emulsifying Drug Delivery Systems (SEDDS) for Prolonged Stability, In Vitro Sustained Release, and Cellular Uptake. . Pharm Nanotechnol, 10(2):146–161, (2022).
- M. Rinaudo. Chitin and chitosan: Properties and applications. . Progress in Polymer Science, 31(7):603–632, (2006).
- A. Malkawi et al. Self-Emulsifying Drug Delivery Systems: Hydrophobic Drug Polymer Complexes Provide a Sustained Release in Vitro. Molecular Pharmaceutics, 2020. 17(10): p. ., 17(10):3709–3719, (2020).
- F. Debacq-Chainiaux et al. Identification of potential anti-photoageing algal compounds using an in-vitro model of photoageing†. . Journal of Pharmacy and Pharmacology, 58(12):1577–1583, (2006).
- A. Haider et al. Advances in chitosan-based drug delivery systems: A comprehensive review for therapeutic applications. European Polymer Journal, 210:112983, (2024).
- R.M. Lucinda-Silva, H.R.N. Salgado, and R.C. Evangelista. Alginate–chitosan systems: In vitro controlled release of triamcinolone and in vivo gastrointestinal transit. Carbohydrate Polymers, 81(2):260–268, (2010).
- I.A. Sogias, A.C. Williams, and V.V. Khutoryanskiy. Why is Chitosan Mucoadhesive? . Biomacromolecules, 9(7):1837–1842, (2008).
- A. Grenha et al. Development of new chitosan/carrageenan nanoparticles for drug delivery applications. J Biomed Mater Res A, 92(4):1265–72, (2010).
- M.A. Mohammed et al. An Overview of Chitosan Nanoparticles and Its Application in Non-Parenteral Drug Delivery. Pharmaceutics, 9(4), (2017).
- Z. Ma, H.H. Yeoh, and L.Y. Lim. Formulation pH modulates the interaction of insulin with chitosan nanoparticles. . Journal of Pharmaceutical Sciences, 2002. 91(6): p., 91(6):1396–1404, (2002).
- T. Higuchi. Mechanism of sustained‐action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. Journal of Pharmaceutical Sciences, 52(12):1145–1149, (1963).
- P. Costa and J.M. Sousa Lobo. Modeling and comparison of dissolution profiles. . European Journal of Pharmaceutical Sciences, 13(2):123–133, (2001).
- R.W. Korsmeyer et al. Mechanisms of solute release from porous hydrophilic polymers. . International Journal of Pharmaceutics, 15(1):25–35, (1983).
- P.L. Ritger and N.A. Peppas. A simple equation for description of solute release I. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. . Journal of Controlled Release, 5(1):23–36, (1987).
- J. Siepmann and N.A. Peppas. Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Advanced Drug Delivery Reviews, 48(2):139–157, (2001).
- I.M. Kaplan, J.S. Wadia, and S.F. Dowdy. Cationic TAT peptide transduction domain enters cells by macropinocytosis. Journal of Controlled Release, 102(1):247–253, (2005).
- P.L. Ritger and N.A. Peppas. A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. . Journal of Controlled Release.
- J. Siepmann and F. Siepmann. Mathematical modeling of drug delivery. International Journal of Pharmaceutics, 364(2):328–343, (2008).
- P. Colombo et al. Analysis of the swelling and release mechanisms from drug delivery systems with emphasis on drug solubility and water transport. Journal of Controlled Release, 39(2):231–237, (1996).
- G.-E. Dinora et al. In vitro characterization of some biopharmaceutical properties of praziquantel. . International Journal of Pharmaceutics, 295(1): 93–99, (2005).
- D.Y. Arifin, L.Y. Lee, and C.-H. Wang. Mathematical modeling and simulation of drug release from microspheres: Implications to drug delivery systems. . Advanced Drug Delivery Reviews, 58(12):1274–1325, (2006).
- M. RMurtomaa et al. Effect of particle morphology on the triboelectrification in dry powder inhalers. . International Journal of Pharmaceutics, 282 (1):107–114, (2004).
- J. Tavakoli and Y. Tang. Hydrogel Based Sensors for Biomedical Applications: An Updated Review. Polymers, 9(8):364, (2017).
- I.M. Kaplan, J.S. Wadia, and S.F. Dowdy. Corrigendum to “Cationic TAT peptide transduction domain enters cells by macropinocytosis”. J. Control. Release, 102(1):247–253, (2005).
- M.A. Khanday, A. Rafiq, and K. Nazir. Mathematical models for drug diffusion through the compartments of blood and tissue medium. Alexandria Journal of Medicine, 53(3):245–249, (2017).
- V. Pillay and R. Fassihi. In vitro release modulation from crosslinked pellets for site-specific drug delivery to the gastrointestinal tract: I. Comparison of pH-responsive drug release and associated kinetics. Journal of Controlled Release, 59:229–242, (1999).
- R.S. Langer and N.A. Peppas. Present and future applications of biomaterials in controlled drug delivery systems. Biomaterials, 2(4):201–214, (1981).

 10.57647/inl.2025.1401.04
10.57647/inl.2025.1401.04