Effects of chirality and functionalization of carbon nanotubes as nanocarriers on the adsorption of Lenalidomide anticancer drug: Insights from molecular dynamics simulation
- Department of Chemistry, Gachsaran Branch, Islamic Azad University, Gachsaran, Iran
- Department of Chemistry, Semnan Branch, Islamic Azad University, Semnan, Iran
- Department of Chemistry, Yasouj Branch, Islamic Azad University, Yasouj, Iran
- Department of Chemical Engineering, Ayatollah Amoli Branch, Islamic Azad University, Amol, Iran

Received: 2024-11-01
Revised: 2025-01-25
Accepted: 2025-02-06
Published in Issue 2025-06-01
Copyright (c) -1 Hamidreza Dehghan, Yadollah Saghapour, Arezoo Tahan, Shiva Joohari, Arezoo Ghadi (Author)

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 307
Abstract
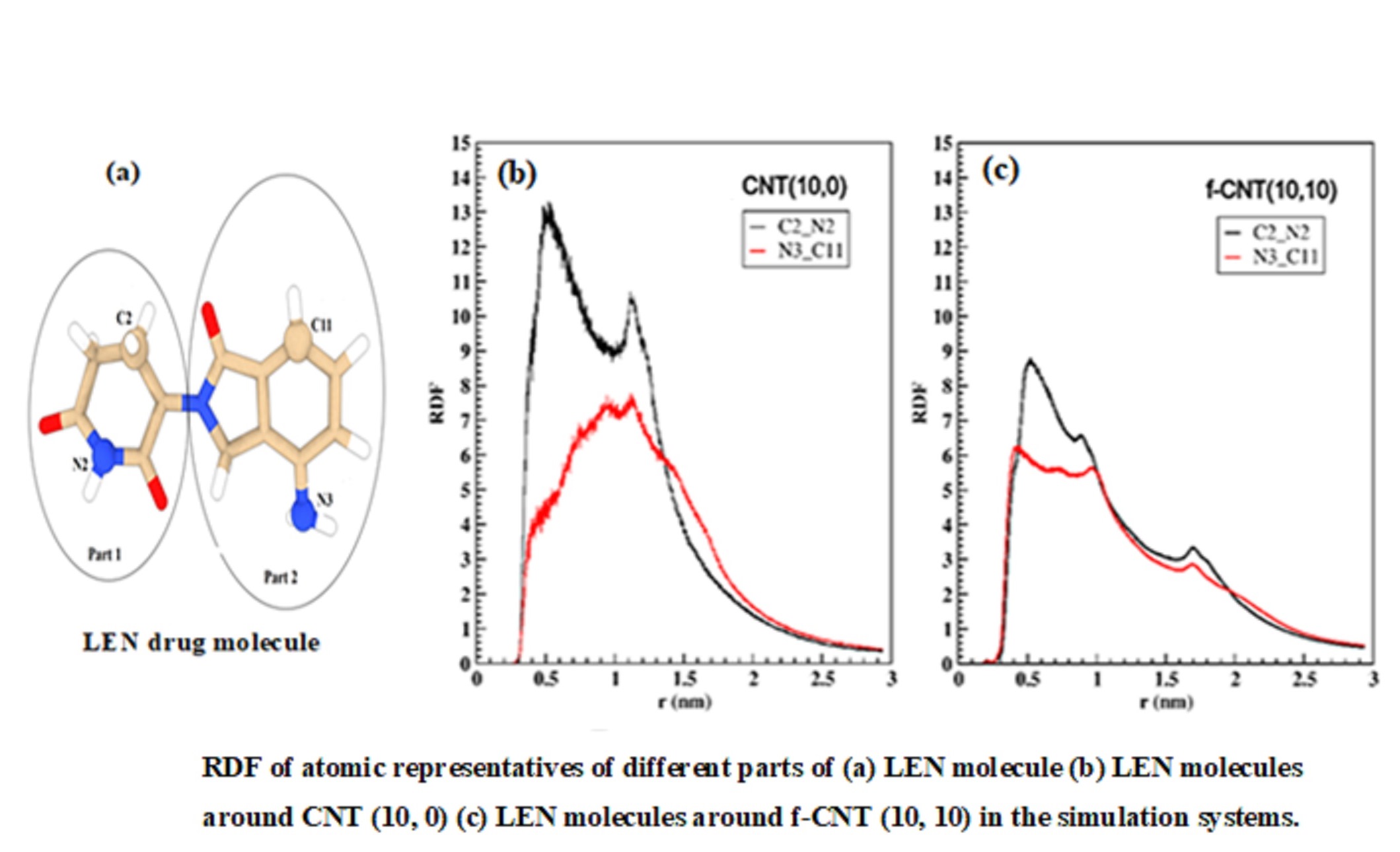
In this study, the adsorption process of Lenalidomide (LEN) anticancer drug on carbon nanotubes (CNTs) with different chiralities was studied using molecular dynamics (MD) simulations. Chirality effects of CNTs (zigzag (10, 0), chiral (10,5) and armchair (10,10)), their functionalization with carboxylic functional groups and solubility were investigated. Results showed that the interaction strength of LEN/water molecules with CNTs and f-CNTs (carbon nanotubes functionalized with carboxylic functional groups) was as a function of chirality. LEN molecules were considerably adsorbed on the f-CNT surface with (10, 10) chirality which was recognized via the most negative van der Waals (vdW) energy and the greatest number of atomic contacts (nAC). Moreover, the number of hydrogen bonds (nHBs) between f-CNT (10, 10) and solvent molecules was higher than for the other f-CNTs. The highest value of solvent accessible surface area (SASA) 45 nm2 for the f-CNT (10,10) compared with the other CNTs confirmed that armchair f-CNT had larger surface area accessible for interacting with the solvent. The functionalization of CNTs led to a decrease in solvation free energy (ΔGsol) and an increase in SASA which resulted in better solubility of f-CNTs in water. Based on the finding, f-CNTs were proposed highly efficient candidate for drug delivery.
Keywords
- Carbon nanotubes,
- Chirality,
- Drug delivery,
- Lenalidomide drug,
- Molecular dynamic simulation
References
- Bartlett J.B., Michael A., Clarke I.A., Dredge K., Nicholson S., Kristeleit H., Polychronis A., Pandha H., Muller G.W., Stirling D.I., (2004), Phase I study to determine the safety, tolerability and immunostimulatory activity of thalidomide analogue CC-5013 in patients with metastatic malignant melanoma and other advanced cancers. British J. Cancer. 90: 955-961. DOI:10.1038/sj.bjc.6601579
- Chen X., Li D., Wang J., Deng Z., Zhang H., (2019), Lenalidomide acesulfamate: Crystal structure, solid state characterization and dissolution performance. J. Mol. Struct. 1175: 852-857. https://doi.org/10.1016/j.molstruc.2018.08.059
- Galustian C., Dalgleish A., (2009), Lenalidomide: A novel anticancer drug with multiple modalities. Exp. Opinion Pharmacotherapy. 10: 125-133. DOI:10.1517/14656560802627903
- Quach H., Ritchie D., Stewart A.K., Neeson P., Harrison S., Smyth M.J., Prince H.M., (2010), Mechanism of action of immunomodulatory drugs (IMiDS) in multiple myeloma. Leukemia. 24: 22-32. DOI:10.1038/leu.2009.236
- Adallahb A.E., Eissa I.H., Mehany A.B.M., Sakr H., Atwa A., El-Adl K., El-Zahabi M.A., (2023), Immunomodulatory quinazoline-based thalidomide analogs: Design, synthesis, apoptosis and anticancer evaluations. J. Mol. Struct. 1281: 135164. DOI:10.1016/j.molstruc.2023.135164
- Kotla V., Goel S., Nischal S., Heuck C., Vivek K., Das B., Verma A., (2009), Mechanism of action of lenalidomide in hematological malignancies. J. Hematology Oncology. 2: 1-10. DOI: 10.1186/1756-8722-2-36
- Armoiry X., Aulagner G., Facon T., (2008), Lenalidomide in the treatment of multiple myeloma: A review. J. Clinic. Pharma. Therapeut. 33: 219-226. DOI:10.1111/j.1365-2710.2008.00920.x
- Meruvia-Rojas Y.V., Molina-Montes E., Hernández-Laguna A., Sainz-Díaz C.I., (2024). Intercalation of the anticancer drug lenalidomide into montmorillonite for bioavailability improvement: a computational study. J. Mol. Modeling. 31: 5. DOI:10.1007/s00894-024-06210-w
- Ghasemi Gol A., Akbari J., Khalaj M., (2023), An Iron-enhanced nanocone assisted drug delivery of Aspirin: DFT assessments. Int. J. Nano Dimens. 14: 339-347. https://doi.org/10.22034/ijnd.2023.1994253.2245
- Sadjadi M.S., Sadeghi B., Zare K., (2007), Natural bond orbital (NBO) population analysis of cyclic thionylphosphazenes, [NSOX (NPCl2)2];X=F(1),X=Cl(2). J. Mol. Struct. THEOCHEM. 817: 27–33. doi:10.1016/j.theochem.2007.04.015
- Zare K., Shadmani N., Pournamdari E., (2013), DFT/NBO study of Nanotube and Calixarene with anti-cancer drug. J. Nanostruct. Chem. 3: 1-6. https://doi.org/10.1186/2193-8865-3-75
- Fei Zh., Yoosefian M., (2021), Design and development of polymeric micelles as nanocarriers for anti-cancer Ribociclib drug. J. Mol. Liq. 329: 115574. https://doi.org/10.1016/j.molliq.2021.115574
- Sadeghy B., Ghammami Sh., (2005), Oxidation of Alcohols with Tetramethylammonium Fluorochromate in Aceticoi Acid. Rus. J. Gen. Chem. 75: 1886–1888. https://doi.org/10.1007/s11176-006-0008-0
- Sadeghi B., Ghammamy Sh., Gholipour Z., Ghorchibeigy M., Amini Nia A., (2011), Gold/hydroxypropyl cellulose hybrid nanocomposite constructed with more complete coverage of gold nano-shell. Micro & Nano Lett. 6: 209–213. https://doi.org/10.1049/mnl.2011.0036
- Esfahani S., Akbari J., Soleimani-Amiri S., (2024), Adsorption of ibuprofen by an iron-doped silicon carbide graphene monolayer: DFT exploration of drug delivery insights. Int. J. Nano Dimens. 15: 63-71. https://creativecommons.org/licenses/by-nc/4.0
- Hakemi P., Ghadi A., Mahjoub S., Zabihi E., Tashakkorian H., (2021), Fabrication of PCL-PEG-PCL nanocarrier for Co-loading of Docetaxel/Quercetin and assessment of its effect on growth inhibition of human liver cancer (Hep-G2) cell line. Int. J. Nano Dimens. 12: 355-368. http://creativecommons.org/licenses/by/4.0
- Sadjadi M.A.S., Meskinfam M., Sadeghi B., Jazdarreh H., Zare K., (2011), In Situ Biomimetic Synthesis and Characterization of Nano Hydroxyapatite in Gelatin Matrix. J. Biomed. Nanotechnol. 7: 450-454. https://doi.org/10.1166/jbn.2011.1305
- Amininia A., Pourshamsian K., Sadeghi B., (2020), Nano-ZnO Impregnated on Starch—A Highly Efficient Heterogeneous Bio-Based Catalyst for One-Pot Synthesis of Pyranopyrimidinone and Xanthene Derivatives as Potential Antibacterial Agents. Russ. J. Org. Chem. 56: 1279–1288. https://doi.org/10.1134/S1070428020070234
- Ajori S., Haghighi S., Ansari R., (2017). Buckling Behavior of Carbon Nanotubes Functionalized with Carbene under Physical Adsorption of Polymer Chains: a Molecular Dynamics Study. Braz. J. Phys. 47: 606-616. https://doi.org/10.1007/s13538-017-0528-6
- Ajori S., Haghighi S., Ansari R., (2019). Tensile characteristics of carbene-functionalized CNTs subjected to physisorption of polymer chains: a molecular dynamics study. J. Mol. Model. 25: 318. https://doi.org/10.1007/s00894-019-4189-y
- Esmaeili A., Yoosefian M., Mahani M., (2023). Molecular Dynamics simulation of lenalidomide interaction with cRBN Protein: a target for immunomodulatory Drugs. South African J. Chem. 77: 157-162. https://doi.org/10.17159/0379-4350/2023/v77a20
- Rezazade M., Ketabi S., Qomi M., (2024), Effect of functionalization on the adsorption performance of carbon nanotube as a drug delivery system for imatinib: molecular simulation study. BMC Chem. 18: 85. https://doi.org/10.1186/s13065-024-01197-0
- Arib C., Spadavecchia J., (2020), Lenalidomide (LENA) Hybrid Gold Complex Nanoparticles: Synthesis, Physicochemical Evaluation, and Perspectives in Nanomedicine. ACS Omega. 5: 28483-28492. DOI:10.1021/acsomega.0c02644
- Mallina S.A., Sundararajan R., (2018), Lenalidomide loaded lactoferrin nanoparticle for controlled delivery and enhanced therapeutic efficacy. Res. J. Pharma. Technol. 11: 4010-4014. DOI:10.5958/0974-360X.2018.00737.0
- Ajori S., Haghighi S., Ansari R., (2018). A molecular dynamics study on the buckling behavior of cross-linked functionalized carbon nanotubes under physical adsorption of polymer chains. Applied Surface Sci. 427(1): 704-714. https://doi.org/10.1016/j.apsusc.2017.08.049
- Moghaddam Jafari A., Morsali A., Bozorgmehr M.R., Beyramabadi S.A., Mohseni Sh., (2024), Modeling and characterization of lenalidomide-loaded tripolyphosphate-crosslinked chitosan nanoparticles for anticancer drug delivery. Int. J. Biolog. Macromol. 260: 129360. DOI: 10.1016/j.ijbiomac.2024.129360
- Harati H., Morsali A., Bozorgmehr M.R., Beyramabadi S.A., (2021), β-cyclodextrin-lenalidomide anticancer drug delivery nanosystem: A quantum chemical approach. J. Mol. Liq. 344: 117762. DOI:10.1016/j.molliq.2021.117762
- Sharifi S., Sheikhi M., Shahab S., Kaviani S., Kumar R., (2022), DFT Study on the Interaction of Lenalidomide Anticancer Drug on the Surface of B12N12 Nanocluster. Lett. Org. Chem. 19: 583-595. DOI: 10.2174/1570178618666211027102305
- Sayiner H.S., Kandemirli F., Dalgic S.S., Monajjemi M., Mollaamin F., (2022), Carbazochrome carbon nanotube as drug delivery nanocarrier for anti bleeding drug: quantum chemical study. J. Mol. Modeling. 28: 11. https://doi.org/10.1007/s00894-021-04948-1
- Rezaei A., Morsali A., Bozorgmehr M.R., Nasrabadi M., (2021), Quantum chemical analysis of 5-aminolevulinic acid anticancer drug delivery systems: Carbon nanotube, –COOH functionalized carbon nanotube and iron oxide nanoparticle. J. Mol. Liq. 340: 117182. https://doi.org/10.1016/j.molliq.2021.117182
- Ahamed S.B.B., Ibrahim F.B., Srinivasan H., (2021), Cancer nanomedicine: A review on approaches and applications towards targeted drug delivery. Int. J. Nano Dimens. 12: 310-327. http://creativecommons.org/licenses/by/4.0
- Rahmanifar E., Yoosefian M., Karimi-Maleh H., (2016). Electronic properties and reactivity trend for defect functionalization of single-walled carbon nanotube with B, Al, Ga atoms. Synthetic Metals. 221: 242-246. https://doi.org/10.1016/j.synthmet.2016.09.017
- Zaboli M., Raissi H., Zaboli M., (2022), Investigation of nanotubes as the smart carriers for targeted delivery of mercaptopurine anticancer drug. J. Biomol. Struct. Dynamics. 40: 4579-4592. DOI:10.1080/07391102.2020.1860823
- Sun H., She P., Lu G., Xu K., Zhang W., Liu Z., (2014), Recent advances in the development of functionalized carbon nanotubes: A versatile vector for drug delivery. J. Mater. Sci. 49: 6845-6854. DOI:10.1007/s10853-014-8436-4
- Hashemzadeh H., Raissi H., (2017), The functionalization of carbon nanotubes to enhance the efficacy of the anticancer drug paclitaxel: a molecular dynamics simulation study. J. Mol. Modeling. 23: 1-10. DOI:10.1007/s00894-017-3391-z
- Narkhede R., More M., Patil S., Patil P., Patil A., Deshmukh P., (2021), Eco-friendly synthesis of surface grafted Carbon nanotubes from sugarcane cubes for the development of prolonged release drug delivery platform. Int. J. Nano Dimens. 12: 211-221. http://creativecommons.org/licenses/by/4.0
- Yoosefian M., Jahani M., (2019). A molecular study on drug delivery system based on carbon nanotube for the novel norepinephrine prodrug, Droxidopa. J. Mol. Liq. 284: 258-264. https://doi.org/10.1016/j.molliq.2019.04.016
- Kamel M., Raissi H., Morsali A., Shahabi M., (2018), Assessment of the adsorption mechanism of Flutamide anticancer drug on the functionalized single-walled carbon nanotube surface as a drug delivery vehicle: An alternative theoretical approach based on DFT and MD. Appl. Surface Sci. 434: 492-503. DOI: 10.1016/j.apsusc.2017.10.165
- Jo S., Kim T., Iyer V.G., Im W., (2008), CHARMM‐GUI: a web‐based graphical user interface for CHARMM. J. Computat. Chem. 29: 1859-1865. DOI:10.1002/jcc.20945
- Abraham M.J., Murtola T., Schulz R., Páll S., Smith J.C., Hess B., Lindahl E., (2015), GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. Software X. 1-2: 19-25. https://doi.org/10.1016/j.softx.2015.06.001
- Huang J., MacKerell Jr A.D., (2013), CHARMM36 all‐atom additive protein force field: Validation based on comparison to NMR data. J. Computat. Chem.,34: 2135-2145. DOI: 10.1002/jcc.23354
- Jorgensen W.L., Chandrasekhar J., Madura J.D., Impey R.W., Klein M.L., (1983), Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79: 926-935. https://doi.org/10.1063/1.445869
- Darden T., York D., Pedersen L., (1993), Particle mesh Ewald: An W log (N) method for Ewald sums in large systems. J. Chem. Phys. 98: 10089-10092. https://doi.org/10.1063/1.464397
- Yousefi M., Salehi Rad M., Shakibazadeh R., Ghodrati L., Ataie Kachoie M., (2022), Simulating a heteroatomic CBN fullerene-like nanocage towards the drug delivery of fluorouracil. Mol. Simul. 48: 1284-1292. DOI:10.1080/08927022.2022.2086252
- Chen T., Li M., Liu J., (2018), π–π stacking interaction: a nondestructive and facile means in material engineering for bioapplications. Crystal Growth Design. 18: 2765-2783. https://doi.org/10.1021/acs.cgd.7b01503
- Razavi R., Ahmadi S.A., (2021), Molecular machine based on Rotaxane@Tricyclic antidepressant carrier: Theoretical molecular dynamic simulation. Computat. Theore. Chem. 1197: 113138. https://doi.org/10.1016/j.comptc.2020.113138
- Kordzadeh A., Amjad-Iranagh S., Zarif M., Modarress H., (2019), Adsorption and encapsulation of the drug doxorubicin on covalent functionalized carbon nanotubes: A scrutinized study by using molecular dynamics simulation and quantum mechanics calculation. J. Mol. Graph. Model. 88: 11-22. DOI: 10.1016/j.jmgm.2018.12.009
- Kordzadeh A., Zarif M., Amjad-Iranagh S., (2023), Molecular dynamics insight of interaction between the functionalized-carbon nanotube and cancerous cell membrane in doxorubicin delivery. Comput. Methods Prog. Biomed. 230: 107332. https://doi.org/10.1016/j.cmpb.2022.107332
- Najafi F., (2020), Thermodynamic studies of carbon nanotube interaction with Gemcitabine anticancer drug: DFT calculations. J. Nanostruct. Chem. 10: 227-242. DOI:10.1007/s40097-020-00344-y
- Berisha A., (2023). Density functional theory and quantum mechanics studies of 2D carbon nanostructures (graphene and graphene oxide) for Lenalidomide anticancer drug delivery. Computat. Theore. Chem. 1230: 114371. https://doi.org/10.1016/j.comptc.2023.114371

 10.57647/j.ijnd.2025.1603.23
10.57647/j.ijnd.2025.1603.23