Results and discussions
Characterization
TEM images of the catalyst are shown in Fig.
1
. It can be observed that particles tend to agglomerate at dimensions near 250 nm. The structure and morphology of the catalyst were investigated by Scanning Electron Microscopy. SEM image of the catalyst is depicted in Fig.
2
. Porous structure of the catalyst is revealed in this nanoscale image.
TEM images of images of iron-modified titanium dioxide SEM image of iron-modified titanium dioxideFig. 1

Fig. 2

Figure
3
shows the typical XPS survey scan of the Fe/TiO
2
sample. No iron could be detected in the surface of sample, which could be because of low concentration of iron being dopant. TiO
2
rutile structure has open diffusion channels with a radius of ~0.77 Å that run parallel to the
c
-axis [
30
]. Since the radius of Fe
3+
is 0.64 Å, which is smaller than both the 0.77 Å channels in the porous structure of pure TiO
2
and the radius of Ti
4+
(0.68 Å) [
31
,
32
], it is possible that Fe
3+
diffuses along the
c
-axis and substitutes Ti
4+
in the TiO
2
lattice [
31
]. Figure
3
a, b show the high-resolution XPS spectra of titanium and oxygen. From Fig.
3
a, it is clear that there are four contributions to the Ti 2
p
region: the Ti (2
p
3/2) and Ti (2
p
1/2) at binding energies of 458.8 and 464.4 eV, respectively. These values for binding energies are in agreement with the values relevant to TiO
2
, moreover, the difference between these two binding energies represents the standard binding energy for Ti element [
33
]. Upon substitution of Ti
4+
with Fe
3+
, the number of electrons of the conduction band of TiO
2
is increased, this bring about the migration of these electrons to iron which results in a decrease in the outer electron cloud density of Ti ions, and overall, It leads to the corresponding increase in generation of surface state of Ti
3+
. Hence, the existence of the Ti
3+
means the presence of Fe
3+
. Existence of Ti
3+
will retard the recombination of the h
+
and e
−
, which has a positive effect on the catalytic activity of Fe/TiO
2
[
34
,
35
]. Figure
3
b shows the corresponding spectra of oxygen with the following composition: The main contribution of oxygen in the Fe/TiO
2
is composed of O
2−
(58.1 %), which can be assigned to the oxygen bounded in the TiO
2
lattice (Ti–O). Its peak is corresponded to binding energy of 530.10 eV. Moreover, 16.6 % of the total oxygen is associated with O
−
which is indicated the presence of surface hydroxyl groups (Ti–OH) as chemisorbed water, The relevant peak is located around 531.8 eV and finally 25.3 % is in the form of physically absorbed water, pertaining to the peak at 533.4 eV [
34
,
35
]. Figure
3
c indicates that iron concentration is below the detection level of XPS. The enhanced activity of Fe/TiO
2
compared with TiO
2
is experimentally shown in Fig.
4
, which confirms that Fe
3+
ions are absorbed on the TiO
2
surface, acting as electron trapping centers. Thus, they increase the electron–hole pair separation efficiency and accordingly, superior activity of Fe/TiO
2
confirms the presence of iron in the surface of the photocatalyst.
XPS survey scan of the Fe/TiO
2
sample.
a
XPS spectra of titanium,
b
XPS spectra of oxygen,
c
XPS resolution spectrum of Fe The effect of Fe photocatalytic activity of the catalyst (Initial phenol concentration: 200 ppm, catalyst loading: 0.5 g/l, H
2
O
2
: 12.5 ml, 30 wt%, reaction time: 120 min, UV light intensity of 757.38 mW/cm
2
)Fig. 3

Fig. 4

The results of XRF analysis for the catalyst characterization are presented in Table
1
. Chemical composition of the synthesized catalyst reveals that it is consisted of TiO
2
(95.85 %), Fe
2
O
3
(0.20 %) and Na
2
O (0.42 %), Al
2
O
3
(0.69 %), SiO
2
(2.84 %) and the ratio of (Fe
2
O
3
/TiO
2
) is (0.2 %/95.85 % = 0.21 %).
Chemical composition of the catalyst Z Symbol Element Concentration (%) 22 TiO2 Titanium 95.85 26 Fe2O3 Iron 0.20 11 Na2O Sodium 0.42 13 Al2O3 Aluminum 0.69 14 SiO2 Silicon 2.84Table 1
XRD pattern of the catalyst is shown in Fig.
5
. Peaks at 32.0095°, 42.1696° and 64.0611° are attributed to the rutile phase (JCPDS File no. 21-1276) and the peak at 29.5090° is attributed to the anatase phase (ICPDS File no. 21-1272). These results imply that both rutile and anatase phases exist in the catalyst. In addition to the peaks belonging to rutile and anatase phases other peaks are also recognized which correspond to iron oxides of Fe
2
O
3
(JCPDS File no. 39-1346). It confirms the existence of iron as dopant in the sample and proves that iron is in its oxide form.
XRD pattern of Fe/TiO2Fig. 5

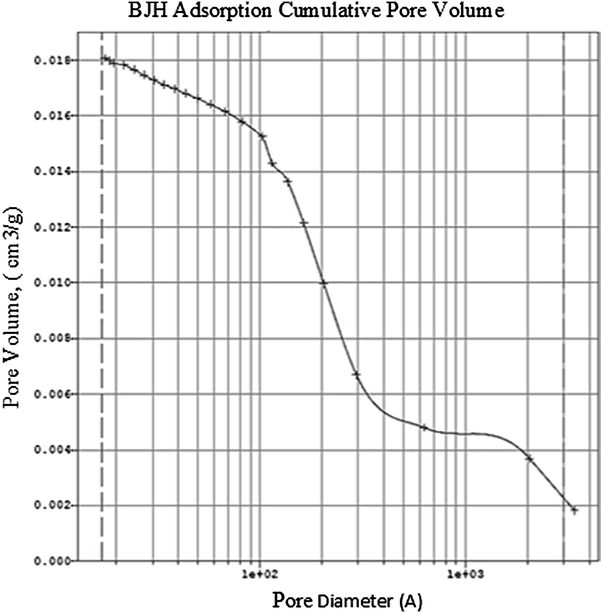
BET model, which was developed by Brunauer, Emmet and Teller is applied for measuring the surface of the catalyst. Based on the IUPAC classification, porous structures are categorized based on their pore sizes, micro pores (pores with diameters less than 2 nm), mesopores (pores with 2–50 nm diameter), and macropores (pores with diameters greater than 50 nm) [
36
]. Since the average pore diameter of Fe/TiO
2
(4 V/A by BET) is 13.9838 nm, the catalyst is considered mesoporous, according to IUPAC classification and specific surface area of the catalyst is 5.1268 m
2
/g. Pore size distribution is multimodal and the decreased prevalence of the pores with increased pore size is evident in Fig.
6
.
Pore size distribution according to BJH adsorption of Fe/TiO2Fig. 6
Figure
7
shows the thermal gravimetric analysis (TGA) and differential thermal analysis (DTA) of Fe/TiO
2
catalyst. In TGA curve, three regions with different slopes are recognized. These regions are 33.5–200.6, 200.6–416.5, and 416.5–650 °C, respectively. In the first region, weight loss is attributed to the vaporization of water and the degradation of volatile organic materials. In the second region, 200.6–416.5 °C, decomposition of the organic materials with strong bound has occurred. Weight loss in the third region, 416.5–600 °C, is negligible and insignificant. In DTA curve, an endothermic broad peak around 100 °C is observed which is attributed to the vaporization of water. The following two exothermic peaks around 200 °C are attributed to the combustion of organics. The broad exothermic peak with onset of 400 °C is representative of the crystallization process. It is notable that below this temperature, the material is amorphous; at 400 °C crystallization begins with the formation of anatase followed by phase transformation from anatase to rutile. The results show excellent agreement with the work of Mesgari et al. [
37
].
TGA–DTA curve of the Fe/TiO2Fig. 7

Activity tests
The photocatalytic activity of Fe-doped TiO
2
nanoparticles was examined by studying the degradation of phenol. A specified amount of the synthesized catalyst (0.5 g/l) was added to the phenol solution (200 ppm, 200 ml), which was selected as the synthetic wastewater. The amount of Fe-loading of Fe/TiO
2
catalyst, initial phenol concentration, and catalyst dosage were investigated and optimized in our previous work [
38
]. Hydrogen peroxide (H
2
O
2
: 30 wt%) was added to the solution as an oxidizing agent. The mixture was stirred at a constant rate and illuminated by the UV lamp (with light intensity of 757.38 (mW/cm
2
) which was fixed 19.5 cm above the batch reactor (Pyrex vessel capacity: 2 l). In this work, air bubbling is carried out by two parallel air pumps. One RS Electrical (model: RS-610) with the maximum flow of 3.5 l/min and the other is HAILEA (model: ACO-5505), with maximum flow is 5.5 l/min. The setup was shielded by aluminum foil during the reaction time to prevent the interference with outside light. After 2 h of reaction, the mixture was centrifuged and the absorbance of the supernatant solution was measured at 270 nm using a Specord 210 UV spectrometer. The experiments were repeated for the blank. All the conditions in the blank were the same as the sample, but it had no catalyst. The concentration of phenol in the solution was measured using Beer–Lambert law and photo degradation of phenol was calculated using following equation:
H2O2
Adding proper amount of electron acceptor results in preventing the electron–hole recombination and increasing the number of hydroxyl radicals and other oxidizing agents. Optimization of H
2
O
2
dosage is a fundamental factor in the photocatalytic degradation of phenol. Hydroxyl generation can be executed through three routes, the first route is H
2
O
2
photo-dissociation, the second route is the reaction between H
2
O
2
photogenerated electrons on TiO
2
surface, and the third one is the reaction between H
2
O
2
and peroxyl ion that is produced from the reaction of dissolved O
2
and electrons of conduction band [
39
]. An experiment was conducted with different dosages of H
2
O
2
and the results are given in Fig.
8
. It has been shown that at low H
2
O
2
concentrations, the produced
·
OH radicals are not sufficient for phenol photocatalytic degradation. However, high concentrations of hydrogen peroxide will lead to a decrease in phenol degradation, since
·
OH radicals react with the excess H
2
O
2
. This reaction consumes hydroxyl radicals and competes with phenol oxidation. According to Fig.
8
, increasing the H
2
O
2
dosage up to 12.5 ml enhances the activity of Fe/TiO
2
and further increase will lead to lower efficiencies. Consequently, the optimum amount for H
2
O
2
is 12.5 ml.
The optimization of H
2
O
2
dosage in the phenol degradation processFig. 8

Air Bubbling
The role of aeration in photocatalytic degradation is inevitable. Dissolved oxygen (DO) increases quantum efficiencies of photocatalytic degradation partly by inhibiting electron–hole recombination. In fact, DO acts as electron acceptor during superoxide radical anion formation, which leads to separate electrons and holes. It has been reported that the reaction of O
2
with electrons at the surface of catalyst is slow and it is regarded as the rate-determining step in photocatalysis. Air bubbling is the simplest way to supply molecular oxygen; moreover, it provides turbulence, which assists mass transfer [
40
,
41
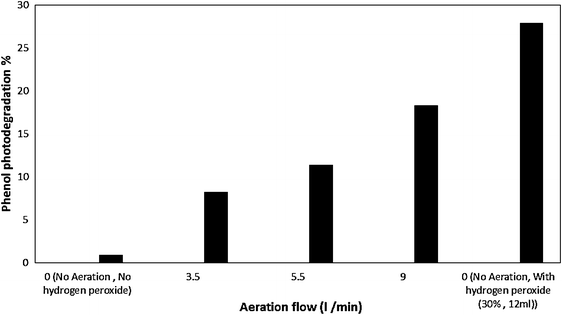
]. The effect of aeration on phenol degradation is displayed in Fig.
9
. It is observed that after 120 min, When there is no oxidizing agent (no aeration, and no H
2
O
2
), phenol degradation is negligible. According to Fig.
9
, increasing the aeration flow rate leads to enhance the phenol degradation. As a result, the highest flow rate (9 l/min) is considered the optimum. Albeit, it is of great importance that compared to H
2
O
2
, aeration is less efficient.
The effect of aeration on phenol photodegradationFig. 9
Temperature
In general, the activation energy of photocatalytic reactions is provided by photons and operations take place at room temperature. To study the influence of temperature, an experiment was performed at 27, 37, and 47 °C. Figure
10
shows the effect of temperature rise on the photodegradation of phenol. It represents that the higher the temperature, the less phenol photodegradation. As it can be seen in Fig.
10
twenty degrees of temperature rise above room temperature drastically drops phenol degradation. In fact, degradation of phenol at 47 °C is inconsiderable. It could be the result of the decrease in dissolved oxygen at higher temperatures. Presence of oxygen is favorable and it prevents the recombination of e
−
and h
+
of the Fe/TiO
2
photocatalyst [
42
,
43
]. Based on the obtained results, the catalytic degradation of phenol is recommended to be performed at ambient temperature.
The effect of temperature on phenol photodegradationFig. 10

Inorganic ions
The existence of inorganic ions is shown to have a significant effect on the photocatalytic degradation of organic compounds. In this test, the effect of the existence of different inorganic ions including Ni
3+
, Cu
2+
, SO
4
2−
, and Cl
−
in the phenol synthetic wastewater was investigated. The concentration of additive ions was the same (1 mM) in each experiment. The results are shown in Fig.
11
. Anions and phenol are in a competition to react with OH radicals, and thus they hinder phenol photodegradation. The low efficiency of the degradation process (less than 5 %) once SO
4
2−
, and Cl
−
being added to phenol solution highlights this finding. KASHIF Naeem and Ouyang Feng have enunciated that anions affect the phenol photocatalytic degradation, they reported the inhibition order of the investigated anions as Cl
−
> SO
4
2−
> NO
3
−
> CO
3
2−
[
44
].
The effect of existence of inorganic ions on the phenol photodegradationFig. 11

Cations absorbing on the surface of iron-modified titanium dioxide are reduced by activated electrons. Therefore, electron and holes will be separated more efficiently and their recombination is avoided. It seems that the existence of cations should have a positive influence on phenol degradation. Figure 11 shows that addition of copper enhances the performance of Fe/TiO 2 ; this arose from electron scavenging effect that inhibits the recombination of the photogenerated electron and holes. Whereas, the addition of Ni 3+ slightly diminishes the efficiency of the catalyst, probably due to reaction with phenol and subsequently, the formation of phenol complexes. Accordingly, the influence of inorganic ions was observed to follow this order: Cu 2+ > No additive > Ni 3+ > Cl − > SO 4 2− . Furthermore, it has been shown that the addition of copper as additive ion to phenol solution boosts the degradation of phenol; the highest phenol degradation efficiency after 120 min (43.85 %) was attained under this condition. From the observed superior performance of Fe/TiO 2 catalyst once copper ion was added to phenol synthetic wastewater, it can be concluded that the nanoparticles of Fe/TiO 2 will be an extremely capable catalyst for treating industrial wastewaters containing both phenol and heavy metals such as copper.