Results and discussion
Morphological properties and phase structure of prepared films
Hexane adsorption–desorption isotherms and pore size distribution analysis of the mesoporous TiO
2
, TiO
2
/SiO
2
and SiO
2
films are illustrated in Fig.
1
.
Hexane adsorption isotherms, and the
distribution function of pore size—
insets
of SiO
2
(
a
), SiO
2
/TiO
2
—10 % (
b
) film on glass substrate sintered at 350 °C and TiO
2
film (
c
) sintered at 500 °CFig. 1
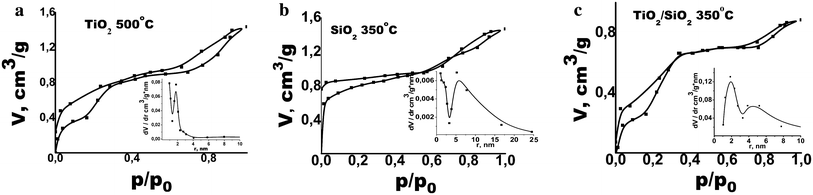
Isotherms of hexane adsorption on the films are of type IV which indicates the presence of mesopores [
21
]. For TiO
2
/SiO
2
, SiO
2
films calcined at 350 °C, and TiO
2
at 500 °C adsorption–desorption isotherms have two hysteresis loops: the first one at high pressures of hexane which indicates the presence of mesopore and the second loop at low pressures, which indicates the irreversible structural deformation of films and formation of the so-called ink bottle type of porosity [
22
]. In this case, desorption of adsorbed molecules is hindered at a given temperature. Moreover, the shape of the hysteresis loop for SiO
2
nanocomposite clearly indicates the presence of micropores in this sample. The total surface area and effective pore radii values yielded from BET analysis of the isotherms are presented in Table
1
.
Hydrophilic properties and efficiency in AY photodegradation of SiO
2
, SiO
2
/TiO
2
and TiO
2
films Sample Water contact angle, grad Adsorption AY, mol/g of the film Rate constants of adsorbed AY decomposition Before After 10 min UV SiO2 350 °C 1580 <2; 12 10 10 2.2 × 10−4 0.6 TiO2/SiO2 350 °C 1300 4; 10 12 5 2.9 × 10−4 3.5 TiO2 500 °C 910 <2; 4 10 ~0 1.8 × 10−4 15Table 1
The shape of the adsorption–desorption isotherms and pore size distribution of prepared TiO 2 , TiO 2 /SiO 2 and SiO 2 films indicate the presence of pores of different diameters. Thus, the pore diameters of 4 and 10 nm dominate in TiO 2 /SiO 2 films. Both TiO 2 and SiO 2 samples have micropores with diameter of less than 2 nm and mesopores with diameter about 4 and 12 nm, respectively. The effective pore radius of the TiO 2 films, calculated from the corresponding SEM images (not shown), was ~8–12 nm.
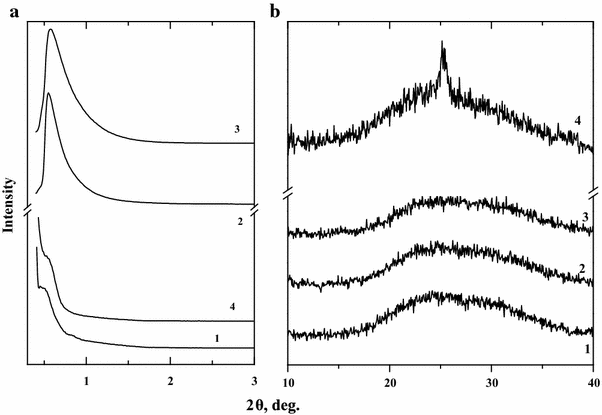
Anatase phase has been identified in XRD patterns for TiO
2
and TiO
2
/SiO
2
(
d
~7 nm) powders prepared from films precursors (Fig.
2
).
Low-angle X-ray diffraction patterns (
a
) and wide-angle XRD patterns (
b
) of the meso-structured SiO
2
—1, SiO
2
/TiO
2
(10 %)—2 and TiO
2
films calcined at 350 °C—3, at 500 °C—4Fig. 2
Hydrophilic properties of the synthesized films
SiO 2 and TiO 2 /SiO 2 films freshly prepared on glass substrates showed highly hydrophilic properties (see data listed in Table 1 ); water contact angles for them were ca. 10°–20°. TiO 2 films after calcination at 350 °C show contact angle near 32 o . Thermal treatment at 500 °C leads to a drop of water contact angle up to 5°–10°. Literature data on the contact angle of water on a clean titania surface are contradictory, ranging from 72° reported in [ 23 ] to 33.3° in [ 24 ] and 15° in [ 25 ] depending on the film structure and storage conditions. Various experimental results relating to this phenomenon have shown that it is caused by defects formed on the surfaces of the as-prepared TiO 2 thin films subjected to high-temperature calcination, especially in case of template syntheses involving high level of organic component as it was used in our work. According to K. Hashimoto and co-workers, the wettability of TiO 2 surfaces can be altered by irradiation [ 23 – 25 ]. After UV illumination (10 min in air under mercury lamp light), mesoporous TiO 2 films exhibited photoinduced super-hydrophilic properties with water contact angle near 0°.
FTIR investigation
The FTIR spectrum of TiO
2
(500 °C) powder prepared from the film precursor contains a single broad high-intensive absorption band at 1000–370 cm
−1
with a maximum at 550 cm
−1
associated with the Ti–O stretching vibrations. In the FTIR spectra of the mesoporous SiO
2
and TiO
2
/SiO
2
calcined at 350 °C (Fig.
3
) a set of vibrations can be seen as characteristic of the materials studied. The band at 794–804 cm
−1
is assigned to symmetric stretching vibrations of the Si–O–Si bond [
26
–
29
]. Intensive band at 947–960 cm
−1
is usually attributed to the stretching vibrations of the Si–O–Ti bond [
27
–
29
] for TiO
2
/SiO
2
mixed oxides prepared through simultaneous hydrolysis of the titanium and silicon containing organometallic precursors. This shows evidences of chemical interaction between titania and silica at molecular level. When titania nanoparticles containing 2-methoxyethoxide side groups interact with growing Si–O–Si chain, Si–O–Ti bonds can be formed at the interface.
FT-IR spectra of the mesoporous SiO
2
(1) and TiO
2
/SiO
2
(2–1 mol.% of TiO
2
; 3–5 mol.% of TiO
2
; 4–10 mol.% of TiO
2
) films deposited onto steel substrates and calcined at 350 °CFig. 3
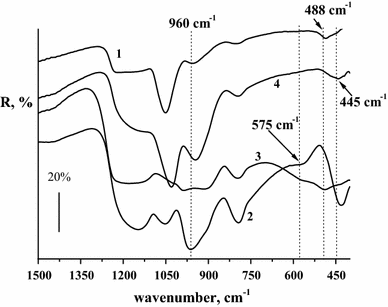
A weak band in the range of 952–958 cm −1 is also seen in the FTIR spectrum of pure SiO 2 and is assigned to the symmetric stretching vibrations of the Si–OH groups [ 27 , 28 ]. Two bands in the range of ~1023–1041 cm −1 and ~1146–1192 cm −1 are attributed to the asymmetric stretching vibrations of the Si–O–Si bond [ 27 – 29 ]. It is accepted that degree of titanium incorporation into silica framework can be monitored by the intensity of the band at ~960 cm −1 corresponding to the vibrations of the Si–O–Ti bond [ 29 ]. In our case, intensity of the band gradually decreases with increasing titanium content into silica framework from 1 to 10 mol.% that can be related to the titania polymer network formation and/or titania segregation into separate phase in the SiO 2 network with increasing its content in the mixed oxides. The presence of two bands in the wave numbers range 440–480 and 550 cm −1 in the IR spectrum of the titanosilicates evidences of the formation of zeolite-type structure in this system [ 30 , 31 ]. First band corresponds to the internal vibrations of the Si–O tetrahedrons and is also presented in the spectrum of pure SiO 2 . It corresponds to the stretching vibrations of the Si–O–Si bond. In the FTIR spectrum of the mesoporous SiO 2 film (Fig. 3 , curve 1), one band can be seen in the range of 484–488 cm −1 . When titania was added (1, 5 mol.%) into SiO 2 films (Fig. 3 , curves 2, 3), we registered two bands of low intensity: the first one at 563–575 cm −1 and the second one at ~432–490 cm −1 . First band is similar to that of five-membered rings of Si–O–T (where T = Si or Ti) in microporous zeolites [ 30 , 31 ]. On the assumption of the integral intensity of the bands of adsorbed water at 1640 cm −1 and associated water molecules with surface hydroxyl groups in the range of 3600–3300 cm −1 we can make a conclusion about surface hydroxylation of the films depending on the TiO 2 content in the SiO 2 matrix. It was observed the increase of the surface hydroxylation of titanium-containing silica in comparison with bare SiO 2 films after similar thermoactivation. According to [ 26 ], the reduction in the absorption frequency of Si–O stretching vibration (near 1100 cm −1 ) indicates the distortion of a SiO 4 tetrahedron in the presence of a heavier neighboring atom linked to a tetrahedron via an oxygen atom. The formation of Ti–O–Si bonds is assumed to be a key factor affecting surface properties such as step of surface hydroxylation and surface acidity and catalytic function of silica–titania [ 9 , 10 , 32 ]. Imamura et al. [ 10 ] reported the change in the amount of acid sites of silica–titania with the change in Ti:Si ratio: weak acid sites were present on both single component oxides. Stronger acid sites appeared after their combination. Maximum amount of sites with acid strength of 1.5 < H o ≤ 3.3 was observed for TiO 2 /SiO 2 with low Ti content up to 10 %.
As we reported previously analysis of the data of XPS investigation of the mixed TiO 2 /SiO 2 as well as bare titania and silica films [ 33 ] proved molecular scale mixing for sol–gel derived titanosilicates. Considerable upward shift of the Ti 2p 1/2 and Ti 2p 3/2 lines was registered for TiO 2 /SiO 2 film as compared with lines position for pure TiO 2 . This upward shift can account for the formation of the Ti–O–Si bonds, as a result of increase of the positive charge on the Ti species resulting from the greater electronegativity of Si via O acting on Ti [ 28 ]. Downward shift of the position of O 1s and Si 2p lines for TiO 2 /SiO 2 film was detected relatively to the position of the lines for pure SiO 2 film. Lee et al. [ 27 ] explained this negative O 1s line shift to the insertion of the Ti 4+ cations into the tetrahedral sites of the silica network to form Ti–O–Si bonds.
Theoretical studies on acridine yellow adsorption and photodegradation on silica, silica–titania, and titania surfaces
When organic substances are adsorbed on silica and titania surfaces, the reactivities of their molecules can change as compared to those in free state.
Elementary stages of reactions as well as the structures of adsorption complexes not always can be determined by experimental methods. To verify probable photodegradation routes of organic dyes, in particular those of acridine yellow, theoretical simulation can be used, especially methods of quantum chemistry [ 34 ]. Density functional theory method (DFT) is used successfully in studies of the mechanisms of both homolytic and heterolytic decomposition of organic matter [ 35 ], as this method describes electron correlation.
Our previous calculations testify [ 36 ] the use of B3LYP functional with extended valence-split 6–31G(d,p) basis set in calculations of energy values of reaction mechanisms to give reasonable results and negligible the basis set superposition error as compared to other, more improved methods. That is why the calculations were performed using the DFT method with B3LYP exchange–correlation functional and 6-31G(d,p) basis set [ 37 , 38 ]. The dispersion correction was taken into account using the DFTD3 model by Grimme et al. [ 39 ]. The equilibrium spatial structures of reactant molecules and reaction products of the elementary stages were found due to gradient norm minimization. All the calculations have been carried out by means of the US GAMESS program [ 40 ].
First, probable adsorption complexes are to be examined of AY (in neutral and protonated HAY + forms) on the surfaces of the sorbents.
The energy values of acridine yellow adsorption on silica, titania–silica, and titania surfaces were found according to Hess law from the total energy values for calculated models as follows:
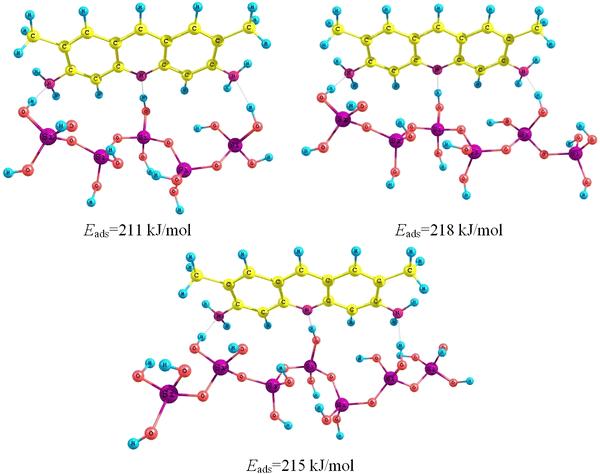
Adsorption complexes of acridine yellow in molecular form with silica surface which containing 5, 6, and 7 silicon–oxygen tetrahedraFig. 4
The silica surface was simulated with a unit of polysilicic acid of Si
5
H
12
O
16
containing 5 silicon–oxygen tetrahedra terminated by hydroxyl groups (Fig.
5
) [
41
].
Adsorption complexes of acridine yellow in molecular (
a
) and protonated (
b
) forms with silica surfaceFig. 5
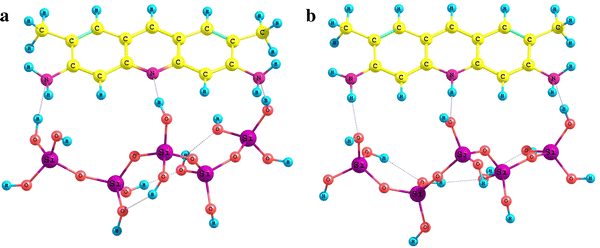
The titania–silica surface in the adsorption complex formed by Brønsted sites was simulated by a TiSi
4
H
12
O
16
structure containing one titanium–oxygen and four silicon–oxygen tetrahedra terminated by hydroxyl groups (Fig.
6
).
Adsorption complexes of acridine yellow in molecular (
a
) and protonated (
b
) forms with titania–silica surface with a weak Brønsted siteFig. 6
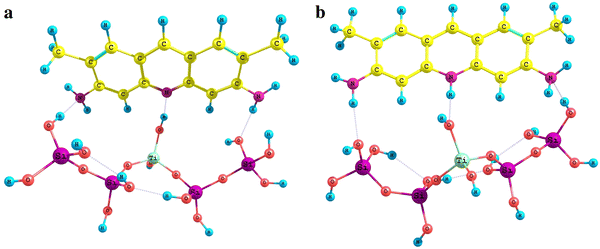
The model of titania–silica with strong Lewis acidic sites is formed due to water molecule addition to the previous model (Fig.
7
). In the model obtained, the hydrogen atom of water molecule is connected to the bridge oxygen atom (between titanium and silicon atoms) and the OH group joins titanium atom to enlarging its coordination number to five.
Adsorption complexes of acridine yellow in molecular (
a
) and protonated (
b
) forms with titania–silica surface with a strong Brønsted siteFig. 7
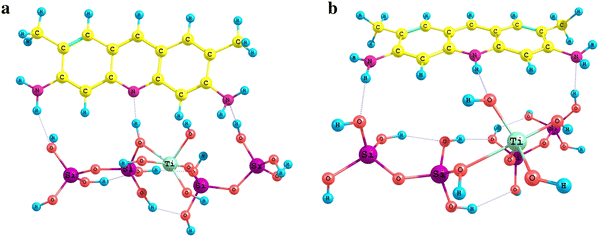
The model of titania–silica with weak Lewis acidic sites is formed from the previous model due to removal of the water molecule: TiSi
4
H
12
O
16
−H
2
O→TiSi
4
H
10
O
15
(Fig.
8
) [
42
] whereas titania surface was simulated by a fragment of anatase face(100) stabilized by four water molecules (Fig.
9
) [
43
,
44
].
Adsorption complexes of acridine yellow in molecular (
a
) and protonated (
b
) forms with titania–silica surface with a Lewis acid site Adsorption complexes of acridine yellow in molecular (
a
) and protonated (
b
) forms with titania surface [a fragment of anatase face(100)]Fig. 8
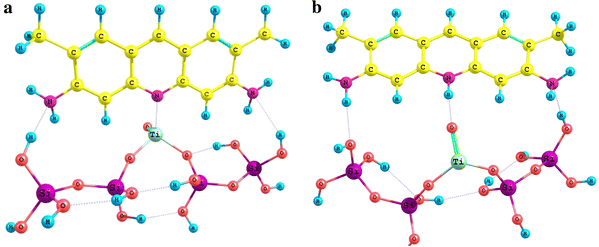
Fig. 9
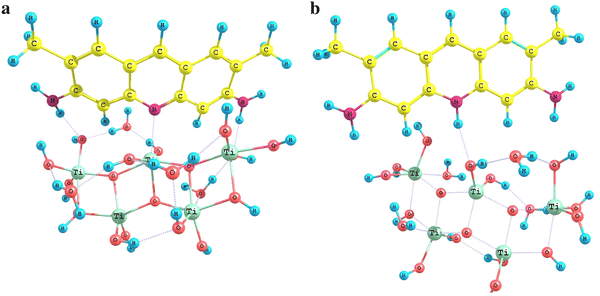
When acridine yellow molecule is adsorbed, its pyridine-type nitrogen atom forms bonds with both Brønsted and Lewis surface acidic sites of the substances mentioned above.
The results of calculations (Table
2
) show the adsorption energy of the molecular state of AY to be the greatest for the complex with titania–silica where Lewis acidic sites are present, the smallest one being related to the complex with titania what corresponds to the experimental data [
45
]. For AY–H
+
, the greatest adsorption energy relates to its interaction with titania surface, whereas the smallest one is a characteristic of titania–silica surface with Lewis acid sites.
Adsorption energy of acridine yellow on the surfaces of TiO
2
, SiO
2
, and titania–silica films, kJ/mol Adsorbent surface Forms of acridine yellow MOLECULAR Protonated Silica −211 −186 Titanium oxide, face(100) −158 −201 Titania–silica (strong Brønsted site) −235 −190 Titania–silica (weak Brønsted site) −223 −194 Titania–silica (Lewis site) −289 −178Table 2
Along with examination of the structures of adsorption complexes, the basicities have been analyzed of both amine-type and pyridine-type nitrogen atoms in acridine yellow molecule via comparing their proton affinity values [ 46 ]. The results of calculations prove the proton affinity of pyridine-type nitrogen atom (1146 kJ/mol) to be for 183 kJ/mol greater than that of amine-type one (963 kJ/mol).
Besides, two probable routes have been examined of acridine yellow photodegradation with similar reaction products.
The energy effects of related routes of photochemical destruction were evaluated analogously to the adsorption energy values; the total energy values of reactants and reaction products were calculated for their triplet states as we assumed the lifetimes of the structures mentioned in the excited singlet states to be too small for the reaction to take place as compared with that of the first triplet state [ 47 ].
The total energy values for reaction products were calculated for various multiplicities; the minimum total energy value was found to relate to the quadruplet for the first route whereas it related to triplet for the second one.
Table
3
shows the energy effects of the probable routes of gas phase acridine yellow destruction; it is seen from the results of calculations that photodegradation of free acridine yellow occurs according to the first route as independent on the calculation method used.
Energy effects of probable destruction routes of gas phase acridine yellow destruction, kJ/mol Photodestruction route ∆ B3LYP/6–31G(d,p) B3LYP-D3/6–31G(d,p) 469 426 598 557Table 3
Table
4
shows the energy effects for acridine yellow photodegradation on silica, titania–silica, and titania surfaces calculated with use of method B3LYP-D3/6–31G(d,p).
Energy effects of acridine yellow photodestruction on various surfaces (kJ/mol) Surface Destruction routes Photodestruction route Silica ( 457 634 ( 473 614 Titania–silica (Brønsted sites) ( 464 642 ( 476 622 Titania–silica (Lewis sites) ( 502 546 ( 570 613 Titania (Lewis sites) face(100) ( 424 493 ( 405 498Table 4
For adsorbed acridine yellow, each route of photodegradation can occur in two ways (Fig.
10
).
A scheme of the probable routes of acridine yellow photodestruction in adsorption complexesFig. 10
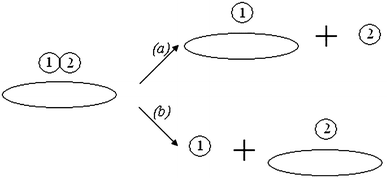
In the first case (Fig. 10 a), one reaction product (1) is adsorbed on the surface and another one (2) is in a free state. In the second case (Fig. 10 b), vice versa, the reaction product (2) is adsorbed on the surface whereas the product (1) is in a free state.
Under conditions of mass spectrometry experiment, elimination of methyl group (Fig.
11
a) or aminogroup (Fig.
11
b) from acridine yellow molecule can occur.
Elimination reactions of methyl (
a
) and aminogroup (
b
) from acridine yellow moleculeFig. 11
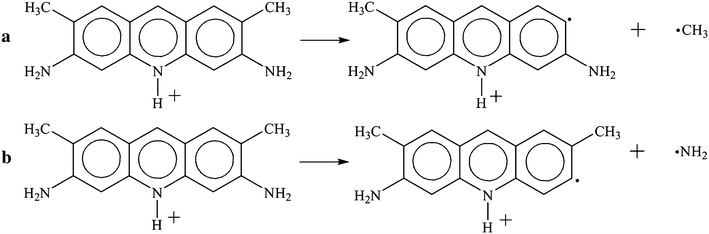
According to the results of quantum chemical calculations, the energy effect values of these two routes (Fig. 11 ) are almost the same as follows: methyl group separation needs 383.2 kJ/mol and that of amino group needs 387.1 kJ/mol. This fact testifies to uniform probability of acridine yellow destruction according to these two routes.
It has been found on the base of the results of quantum chemical calculations that acridine yellow is best adsorbed on titania–silica surface where Lewis sites play a key role whereas it is less adsorbed on titania (anatase) surface. The gas phase destruction of acridine yellow has been found to occur due to formation of the fragments with odd number of electrons. The destruction route preferred is independent on the surface nature. The photodegradation of acridine yellow has been found to occur most easily on the titania surface. In agreement with experiment, the calculated adsorption energy of acridine yellow molecular form is the greatest in case of titania–silica surface where Lewis sites are present, the last one relates to titania surface. For HAY + , the greatest adsorption energy is a characteristic of titania surface whereas the least one relates to titania–silica surface with Lewis acid sites. Thus, cleavage of methyl and amino groups from acridine yellow molecule occurs with the same probability.
Acridine yellow adsorption and photodegradation on the surface SiO2, TiO2 and SiO2/TiO2 films observed by UV–Vis and LDI-MS spectra
Adsorption isotherms of AY on the SiO 2 , TiO 2 and SiO 2 /TiO 2 films demonstrate Langmuir-like behavior. Enhanced adsorption of AY on SiO 2 /TiO 2 film coincides with high acidity, surface area and hydrophilicity of synthesized films (Table 1 ). AY adsorption on the surface of SiO 2 and TiO 2 films is reversible. Dye molecules can be desorbed easily from TiO 2 and SiO 2 films but cannot be leached from TiO 2 /SiO 2 surface by washing in great amount of pure water that can be interpreted in terms of protonation of AY through basic amino groups by acidic OH groups of mixed oxide surface [ 10 ] as was observed by us early for AO [ 48 ] and 9-aminoacridine on silica surface [ 49 , 50 ]. These data are in good agreement with our theoretical calculations.
A broad absorption band (
λ
max
= 430 nm) observed for AY starting aqueous solution (1 × 10
−4
M) used for adsorption is a superposition of the spectra of its monomeric and dimeric forms. Dilution of this solution (up to concentration of 1 × 10
−6
M) leads to short-wavelength shift of absorption band due to decreasing of dimers concentration [
11
]. The absorption maximum at 444 nm corresponds to absorption of the monomeric form of AY in solution. However, as was shown for fluorescence excitation spectra of AY adsorbed on SiO
2
surface [
51
], the dye concentration increase causes a bathochromic shift of the excitation maximum and decreases the emission intensity. It was explained by the formation of various-type associates in the solution and on the oxide surface. The developed porous structure of films is geometrically favorable for the adsorption of two and more dye molecules. Associates are formed [
48
,
49
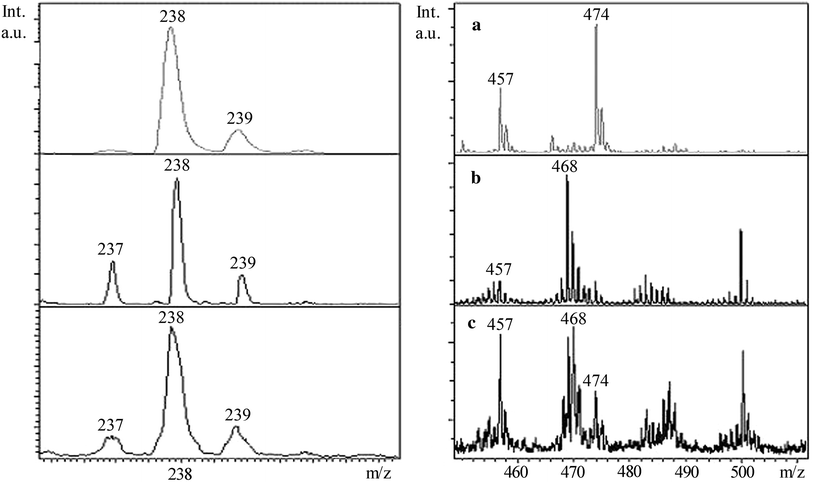
] starting from the covering of ~0.01 % of monolayer what is also confirmed by the presence of peaks with large masses in initial (measured before irradiation) mass spectra (Fig.
12
).
Initial LDI-MS spectra of AY adsorbed on the A-TiO
2
, B-SiO
2
and C-SiO
2
/TiO
2
surfacesFig. 12
The absorption maximum positions of AY dye spectra before irradiation vary with the different synthesized films (Figs.
13
,
14
,
15
a). Monomolecular adsorption of AY with maximum near 442 nm is observed on TiO
2
surface. The short-wave shift of AY absorption bands on the surface of SiO
2
(
λ
max
= 430 nm) and TiO
2
/SiO
2
(
λ
max
= 435 nm) films with stronger acid sites can be a result of both protonation and dimerization of dye molecules. It is known that protonation of acridine dyes proceed via first proton coordination to nitrogen atom of aromatic ring, causing a bathochromic shift of the absorption spectrum [
11
]. Further protonation of AY through basic
N
-dimethylamino groups with formation of double ions is accompanied by a shift of the absorption band towards shorter wavelengths. The proton affinity of pyridine nitrogen atom was found theoretically to be 180 kJ/mol greater than that of amine atom.
Evolution of absorption (
a
) and LDI (
b
) spectra after 30 min irradiation of AY on the surface of mesoporous TiO
2
films. Peak intensity of some AY fragments during UV irradiation (
c
) Evolution of absorption (
a
) and LDI-MS (
b
) spectra after 120 min irradiation of AY on the surface of mesoporous SiO
2
films. Peak intensity of AY fragments during UV irradiation (
c
) Evolution of absorption (
a
) and LDI-MS (
b
) spectra after 90 min irradiation of AY on the surface of mesoporous SiO
2
/TiO
2
films (
inset
shows negative mode). Peak intensity of AY fragments during UV irradiation (
c
)Fig. 13
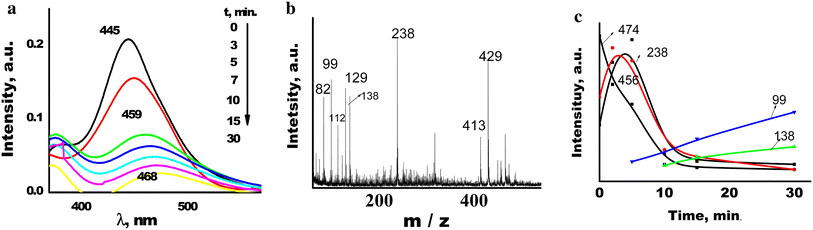
Fig. 14
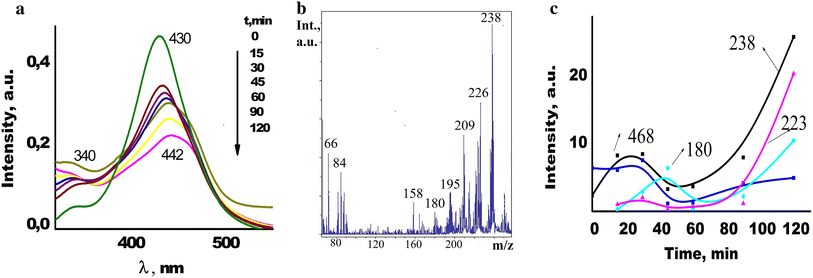
Fig. 15
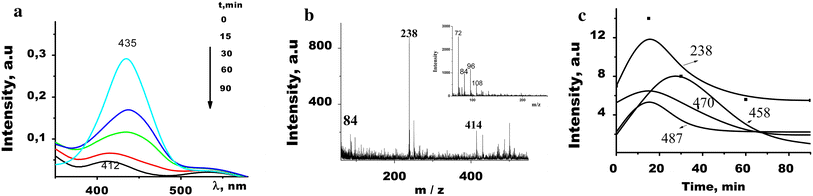
Irradiation of adsorbed AY in the presence of oxygen leads to a decrease in adsorption intensity and to a long-wave shift of the main absorption band of dye on the SiO 2 and TiO 2 surfaces, whereas on the SiO 2 /TiO 2 film a short-wave shift up to 412 nm takes place. Along with a decrease in the main absorption band intensity due to irradiation, an increase in absorption in the 330–340 nm spectral regions (acridine-like absorption) for all the samples is observed. Acridine yellow is capable of forming protonated ions, dimers, and aggregates with corresponding spectral transformations; it is almost impossible to separate these processes relying only on the absorption measurements. Therefore, LDI mass spectra of the samples exposed to UV irradiation for different time intervals (from 0 to 120 min) were obtained and analyzed. It has been found that exposure of three types of films with AY adsorbed to UV light leads to characteristic and reproducible changes in the mass spectra probably associated with different stages of photodegradation of the dye.
Major peaks of the dye cation M + with m/z 237 and peaks in the mass region close to dimer mass were registered in the LDI mass spectra (Fig. 12 , positive ions) of the initial air-dried films with adsorbed AY. The monoisotopic peak of the cation M + ( m/z 237) is accompanied by peaks of [M+1] + ( m/z 238) and [M+2] + ( m/z 239) with intensities higher than the value expected from the contribution of 13 C isotope. The molecular peak M + is negligible in LDI spectra of AY adsorbed on TiO 2 films. In the region of large masses, several peaks of varying intensity were observed. A pronounced peak with the mass equal to that of the dimer 2M + ( m/z 474) was present in LDI spectra of AY adsorbed on TiO 2 and TiO 2 /SiO 2 films.
The magnitude of the peak of protonated form [M+1] + ( m/z 238) increases dramatically in the spectra of all investigated samples under the influence of UV irradiation (5 min for AY adsorbed on TiO 2 and 15—on SiO 2 and TiO 2 /SiO 2 films). The optical density at the absorption band maximum decreased by 50 % after irradiation of AY adsorbed on TiO 2 film for 30 min. Since the electron transfer into the TiO 2 conductive band with the cation-radical formation is the first stage of photocatalysed destruction of the dye, it was highly probable that further photodegradation of AY occurred through a radical mechanism [ 16 ]. Thus, radicals formed after the loss of an amino group ( m / z = 223) were bound to dye molecules to form dimeric aggregates. This was accompanied by a long-wavelength shift of the absorption band maximum to 459 nm (Fig. 13 a). Peaks of the corresponding masses ( m / z = 456, 460, 468, 474) increased in the mass spectrum (Fig. 13 c). This process may be explained by protonation of the side amino groups of AY despite the fact that the short-wavelength shift in the absorption spectrum was masked by absorption of the dimers [ 13 , 14 ]. UV radiation affected mainly highly delocalized electrons of C=C- and C–N- bonds in the dyes leading to photodegradation.
As it has been mentioned above, dye monomers and dimers are present on the film surface. With the onset of irradiation (the first 2–5 min), multiple processes run as follows: (1) breaking of the dimer followed by its protonation which contributes to the sharp increase in the peak of m/z 238 (Fig. 13 c); (2) increase in the surface hydrophilicity that not only leads to the formation of dimers but also associates with the masses over m/z 650–700. In the electronic spectra, this process manifests itself in a shift of the absorption band maximum to longer wavelengths [ 12 ] characteristic of the formation of j-aggregates (brickwork) (3) decomposition of AY with the formation of deaminated and demethylated short-lived species (not recorded in the mass spectra) binding to the protonated cation as judged from the increase in the m/z 456 peak (Fig. 13 c).
This observation agrees well with the results of our theoretical calculations that show similar values of cleavage energy of amino and methyl group; such a cleavage is the first step of AY degradation. Subsequent irradiation leads to a monotonic decrease in the intensity of peaks with large masses in the LDI spectra as well as emergence and increase in peaks with masses less than that of the AY cation (138, 122, 113, 99, 83, 67, etc.), i.e., degradation and mineralization proceed (Fig. 14 c). These masses relate to the AY destruction routes predicted theoretically.
While the processes observed on the TiO 2 film are photocatalytic, a slow photolytic decomposition of acridine yellow is observed on the surface of SiO 2 film (Fig. 15 ).
In this case, multiple stages dominating over other processes can be identified: (1) detachment of amino (peak m/z 223) and methyl groups and formation of acridine (peak m/z 180) appeared after 15 min of UV exposure (Fig. 15 b). It should be mentioned that after 120 min of exposure the mass spectrum still contains peaks corresponding to all stages of cleavage of two amino groups and two methyl groups with the masses of 238, 223, 208, 194, 180; (2) formation of a huge number of dimer associates with the masses of m/z 450–500 difficult to identify and group of peaks with masses of m/z 700 observed after 45 min of irradiation; (3) decomposition of associates with formation of a molecular cations and degradation products with masses less than m/z 180 so indicating destruction of acridine and benzene rings ( m/z 158, 122, 108, 88, 72, 66, 64) (Fig. 15 c).
Most peaks in LDI-MS spectra of AY adsorbed on the mixed oxide surface are much less intensive (under the same laser power) than those on parent oxide surfaces. It is worse noting some peculiarities of photodegradation processes occurred on the TiO 2 /SiO 2 film surface. It is essential that during the irradiation no peak with masses more than m/z 500 was registered; most decomposition products were observed in the mass spectra of negative ions (Fig. 15 , inset). The constants of AY photodegradation rate k 0 evaluated from an inclination of the kinetic curves given in the Table 1 on TiO 2 /SiO 2 are almost six times higher than that for SiO 2 film.
Based on theoretical prediction and investigation of absorption spectra and LDI-MS data on AY degradation on the surfaces of mesoporous TiO
2
, SiO
2
and TiO
2
/SiO
2
films, following scheme (Fig.
16
) illustrating these processes can be proposed:
Scheme of AY photodegradationFig. 16
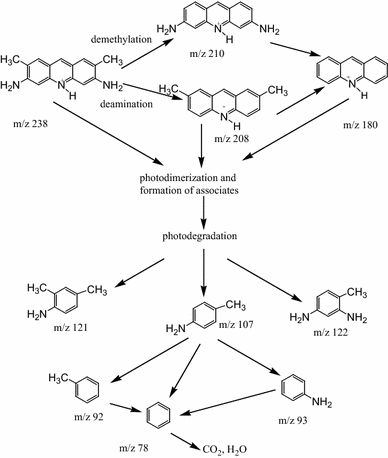
Photodegradation of AY dye adsorbed on the film surface is characterized by different ways of bleaching—a fast process involving photocatalysts on TiO 2 and TiO 2 /SiO 2 films containing anatase nanoparticles (half-time of degradation 4 and 15 min, respectively), or a slow process (more than 90 min) of chemical oxidation of protonated via aminogroup acridine yellow on silica surface in air.