Graphic abstract

Selenium nanoparticles (SeNPs) have the potential to be used for various applications. Therefore, they have attracted more attention in recent years and several synthesis methods have been exploited. Green synthesis using plant extracts has gained popularity because it requires non-toxic solvents and moderate temperatures. Furthermore, it is environmentally friendly and uses a reducing agent that is easily accessible and biodegradable. In this review, we present recent reports concerning the capability of different plant materials for the biosynthesis of selenium nanoparticles. The synthesis conditions (temperature, time, selenium precursor, and extract concentration) are discussed in combination with the characteristics of the obtained product (composition, size, shape, stability). The application of synthesized selenium nanoparticles was briefly presented.

In recent years, there has been growing interest in selenium due to its important function in human health [ 1 , 2 ]. Selenium plays a key role in several major metabolic pathways, such as thyroid hormone metabolism and immune functions. It also prevents cellular damage induced by free radicals by incorporation into antioxidant enzymes. Se deficiency has been linked to a range of serious conditions such as cancer, cardiovascular and inflammatory diseases [ 3 ]. However, long-term Se supplementation or higher concentrations could cause toxicity [ 4 ]. In environmental and biological samples, selenium exists in inorganic forms, mainly as selenite Se(IV) and selenate Se(VI) ions, and as organic species with direct Se-C bonds (methylated compounds, selenoamino acids, selenoproteins, and their derivatives). Some plants have the ability to accumulate inorganic Se forms from soil and transform them into bioactive organic species [ 5 ]. The bioavailability and toxicity or the antioxidant and pro-oxidant effects of selenium depend on its chemical form.
Recently, nanoparticles of elemental selenium (SeNPs) have attracted attention. Their synthesis and application continue to be reported and discussed [ 6 , 7 , 8 , 9 , 10 – 11 ]. SeNPs differ from the properties of its corresponding bulk materials, similar to other nanoparticles. These properties as well as morphology and size depend on several parameters including a method of synthesis, use of surfactants or additives, reaction temperature, and time [ 11 ]. Moreover, their surface can be coated with surfactants and polymers [ 12 ]. The toxicity reported for SeNPs was lower in comparison to inorganic and organic forms of selenium [ 13 ].
Three main methodologies have been applied for the preparation of selenium nanoparticles covering physical, chemical, and biological methods. In the physical approach for SeNPs synthesis, pulsed laser ablation [ 14 , 15 – 16 ], vapour deposition [ 17 ], hydrothermal [ 18 ] and solvothermal methods [ 19 ] were used. The pulsed laser ablation method has the advantage over other methods due to the lack of contamination with chemical reagents, easy collection of NPs by centrifugation, and their high stability.
The most commonly used method for the preparation of SeNPs is a chemical reduction of inorganic selenium forms as the precursors. Ascorbic acid [ 20 , 21 , 22 , 23 – 24 ], glucose [ 24 , 25 – 26 ], fructose [ 27 ], cysteine [ 28 , 29 ], glutathione [ 30 ], sodium metasulfite [ 31 ] and ionic liquid 1-ethyl-3-methylimidazolium thiocyanate [ 32 ] have been used as reducing agents, usually in the presence of stabilizing agent to prevent aggregation of nanoparticles. Water-soluble polymers [ 22 ], natural polysaccharides [ 22 , 33 ], carboxymethyl cellulose [ 34 ] or bovine serum albumin [ 35 ] have been used for this purpose. However, some residuals of these chemicals limit the applications of the formed SeNPs in the pharmaceutical and medicinal areas. Yu et al. [ 36 ] synthesized different Se nanostructures, such as nanoball, nanotube, and multi-armed nanorods, by reducing H 2 SeO 3 with L-asparagine in polyethylene glycol solution. The reaction was supported by microwave irradiation at 100 °C. The diameter and morphology of SeNPs were controlled by the ratio of L-asparagine/H 2 SeO 3 and the microwave irradiation time. Extending reaction time resulted in a higher diameter size of the obtained SeNPs and they were aggregated after 15 min of microwave treatment [ 36 ].
An alternative green approach towards the synthesis of SeNPs has been reported by using the reducing potential of various microorganisms [ 37 ]. Different groups of bacterial strains [ 39 , 40 , 41 , 42 , 43 – 44 ] and fungi [ 44 , 45 , 46 , 47 – 48 ] have the ability to reduce selenite or selenate to nano selenium as a method of detoxification. SeNPs may be formed both within the bacterial cells and/or extracellularly. All aspects of the investigations regarding the microbial transformations of selenium species could be found in the relevant reviews, where different mechanisms are discussed [ 37 , 49 , 50 – 51 ]. The degree of control over the size and shape of SeNP is high in the chemical synthesis while using microorganisms the spherical and polydispersive SeNP with sizes in the range of 50–500 nm were usually obtained [ 37 ].
The utilisation of water plant extracts represents also a better alternative to chemical methods for the synthesis of selenium nanoparticles. That approach requires non-toxic solvents, mild temperatures, and application of the reducing agents that are easily accessible, cheap, biodegradable, and not harmful to the environment. It also reduces the high cost of microorganism’s isolation and a final SeNPs purification, when they will be applied in biomedical sectors. Such biogenic methods of synthesis of SeNPs are becoming preferred over the conventional chemical and physical methods due to their reduced toxicity towards the environment which uses living organisms such as plants, microalgae, and other microorganisms. Selenium nanoparticles produced via the green synthesis method can be an alternative to antibiotics. SeNPs showed an antibacterial effect toward standard and antibiotic-resistant phenotypes of Gram-negative and Gram-positive bacteria in a dose-dependent manner [ 38 ]. The aim of this review is to show the current knowledge concerning the plant materials' capability for the biosynthesis of selenium nanoparticles and their applications.
Several plants that have been already reported for the preparation of SeNPs are presented in Table
1
and the schematic process of their synthesis is depicted in Fig.
1
. In the published papers there is no explanation why this particular plant was chosen for the preparation of selenium nanoparticles. It can be inferred that the determining factor for such choice was the easy availability of the raw materials and some of these plants have traditional and pharmacological uses. Moreover, agricultural waste, such as fruit peels, could be used for synthesis. There is a maximum probability of succeeding by using plant materials which are rich in polyphenols, flavonoids, alkaloids, polysaccharides, saponins, etc. since they are very good reducing and stabilizing agents. Some literature works reported the content of phytochemicals present in prepared plant extracts such as total phenolic compounds and flavonoids [
57
,
77
,
81
], tannins [
57
] and polysaccharides [
81
] or phytochemical screening showing only qualitatively the presence of main active components [
58
,
61
,
72
,
76
].
Synthesis of SeNPs using different plants Plant Extraction Preparation of SeNPs Refs. Hawthorn 10 gof dried fruits + 200 mL H2O, refluxed for 2 h Extract (2 mg mL−1) + Na2SO3 (10 mol L−1) stirred for 12 h 52 Dried raisin ( Refluxed for 30 min with water 10 mL extract + 90 mL H2SeO3 (4 × 10–5 mol L−1) refluxed for 15 min 53 Lemon plant 5 g + 20 mL of Tris–Cl (pH 7.5) 2 mL extract (from 0.5 g) + 20 mL of Na2SO3 (10 mmol L−1), shaken for 24 h at 30 °C in dark conditions 54 100 g of dried flowers + 100 mL water, heated at 70 °C for 5–10 min 10 mL of extract + 90 mL 10 mmol L−1 Na2SeO4 shaken at 36 °C for 5 days 55 Ginger fruit – 1% extract + Na2SeO3 (10 mmol L−1) at room temperature stirred for 75 h 56 Ashwagandha 10 g dried leaves + 500 mL water, boiled for 15 min 0.1 mL extract + H2SeO3 (50 mmol L−1) at room temperature, centrifuged, washed with water and acetone 57 Fenugreek ( 1% extract in water, stirring for 15 min 1 mL of extract + 10 mL H2SeO3 (30 mmol L−1) + 200 µL ascorbic acid (0.04 mol L−1), room temperature 24 h, washed with water, then ethanol 58 Avaram 5 g of powdered leaves + 100 mL water, soaked for 30 min Extract + 100 mL Na2SeO3 (10 mmol L−1), incubated 48 h at room temperature 59 Arauna 5 g + 20 mL of TrisCl (pH 7.5) 2 mL of extract + 20 mL of Na2SeO3 (10 mmol L−1), shaken at 30 0C for 72 h in dark conditions, washed with ethanol 60 Java tea 50 g of dried leaves + 300 mL water, stirring for 15 min, kept for 24 h 1 mL of extract + 10 mL of H2SeO3 (10 mmol L−1) + 200 µL ascorbic acid (40 mmol L−1), 24 h at room temp. washed with water and absolute alcohol 61 Lavender leucas 10 g of dry plant + 400 mL water, boiled for 5 min 2 mL of extract + 10 mL of H2SeO3 (50 mmol L−1) + 200 µL of ascorbic acid (40 mmol L−1) 62 30 g of fresh leaves + 20 mL water 25 mL of Na2SeO3 (5 mmol L−1) + plant extract until color was changed, later kept for 72 h 63 4.69 g of dried leaves + 100 mL of boiling water, 5 min 4.92 mL of extract + 133.03 mL Na2SeO3 (10 mmol L−1) at 121 °C for 15 min 64 Garlic 10 g of garlic buds + water to make a thin paste 25 mL of Na2SeO3 (5 mmol L−1) + extract until color was changed, kept for 72 h. in dark 65 Garlic Garlic buds + water to make a thin paste 2 mL of extract (2%) + 8 mL of H2SeO3 (30 mmol L−1) + 200 µL of ascorbic acid (40 mmol L−1), 48 h for incubation 66 Garlic 10 g of garlic buds + water to make a thin paste Extract was added dropwise to 25 mL Na2SeO3 (5 mmol L−1), kept 72 h in dark 67 Garlic 5 g of crushed gloves macerated with 30 mL of Tris–Cl (pH 7.5) 20 mL of extract + dropwise 20 mL of Na2SeO3 (10 mmol L−1), 5–7 days at 36 °C 68 Garlic ( – 0.06 g of garlic extract + 20 mL of Na2SeO3 (10 mmol L−1), incubated 4, 24, 48 and 72 h 69 Walnut ( 1 g of leave powder + 100 mL water, boiling for 5 min 5 mL of extract + Na2SeO3 (10 mmol L−1), heated using microwave oven (800 W for 4 min) 70 Horseshoe geranium ( 2 g of powdered leaves + 100 mL of boiling water, mixed for 5 min 1.48 mL of extract + 15 mL of Na2SeO3 (10 mmol L−1), heated using microwave oven (800 W for 4 min) 71 2 g of powdered aerial part + 200 mL water, boiled for 1 h 75 mL of extract + 25 mL of Na2SeO3 (10 mmol L−1), incubated in dark for 24 h 72 10 g of flowers + 100 mL water, heated at 90 °C for 5 min 10 mL of extract + 90 mL of Na2SeO3 (10 mmol L−1), kept at 36 °C for a week 73 Orange peel 50 g of grounded peels + 150 mL water, boiled for 15 min 50 mL of extract + 5 mL of Na2SeO3 (100 mmol L−1) 74 10 g of powdered leaves + 100 mL water, boiled at 60 °C for 5 min 12 mL of extract + 88 mL of selenium powder (1 mmol L−1) 75 Mountain persimmon ( – 10 mL of extract + 100 mL of H2SeO3 (300 mmol L−1) + 2 mL of ascorbic acid (400 mmol L−1). Incubation for 24 h 76 100 g of edible grounded fruit + water (1:2, w/v) 2 mL of extract + 10 mL of Na2SeO3 (100 mmol L−1), in dark for 24 h 77 25 g of dry leaves + 100 mL of water, boiled at 100 °C Se powder (0.6 mol L−1) + Na2S (3.87 mol L−1) dissolved in 10 mL of water, refluxed at 90 °C for 1 h. Then the formed SeS2 (0.5 mol L−1) + 100 mL of water + 5 mL of extract, stirred for 10 min at room temperature 78 Drumstick ( Leaves soaked in 100 mL of water at 67 °C for 4 h Selenium salt (1 g/100 mL) + extract, kept for 24 h at room temperature 79 Guava ( 10 g of leaves + 100 mL 60% ethanol, boiled for 2 min 100 mL of extract + 900 mL of Na2SeO3 (25 mmol L−1), at 60°, incubating for 3 h 80 Cacao bean shell ( 6 g of grounded material + water, adjusted to pH 2, heated in the microwave oven (500 W) at 100 °C for 5 min 50 mL of extract + 0.14 g of Na2SeO3, heating in the microwave oven (788.6 W) for 15.6 min 81 Scheme for the synthesis of selenium nanoparticles using plant extractTable 1
Fig. 1
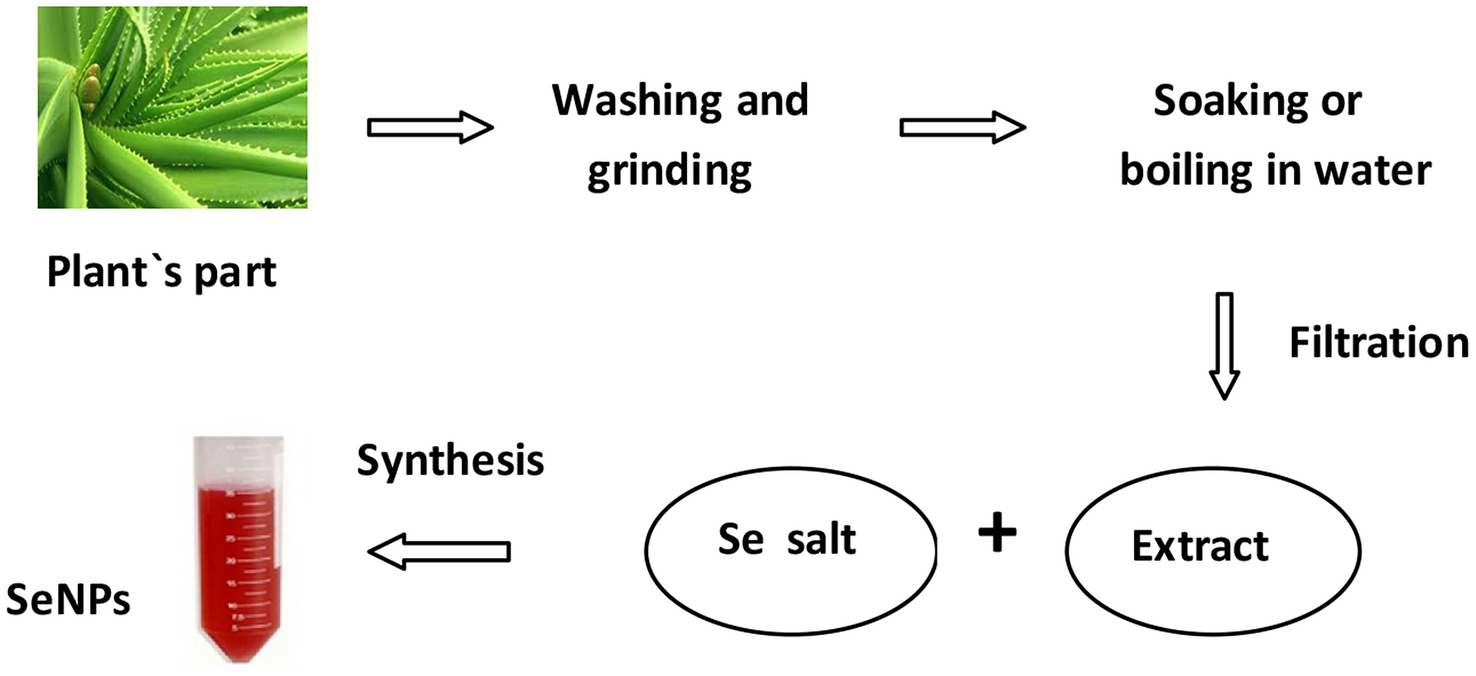
Generally, the chosen part of the plant (leaves, buds, fruit, peel, nuts, seed or pulp) was washed thoroughly with deionised water, dried, ground in a mortar and then boiled with water. In some procedures simply soaking process under the continuous stirring condition without heating was applied [ 54 , 57 , 58 , 59 , 60 – 61 , 63 , 68 , 71 , 76 ]. Garlic cloves were crushed with a mortar and diluted with water [ 65 , 66 – 67 ] or Tris–Cl buffer (pH 7.5) [ 68 ] to make a thin paste. Microwave irradiation was applied for extraction from cacao bean shell [ 81 ]. After this, the solution was filtered or centrifuged and the liquid part was used in the experiments. The differences amongst the prepared plant extracts exist as a result of using different kinds of plants and their different parts as well as extraction temperature and time; e.g . extract prepared from Java tea was filtered only after 24 h [ 60 ], while lavender leucas extraction was conducted within 5 min [ 62 ].
In SeNPs biosynthesis process, reducing and stabilizing agents are the main factors. Biomolecules present in the extracts of plants, such as polysaccharides, phenolic compounds, flavonoids, tannins, saponins, amino acids, enzymes, proteins, and sugars, are known to be potential reducing agents of selenium and have medicinal importance. Nanoparticles tend to agglomerate, thus, a stabilizer is often used to inhibit their over-growth by coating with a single layer of polymer or surfactant which reduces the interactions between nanoparticles [ 22 , 30 , 53 , 59 ]. Some authors reported that the used plant extracts contained phytochemicals that exhibited stabilizing properties [ 24 , 25 , 28 , 32 , 57 ].
In the synthesis of nanoparticles, the main goal is their formation with minimum particle size and maximum stability [ 82 ]. Many factors affect the obtained nanoparticles, including temperature, pH, extract concentration, type, and concentration of the used precursor. In some papers response surface methodology (RSM) was applied to develop the empirical models for the prediction of the optimal synthesis conditions [ 64 , 71 , 81 ].
In a typical experiment, selenium precursor (sodium selenite or selenate, selenous acid, sodium selenosulphate) was mixed with the plant extract solution at different ratio. The addition of ascorbic acid was used in some procedures as an initiator of reduction reaction [ 58 , 61 , 62 , 66 , 76 ]. The reacting solution was stirred at room temperature in different time intervals; from 12 h [ 52 ] to 72 h [ 53 , 57 , 60 , 62 , 64 ] and even for 5–7 days [ 68 , 73 ], sometimes in the dark. Another group of proposed procedures includes heating the mixture at the desired temperature and time. During that incubation process, the colour of the reaction media changes to red, indicating the formation of Se nanoparticles. Finally, the SeNPs can be separated by centrifuging at high speed, washed thoroughly in water and solvent.
The proposed methods for selenium nanoparticles using plant extracts show similarity in the synthesis procedure with little modifications (Table 1 ). Deepa and Ganesa [ 73 ] used Na 2 SeO 4 as the selenium precursor for reduction reaction, while most often Se(IV) species are applied. This fact was explained by the authors by a long incubation time of 1 week for SeNPs preparation using extract from the flower of Catharanthus roseus . Santanu et al. [ 74 ] reported that the time required for conversion of selenium selenite to SeNPs was indirectly related to the reduction ability of orange peel extract. The extraction process was found to be the most effective when conducted for 15 min at 40 °C and pH 4.
Melinas et al. [ 81 ] proposed the extract from Theobroma cacao L. bean shell as a reductant and capping agent for SeNPs synthesis. The kinetic of reaction was increased due to the use of microwave irradiation with rapid initial heating. It was concluded that SeNPs crystalline size was not significantly affected by time and microwave power and only low amounts of the Se precursor were needed to obtain small-sized nanoparticles. The optimized through RSM synthesis parameters were: 15.6 min, 788.6 W, 0.14 g of sodium selenite and 50 mL of extract solution.
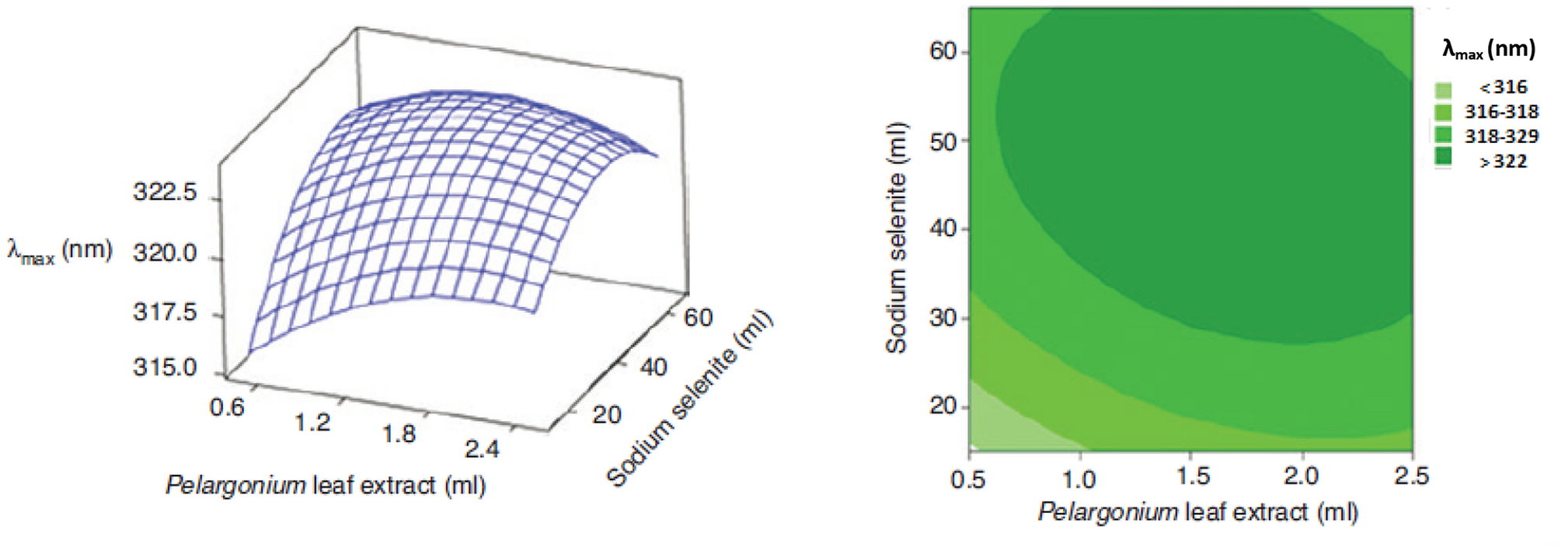
Fardsadegh et al. [
71
] also used RSM methodology to obtain the optimal condition for fabrication of selenium nanoparticles using
Pelargonium zonale
leaf extract. They evaluated the effects of selenium precursor amount and plant extract on the λ
max
value, as higher λ
max
correlated to larger particle size. The increase in the amount of Na
2
SeO
3
at a constant amount of the plant extract, generated the increase of λ
max
(Fig.
2
). Similar trend was observed for increasing amount of extract at constant amount of Na
2
SeO
3
solution.
Surface and contour plots for
λmax
of the synthesized SeNPs as a function of amounts of Na
2
SeO
3
solution and
Pelargonium zonale
leaf extract. Reprinted with permission by De Gruyter GmbH from Ref. [
71
]Fig. 2
The synthesis with
Aloe vera
leaf extract was conducted under subcritical water conditions due to its capability to generate simultaneously high pressure and high temperature [
64
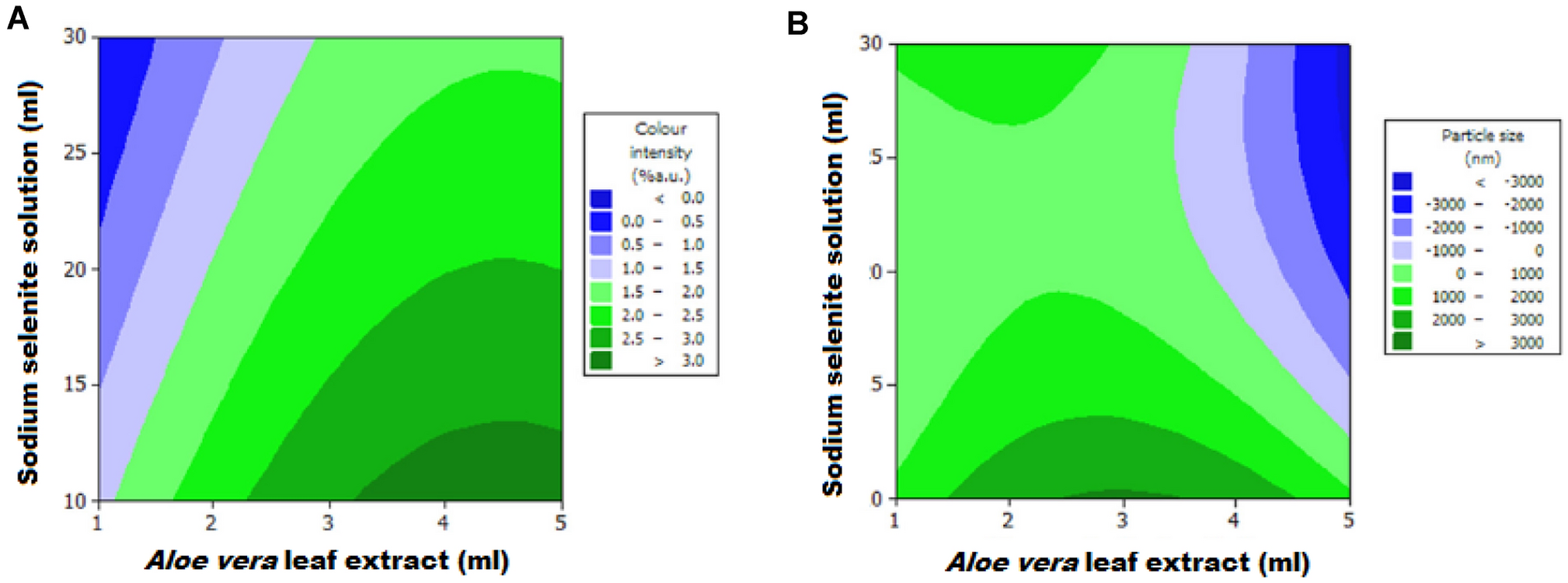
]. This hydrothermal synthesis was completed within 15 min. Additionally, central composite design with RSM models were used to evaluate the effects of synthesis parameters on the colour intensity and particle size (Fig.
3
). It was found that at a constant amount of Na
2
SeO
3
, the increase in the amount of extract also caused the increase in SeNPs concentration, observed as the increase in colour intensity. On the other side, at a constant amount of
Aloe vera
extract, the colour intensity decreased with the increase of the amount of Na
2
SeO
3
solution. Such result was explained by the fact that in the small amounts of plant extract, reducing agents react with selenium ions completely. In fact, the increase in Na
2
SeO
3
solution amount was causing the increase in the amount of Se ions, while the number of synthesized SeNPs was constant. Their concentration decreased due to an increase in the volume of the reaction solution.
Contour plots of (A) color intensity and (B) particle size of the solution containing SeNPs as a function of the amount of Na
2
SeO
3
solution and
Aloe vera
leaf extract. Reprinted with permission by De Gruyter GmbH from Ref. [
64
]Fig. 3
The prepared SeNPS were characterized by various spectroscopic and microscopic methods to evaluate their elemental composition, exact morphology and also other physicochemical properties. UV–Vis absorption spectra were recorded for the indication of the Se nanoparticles formation. The color change of the reaction mixture from uncoloured to reddish confirms the synthesis of SeNPs. It was further validated by the progressive rise in optical phenomenon within the characteristic peak between 200 and 400 nm with the increase in reaction time. In the absorption spectra of SeNPs the red-shift occurs because the particle sizes increase [ 52 , 53 , 66 , 68 ]. It is worth mentioning that SeNPs synthesized using the reducing potential of various bacteria gave a characteristic peak at 450–600 nm. It had be explained by the diversity of enzymes which catalyze the reduction reaction [ 43 ]. Atomic absorption spectroscopy was also used to study the conversion of selenium ions at various time intervals by determining its remaining concentration in supernatant [ 50 ].
Scanning emission microscopy (SEM) and transmission electron microscopy (TEM) measurements revealed their size, shape, agglomeration and distribution. Dynamic light scattering (DLS) also helps in the measurement of the particle size and distribution. In some cases, the disagreement between the sizes obtained by TEM and DLS was observed due to the fact that TEM analyzes the metallic core while DLS measures the hydrodynamic volume.
Energy dispersive spectroscopy (EDS) was used for the examination of elemental composition and purity of nanoparticles. The highest content of selenium (82%) in nanoparticles was obtained using the leaf extract of Cassia auriculata [ 59 ], followed by Clausena dentate (72.6%) [ 75 ] and Emblica officinalis (61.6%) [ 77 ]. EDS analysis of SeNPs revealed the proportion of Se equals to 55% when the extract of garlic bulbs was utilized [ 63 , 65 ]. Similar values of 54% were reported for ginger fruit [ 56 ] and herb Withania sonnitfera extracts [ 57 ]. The other peaks being observed in EDS spectrum were carbon, oxygen, nitrogen, and sodium.
Fourier transform infrared (FTIR) spectroscopy was used to confirm the involvement of O–H, N–H, C=O and C–O functional groups during the formation of SeNPs, which were associated with bioactive molecules capping their surface [ 53 , 54 , 55 , 56 , 57 – 58 , 62 , 63 , 64 , 65 , 66 , 67 – 68 , 71 , 75 , 76 – 77 ]. The peaks at 1375 cm −1 , 1030 cm −1 , 1462 cm −1 and 1250 cm −1 representing phenolic OH, aromatic in-plane C–H bending, asymmetric C-H bending (in CH 3 and –CH 2 –) and secondary OH, respectively can be found after the green synthesis of SeNPs. The peaks at 2840 and 2930 cm −1 representing ether-methoxy-OCH 3 groups, show the presence of the biopolymer lignin associated with SeNPs [ 53 ]. Also, novel composite containing TEMPO cellulose, chitosan, starch and selenium nanoparticles was synthesized confirming its structure by FTIR [ 78 ]. X-ray diffraction (XRD) pattern reflects the morphology of selenium nanoparticles and their crystalline nature. [ 42 , 48 , 53 , 60 , 73 , 75 , 76 – 77 ]. Raman spectroscopy analysis provides the unique feature of vibrational characteristics of crystal or amorphous nature of SeNPs [ 66 , 77 , 84 ].
The stability of selenium nanoparticles was evaluated by measuring the zeta potential. SeNPs synthesised by plant extracts are covered by a bioorganic layer comprising proteins, polysaccharides and lipids, with a significant proportion of ionised carboxylic groups. These groups, which are typical both of side-chains of some amino acid residues and carboxylated polysaccharides, are responsible for the negative values of SeNPs zeta potentials. The obtained magnitudes deliver information on particle stability. When this absolute value is higher than 30 mV, the colloidal solution can be considered stable, and there is no agglomeration or flocculation [
85
]. From literature, it may be observed that the lowest values of zeta potential equals to − 36 V were reported for SeNPs synthesized using the extract from lemon plant [
53
] and ginger fruit [
54
], followed by the extract of Java tea (− 34.9 V) [
59
] and cacao bean shell (− 28.6 V) [
81
]. The synthesized SeNPs with the spherical average of 24.3 nm found to be stable for more than three months without forming any aggregates [
54
]. Mellinas et al. [
82
] observed a significant interaction between the reaction time and the amount of Na
2
SeO
3
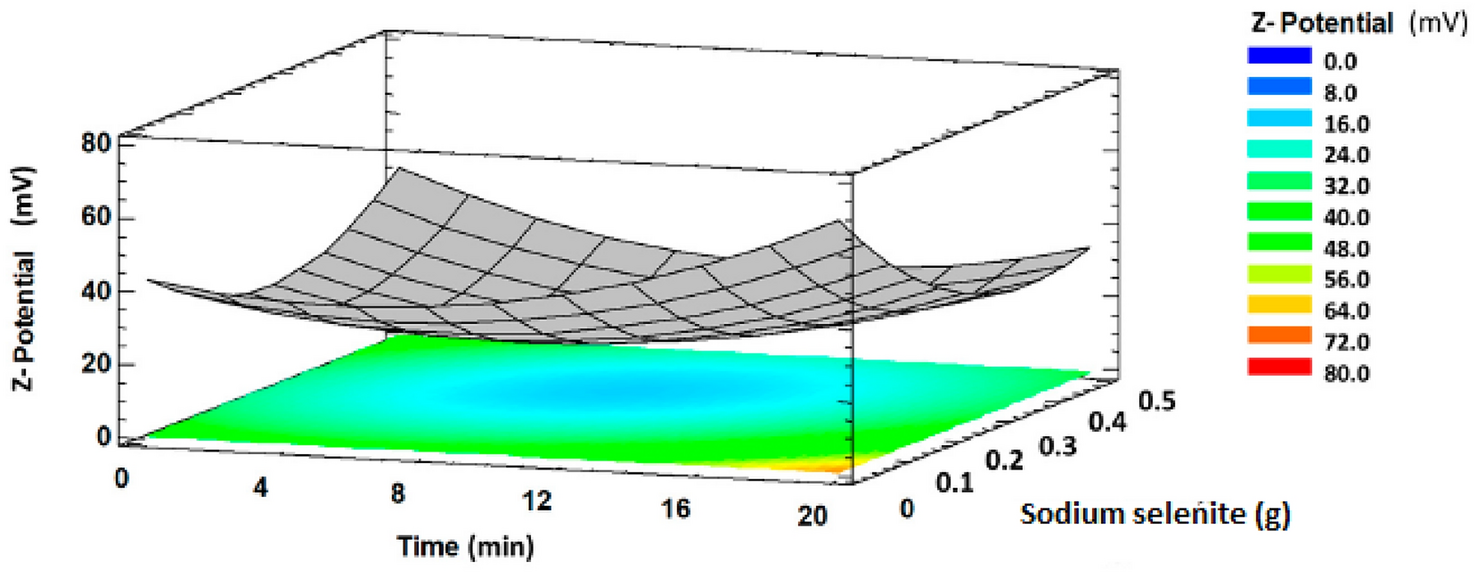
(used for the preparation of the precursor solution) on Z-potential. As shown in Fig.
4
, the Z-potential as a function of these variables has a minimum. Initially, the Z-potential value decreased with increasing reaction time and the concentration of precursor. After reaching the minimum, the Z-potential increased with increasing both reaction time and the amount of sodium selenite. This was explained by the increase of the free energy of the system that favours the aggregation of nanoparticles and then the decrease in their stability. When the time of synthesis was increased and the amount of Se precursor decreased, SeNPs showed higher stability, which was largely attributed to the stabilizing effect of compounds present in the plant extract.
Response surface plot showing synthesis time
vs
. Na
2
SeO
3
amount on Z-potential of SeNPs using
Theobroma cacao
bean shell extract. The Z-potential values on the vertical axis are presented as absolute values. Reprinted with permission by MDPI from Ref. [
81
]Fig. 4
The incubation temperature during the synthesis of SeNPs could affects their size, shape, and bioavailability. Zhang et al. [ 86 ] reported that the 80 nm SeNPs after incubation at 90 °C for 1 h resulted in aggregated into larger 110 nm particles and nanorods (290 nm × 70 nm).
The functions of selenium in the organism are mainly connected with its antioxidant properties, as it is an essential component of antioxidant enzymes such as the glutathione peroxidise group and thioredoxin reductase. They have important roles in scavenging free radicals such as reactive oxygen and nitrogen species, regulating their content produced during the biochemical reaction, Thus, they protect cells from damage and oxidative stress. SeNPs can increase selenoenzyme activities with equal efficiency but less toxicity, compare to other selenium species [ 9 , 87 ].
The antioxidant activity of different Se species is still studied [ 87 , 88 ], but the newest research has also involved selenium nanoparticles [ 56 , 57 , 63 , 67 , 76 , 82 , 83 , 90 , 91 ]. The evaluation of the antioxidant potential of SeNPs was performed using various assays, including 2,2′-diphenylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) as well as ferric reducing antioxidant power (FRAP) using standard procedures [ 92 ]. However, these methods vary in terms of antioxidant mechanism, redox potential, type of substrate, chemical conditions as well as the way of presenting the results. There is a strong recommendation to use at least two assays which measure different aspects of the antioxidant behavior due to the lack of a standard method for this purpose [ 93 ].
Menon et al. [ 56 ] compared the percentage of inhibition ratio of SeNPs synthesized using extract of ginger with that of ascorbic acid as a standard compound in a dose-dependent manner in DPPH assay. It showed moderate antioxidant activity as the IC 50 value of SeNPs was estimated at 125 µg mL −1 , while for ascorbic acid was 250 µg mL −1 . As IC 50 value for SeNPs was lower than that for ascorbic acid, so a higher concentration of this compound is needed to produce a similar effect. Alagesan and Venugopal [ 57 ] reported the IC 50 value of 14.81 µg mg −1 for similar studies using leaves extract of Withania somnifera for preparation of SeNPs within the diameter range of 45–90 nm.
Completely opposite results were obtained for Diospyros montana leaf extract-mediated synthesis of selenium nanoparticles (4–16 nm) which exhibited 61% of scavenging activity in DPPH method at the concentration of 200 μg mL −1 , compared to the same concentration of ascorbic acid (almost 100%) [ 76 ]. The IC 50 value of the nanoparticles was 0.225 μg mL −1 , while 0.138 μg mL −1 for ascorbic acid. Also in FRAP method, the synthesized SeNPs exhibited better reducing power than that of standard ascorbic acid as the EC 50 values of 0.435 μg mL −1 and 0.083 μg mL −1 were reported [ 76 ]. The size of the SeNPs may affect their antioxidant property and the nanoparticles with smaller diameters showed the stronger antioxidant activity [ 81 , 93 ].
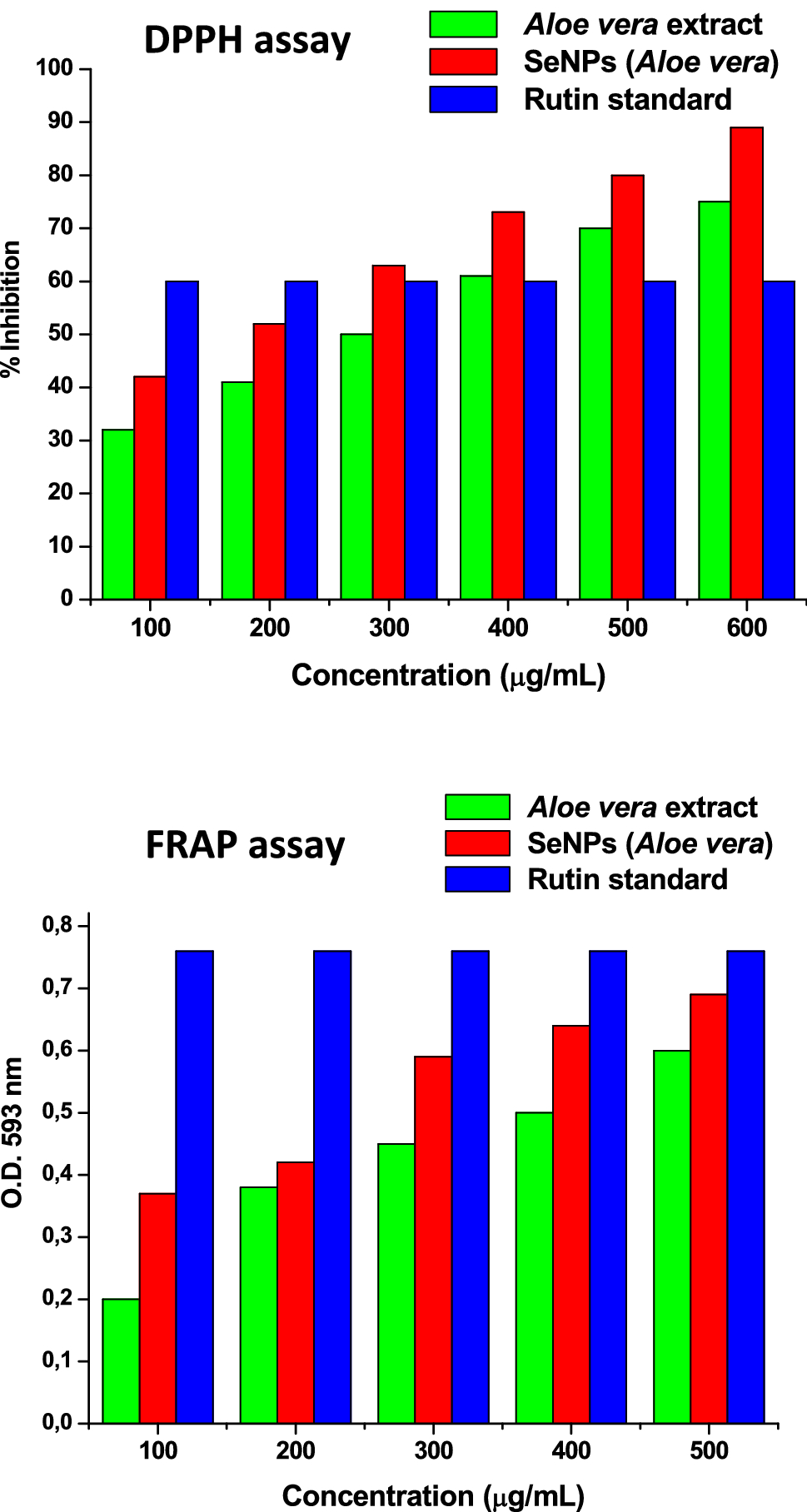
Vyas and Rana found that the average inhibition percentage in DPPH and ABTS assays were 75% and 76%, respectively, in comparison to single
Aloe vera
[
63
]
and Allium sativum
[
67
] extracts for SeNPs synthesized using these plant extracts. The obtained results were also higher than those determined for rutin as a standard compound (Fig.
5
). However, in FRAP assay rutin showed more reducing activity than both biosynthesized selenium nanoparticles.
Antioxidant properties of SeNPs synthesized by
Aloe vera
extract evaluated using DPPH and FRAP assays. Reprinted with permission by Innovare Academic Sciences Pvt. Ltd. from Ref. [
63
]Fig. 5
The values in ABTS and FRAP assays of 28.6 and 12.4 mg of Trolox equivalent per gram of nanoparticles, respectively, were reported for SeNPs synthesized with Theobroma cacao L. bean shell extract [ 82 ]. No significant differences ( p = 0.05) were observed between samples analysed at different time up to 55 days. These nanoparticles possess higher antioxidant activity than the used plant extract itself, such as for SeNPs synthesized with other natural extracts [ 63 , 67 , 94 ].
Biosynthesized SeNPs have been used in different areas such as medical, agriculture, and industrial sectors because they exhibit low toxicity and are environmentally friendly (Table
2
). In this section of review, the main applications of selenium nanoparticles synthesized using the extracts of plant materials are briefly described. More details can be found in several recent review papers [
6
,
7
,
8
,
9
–
10
,
95
,
96
,
97
,
98
–
99
].
Morphology, particle size, Se content and potential applications of SeNPs synthesized using corresponding plant extract Plant Morphology Size by SEM/TEM studies (nm) Se content (%) Medical effect/application Refs. Hawthorn spherical average 113 25.6 Antitumor activity towards HepG2 cells Chemotherapic agent for human liver cancer 52 Dried raisin nanoballs 3—18 – Potential pharmaceutical 53 Lemon plant spherical 60 -80 48 Prevention of DNA damage when cells were exposed to UVB. Diagnostic agent 54 Bugenwilla hollow and spherical 18–35 – – 55 Ginger fruit spherical 100–150 54 Antimicrobal activity against 56 Ashwagandha spherical 40–90 – Antibacterial activity against 57 Fenugreek oval 50–150 – Inhibiting the breast-cancer cells (MCF-7) growth 58 Avaram spherical 10–20 82 Antileukemia activity 59 Arauna spherical 10–80 – Minimizing arsenic-induced toxicity on human lymphocytes 60 Java tea spherical 88–141 12.6 Cytotoxic effect against L6 cell lines 61 Lavender leucas spherical 56–75 – Antibacterial and anti-fungal activity 62 spherical 7–48 25 Chemopreventive agent in cancer diagnosis 63 spherical 121 – Antimicrobial activity against spoilage fungi and pathogenic bacteria strain 64 Garlic hollow and spherical 8–52 55 Antimicrobial activity against gram-positive bacteria 65 Garlic spherical 10–80 – Direct interaction with DNA. Anticancer agent 66 Garlic spherical 7–45 55 Potential antioxidant agent 67 Garlic spherical 40–100 – Cytotoxicity against Vero cells 68 Garlic spherical 21–40 – Antibacterial agent against 69 Walnut spherical 150 – Antibacterial activity against 70 Horseshoe geranium spherical 50 – Antibacterial activity against 71 spherical 21,6 13.9 Inhibiting HepG2 cell proliferation 72 spherical 32 32.6 Possible medical applications 73 Orange peel spherical 70 – Inhibition of algal blooms 74 spherical 46–79 72.6 Insecticidal agent against mosquito vectors 75 Mountain persimmon spherical 4–16 – Antimicrobial activity 76 20–60 61.6 Antioxidant and antimicrobial agent 77 spherical 45–95 – Photocatalytic degradation of methylene blue dye 78 Drumstick spherical 23–35 44.6 Inhibition of Caco-2, HepG2 and Mcl-7 cells Photocatalytic degradation of sunset yellow dye 79 Guava spherical 8–20 – Antibacterial activity 80 Cacao bean shell spherical 1–3 – Antioxidant material 81Table 2
Several reports have confirmed the anticancer effect of SeNPs obtained by green synthesis [ 52 , 54 , 57 , 58 – 59 , 66 , 72 , 76 , 77 ]. Various mechanisms have been suggested for Se anticancer activity including cell cycle arrest, antioxidation, apoptosis, and interruption of cell signalling pathway and there is a lot of literature available on this topic [ 7 , 9 , 90 , 100 , 101 , 102 , 103 – 104 ]. SeNPs can act as a carrier for chemotherapeutic drugs delivering them to the target side, selectively accumulated inside the malignant cells, and caused the reactive oxygen species formation. The production of these reactive species was more efficient than by Se(IV) in a mouse model, generating potent therapeutic effects and promising strategy for cancer therapy [ 87 ]. Cui et al. [ 52 ] reported that selenium nanoparticles synthesized with hawthorn fruit extract induced HepG2 cells apoptosis mediated through the mitochondria pathway with the promotion of reactive oxygen species. MTT assay revealed higher growth control against A549 lung carcinoma [ 57 ] and MCF 7 human breast [ 58 ] cancer cells as well as showed good viability against L6 rat skeletal muscle cell line [ 61 ] on treatment with SeNPs with minimal toxicity. Additionally, the combination of SeNPs with doxorubicin (chemotherapy drug used to treat cancer) enhanced the cytotoxic effect in comparison to individual ones [ 53 ].
Casia auriculata leaf extract generated SeNPs exhibited cytotoxicity against Vero cells with the cytotoxic concentration required for the reduction of cell viability by 50% (CC 50 ) of 7.00 µg mL −1 [ 59 ] and for those synthesized using garlic gloves CC 50 value was higher (31.8 µg mL −1 ), while Vero cells treated with chemically synthesized nanoparticles (in the presence of polyvinylalcohol as a stabilizer) led to CC 50 of 18.8 µg mL −1 [ 68 ]. This proves that cytotoxicity of SeNPs differs depending on the technology used to synthesis of nanomaterial. Biosynthesized SeNPs exhibited also antimutagenic activity against UVB-induced DNA damage of isolated lymphocytes [ 54 ].
Selenium nanoparticles have been widely used in biomedical applications due to their antimicrobial effect to control the growth of many pathogenic bacteria and fungi [ 23 , 56 , 57 , 62 , 64 , 65 , 69 , 71 , 76 , 77 ]. The antimicrobial activity was evaluated by well diffusion method and calculated as Minimum Inhibitory Concentration (MIC), defined as the lowest concentration of the antimicrobial agent required to inhibit the growth of given microbial species, or as the diameter of inhibition zone expressed in mm. The inhibitory effect of SeNPs on bacteria and fungi may be due to their sorption on the cell wall, penetration through membrane, and entrance to kill the cell [ 16 ]. The nanoparticles with a smaller diameter can possibly diffuse the bacteria cell better than the larger ones. Sribenjarat et al. [ 69 ] found that SeNPs with size range within 21–40 nm showed a slightly higher inhibition activity than those of 41–50 mm diameter size. Boroumand et al. [ 22 ] evaluated the antibacterial activity of SeNPs on several kinds of gram-positive bacteria ( Staphylococcus aureus , Streptococcus epidermidis ) and gram-negative bacteria ( Escherichia coli , Pseudomonas aeruginosa ) and found that they exhibited dose-dependent activity against all tested bacterial strains. Particularly significant effects against the gram-positive bacteria were observed with MIC equals to 125 mg L −1 . The MIC value of SeNPs against Proteus sp . of 250 mg L −1 was reported [ 56 ]. Selenium nanoparticles synthesized using Mountain persimmon leaf extract showed antibacterial effect not only against S. aureus bacteria (zone of inhibition 8 mm) and E. coli (7 mm) but also against Aspergillus niger fungi (12 mm) [ 76 ]. Likewise, SeNPs synthesized with Aloe vera leaf extract exhibited high antimicrobial activity against pathogenic bacteria and spoilage fungi strains [ 64 ].
The surface of medical devices may be coated by SeNPs to prevent biofilm formation. They strongly inhibited the growth of S. aureus bacteria on the surface by 91% and 73% after 24 and 72 h respectively, in comparison to uncoated polycarbonate surfaces [ 105 ]. Numerous paper products such as filter paper, air purifying systems, and wrapping paper used in the food industry were also coated by precipitation of selenium nanoparticles [ 106 ]. The Se-coated paper showed 90% of S. aureus and S. epidermidi inhibition after 72 h of treatment. The results for P. aeruginosa and E. coli growth were about 57% and 84%, respectively. Thus, the antibacterial and antifungal activity of selenium nanoparticles can be used as an antimicrobial agent. Microbial infection is responsible for many diseases in humans and animals and treatment with antibiotics is not always effective due to their repeated uses resulted in resistance against pathogenic organisms.
SeNPs could be used in the drug delivery system due to easy manipulation in their size and low toxicity [ 101 , 102 – 103 ]. They are taken up by cells more efficiently than larger biomolecules. Drugs can either be integrated into the matrix of the particle or attached to its surface. Deng et al. [ 102 , 103 – 104 ] developed selenium-layered nanoparticles for oral delivery of mulberry leaf and Pueraria lobata extracts with significant hypoglycemic activities. SeNPs exhibited a slow drug release and good physiological stability in the simulated digestive fluid.
A significant number of countries in the world today are affected by groundwater arsenic contamination, which is linked with a significant increase in the risk of cancers. Prasad and Selvaraj [ 60 ] reported the protective effect of biogenic SeNP against As(III)-induced cytotoxic and genotoxic effect on human lymphocytes. The enhancement of cells viability had been noticed when arsenite-treated cells were mixed with selenium nanoparticles preventing the death of lymphocytes. Moreover, the addition of SeNPs into lymphocytes prevented As(III)-induced DNA damage. Selenium nanoparticles have also the ability to decrease the toxicity of other metal ions such Cd, Hg or Cu contained in wastewater or soil [ 110 , 111 , 112 – 113 ].
Nanoscale selenium is of great interest as an additive to fertilizers [ 6 , 96 , 114 , 115 ]. The large surface area and small size of the nanomaterials could allow for enhanced interaction and efficient uptake of selenium for crop. The inorganic selenium compounds used as fertilizers are leached along with rains, while organic compounds can be degraded after applying. SeNPs do not dissolve in water and their transition to plants occurs as a result of gradual oxidation of their surface and release as oxides [ 114 ]. Mainly, SeNPs act as stimulator and/or stressor increasing antioxidant defence system, improving vegetative growth and nutritional value of plants [ 114 , 115 , 116 , 117 – 118 ]. The eggplant and tomato growth on the soil with the Se nanoparticle addition at a concentration of 10 μg kg –1 showed almost twice the plant leaf plate surface area compared to these plants in untreated soil [ 115 ]. Zsiros et al. [ 116 ] found that SeNPs even at a concentration of 100 mg L −1 exerted no negative effect on the growth of plantlets in tobacco cultures.
Selenium nanoparticles synthesized using extract of plants are also very effective materials for photocatalytic degradation of dyes, due to their high surface to volume ratio [ 57 , 78 , 79 ]. These dyes are largely used as colorants in the common food and pharmaceutical industry. Around 60% degradation of methylene blue dye was achieved in 40 min using Fiscus benglalensis leaf extract. Its degradation followed first-order kinetics with the rate constant of 1.3 min −1 [ 77 ], while the rate constant of 0.173 min −1 was reported for photodegradation reaction of sunset yellow dye using SeNPs generated with the leaf extract of drumstick tree [ 80 ]. Thus, this is an eco-friendly approach for the treatment of wastewater containing dye effluents before being released into an aquatic system.
Selenium nanoparticles have attracted attention and their synthesis can be potentially useful in various fields. Compared to inorganic and organic selenium species, they display better bioavailability, higher biological activity and lower toxicity. The methods for SeNPs synthesis using plant extracts do not require the use of toxic chemicals, the precursors are easily accessible, inexpensive and do not need any special conditions. That way of synthesis also enables control over the size, shape and stability of nanoparticles. SeNPs synthesized in this manner exhibit particular potential in biomedical applications such as cancer therapy, targeted chemotherapy, molecular diagnosis and drug delivery system. They could also be found in suitable applications as efficient antioxidants and antibacterial agents in the food and pharmaceutical industry. A lot of work has been carried out emphasizing numerous applications of SeNPs in the technology and agriculture sectors.
The future prospects of selenium nanoparticles include the development of new fast and environment-friendly methodology for their synthesis to obtain nanomaterial with the corresponding size, shape, and properties for the desired application. It will require a better understanding of physical and chemical properties related to the specific use of SeNPs. Due to their less toxicity to normal cells, it is expected that the drugs based on selenium nanoparticles may be commercially available.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.