The astonishing ultra-small Iron Oxide nanoparticles as positive contrast agents for MR Imaging of cancerous tissues: A review
- Social Determinants of Health Research Center, School of Medicine, Lorestan University of Medical Sciences, Khorramabad, Iran.
- Finetech in Medicine Research Center, Iran University of Medical Sciences, Tehran, Iran.

Published in Issue 2024-03-30
How to Cite
Amraee, A., Sarikhani, A., & Rasaneh, S. (2024). The astonishing ultra-small Iron Oxide nanoparticles as positive contrast agents for MR Imaging of cancerous tissues: A review. International Journal of Nano Dimension, 15(2 (April 2024). https://doi.org/10.57647/j.ijnd.2024.1502.09
HTML views: 28
PDF views: 271
Abstract
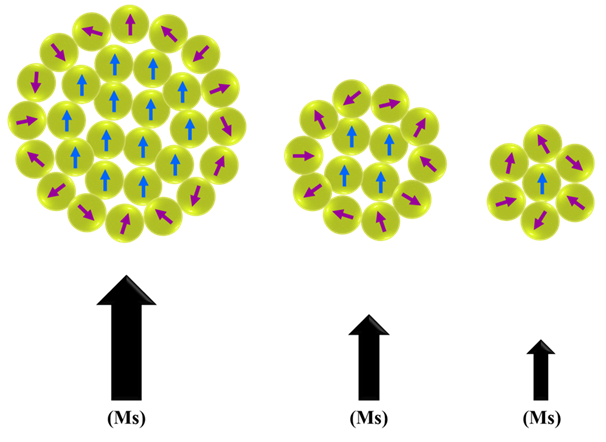
Magnetic nanoparticles, which make up a large part of nanomaterials, have the potential for clinical diagnosis and treatment due to their unique properties such as magnetic and superparamagnetic torque and the power of biological interactions at the cellular and molecular levels. The unique properties of these nanoparticles include super-saturation, superparamagnetic and magnetic susceptibility, which are derived from their inherent magnetic properties. In this review, we investigated the properties of ultra-small iron oxide nanoparticles as a positive contrast agent in MRI. As a result, ultra-small iron oxide nanoparticles have a high potential for use as a T1 weight contrast agent in the clinic. Ultra-small iron oxide nanoparticles have been widely used as a contrast agent in MR imaging. In ultra-small iron oxide nanoparticles, it has been shown that the states change from superparamagnetic to paramagnetic by decreasing in size. Therefore, it can be said that as a result of reducing the size of these nanoparticles, they change from being the T2 contrast agent to a T1 contrast agent. Unlike iron oxide nanoparticles larger than 5 nm, these nanoparticles can create a positive contrast that will facilitate detection. Also, ultra-small iron oxide nanoparticles could solve the problem of gadolinium toxicity and the high magnetic momentum of iron oxide. As a result, this nanoparticle has a high potential for use as a T1 weight contrast agent in the clinic.Keywords
- Contrast agent,
- Magnetic nanoparticles,
- MRI,
- Relaxation Times,
- T1-Weighted,
- Ultra-Small Iron Oxide

 10.57647/j.ijnd.2024.1502.09
10.57647/j.ijnd.2024.1502.09