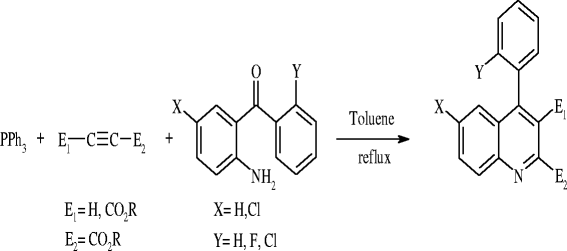
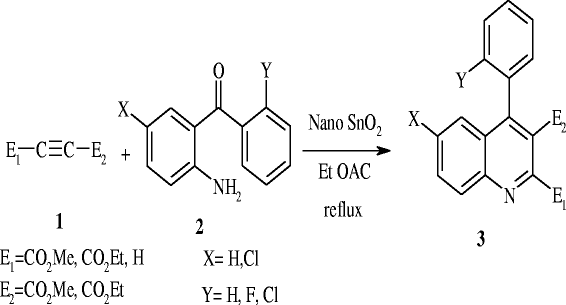
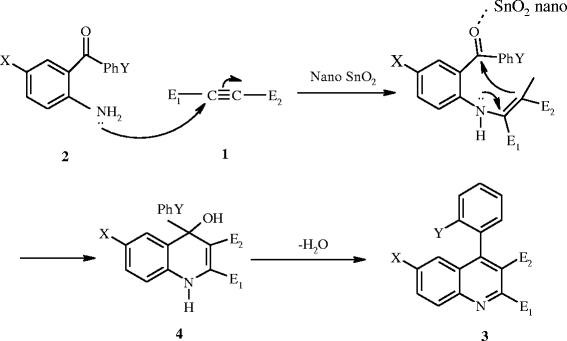
To a stirred mixture of 2-aminobenzophenone (2 mmol) and acetylenic ester (2 mmol) in EtOAc, 5 ml SnO
2
nanoparticles (5 mol%) was added. The reaction mixture was refluxed for 1 h. The progress of the reaction was monitored by TLC. After completion of the reaction, the solid product was filtered and purified by dissolving in chloroform, and the nanocatalyst was removed by a centrifuge and pure product
3a
was obtained by removing the solvent. The nanocatalyst was recovered to be reused in subsequent reactions without losing any activity. The structure of the product was confirmed by melting point, IR, and
1
H NMR spectra. The results were compared with authentic samples.
Dimethyl 4-phenyl-2,3-quinolinedicarboxylate (3a):
White powder; yield: 90%. mp 122°C to 124°C. IR (KBr) (
υ
max
/cm
−1
): 3,055 (C
sp2
-H), 2,994 (C
sp3
-H), 1,725 (CO);
1
H NMR (400 MHz, CDCl
3
):
δ
H
3.61 (s, 3
H
, OCH), 4.04 (s, 3
H
, OCH), 7.34 (dd,
3
J
HH
= 6.4 Hz,
3
J
HH
= 2.0 Hz, 2H, 2CH), 7.47 (d,
3
J
HH
= 2.0 Hz, 2H, 2CH), 7.48 (t,
3
J
HH
= 6.4 Hz, 1H, CH), 7.55 (t,
3
J
HH
= 7.7 Hz, 1H, CH), 7.62 (d,
3
J
HH
= 7.7 Hz, 1H, CH), 7.79 (ddd,
3
J
HH
= 8.5 Hz,
3
J
HH
= 7.7 Hz,
4
J
HH
= 1.4 Hz, 1H, CH), 8.3 (d,
3
J
HH
= 8.5 Hz,1 H, CH).
Diethyl 4-phenyl-2,3-quinolinedicarboxylate (3b):
White powder; yield: 87%. mp 96°C to 98°C. IR (KBr) (
υ
max
/cm
−1
): 3,032 (C
sp2
-H), 2,985 (C
sp3
-H), 1,728(CO);
1
H NMR (400 MHz, CDCl
3
):
δ
H
0.96 (t,
3
J
HH
= 7.1 Hz, H, CH
3
), 1.43 (t,
3
J
HH
= 7.1 Hz, 3H, CH
3
), 4.06 (q,
3
J
HH
= 7.1 Hz, 2H, OCH
2
), 4.50 (q,
3
J
HH
= 7.1 Hz, 2H), 7.33 (dd,
3
J
HH
= 6.5 Hz,
3
J
HH
= 2.2 Hz, 2H, 2CH), 7.45 (d,
3
J
HH
= 2.2 Hz, 2H, 2CH), 7.46 (t,
3
J
HH
= 6.5 Hz, 1H, CH), 7.53 (t,
3
J
HH
= 7.8 Hz, 1H, CH), 7.59 (d,
3
J
HH
= 7.8 Hz, 1H, CH), 7.79 (ddd,
3
J
HH
= 8.5 Hz,
3
J
HH
= 7.8 Hz,
4
J
HH
= 1.4 Hz, 1H, CH), 8.29 (d,
3
J
HH
= 8.5 Hz, 1 H, CH).
Methyl 4-phenyl-2-quinolinecarboxylate (3c):
White powder; yield: 60%. mp 101°C to 103°C. IR (KBr) (
υ
max
/cm
−1
): 3,010 (C
sp2
-H), 2,995 (C
sp3
-H), 1,725 (CO);
1
H NMR (400 MHz, CDCl
3
):
δ
H
4.07 (s, 3H, OCH
3
), 7.49 to 7.54 (m, 5H, -Ph), 7.58 (dd,
3
J
HH
= 8.3,
3
J
HH
= 7.7 Hz,
J
HH
= 1.1 Hz, 1H, CH), 7.7 (ddd,
3
J
HH
= 8.4 Hz,
3
J
HH
= 7.7 Hz,
4
J
HH
= 1.4 Hz, 1H, CH), 7.96 (d,
3
J
HH
= 8.3 Hz, 1H, CH), 8.14 (s, 1H, CH
3
), 8.36 (d,
3
J
HH
= 8.4 Hz, 1H, CH).
Ethyl 4-phenyl-2-quinolinecarboxylate (3d):
White powder; yield: 65%. mp 125°C to 127°C. IR (KBr) (
υ
max
/cm
−1
): 3,045 (C
sp2
-H), 2,985 (C
sp3
-H), 1,724 (CO).
Dimethyl 6-chloro-4-phenyl-2,3-quinolinedicarboxylate (3e):
White powder; yield: 80%. mp 158°C to 160°C. IR (KBr) (
υ
max
/cm
−1
): 3,074 (C
sp2
-H), 2,958 (C
sp3
-H), 1,732 and 1,729 (CO).
Diethyl 6-chloro-4-phenyl-2,3-quinolinedicarboxylate (3f):
White powder; yield: 90%. mp 163°C to 165°C. IR (KBr) (
υ
max
/cm
−1
): 3,075 (C
sp2
-H), 2,988 (C
sp3
-H), 1,739 and 1,724 (CO).
Methyl 6-chloro-4-phenyl-2-quinolinecarboxylate (3g):
White powder; yield: 70%. mp 177°C to 179°C. IR (KBr) (
υ
max
/cm
−1
): 3,054 (C
sp2
-H), 2,998 (C
sp3
-H), 1,719 (CO).
Ethyl 6-chloro-4-phenyl-2-quinolinecarboxylate (3h):
Yellow powder; yield: 75%. mp 171°C to 173°C. IR (KBr) (
υ
max
/cm
−1
): 3,064 (C
sp2
-H), 2,983 (C
sp3
-H), 1,729 (CO).
Dimethyl 6-chloro-4-(2-chlorophenyl)-2,3-quinolinedicarboxylate (3i):
Yellow powder; yield: 74%. mp 190°C to 192°C. IR (KBr) (
υ
max
/cm
−1
): 3,070 (C
sp2
-H), 2,985 (C
sp3
-H), 1,729 and 1,720 (CO).
Diethyl 6-chloro-4-(2-chlorophenyl)-2,3-quinolinedicarboxtlate (3j):
White powder; yield: 87%. mp 127°C to 129°C. IR (KBr) (
υ
max
/cm
−1
): 3,074 (C
sp2
-H), 2,983 (C
sp3
-H), 1,729 and 1,724 (CO).
Dimethyl 6-chloro-4-(2-fluorophenyl)-2,3-quinolinedicarboxylate (3k):
White powder; yield: 92%. mp 210°C to 212°C. IR (KBr) (
υ
max
/cm
−1
): 3,079 (C
sp2
-H), 2,953 (C
sp3
-H), 1,729 and 1,718 (CO).
Diethyl 6-chloro-4-(2-fluorophenyl)-2,3-quinolinedicarboxylate (3l):
White powder; yield: 95%. mp 135°C to 137°C. IR (KBr) (
υ
max
/cm
−1
): 3,084 (C
sp2
-H), 2,983 (C
sp3
-H), 1,729 and 1,719 (CO).