In vitro protective effects of Helichrysum cymosum (L.) D.Don (Asteraceae) on TM4 Sertoli cell
- Phytomedicine and Phytochemistry Group, Department of Biomedical Sciences, Faculty of Health and Wellness Sciences, Cape Peninsula University of Technology, Bellville 7535, South Africa
- Department of Medical Bioscience, University of the Western Cape, Bellville, Cape Town 7530, South Africa
- Department of Biological Sciences, College of Medicine and Health Sciences, Khalifa University, Abu Dhabi P.O. Box 127788, United Arab Emirates

Received: 2024-06-17
Revised: 2025-09-05
Accepted: 2025-09-14
Published in Issue 2025-09-16

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
PDF views: 53
Abstract
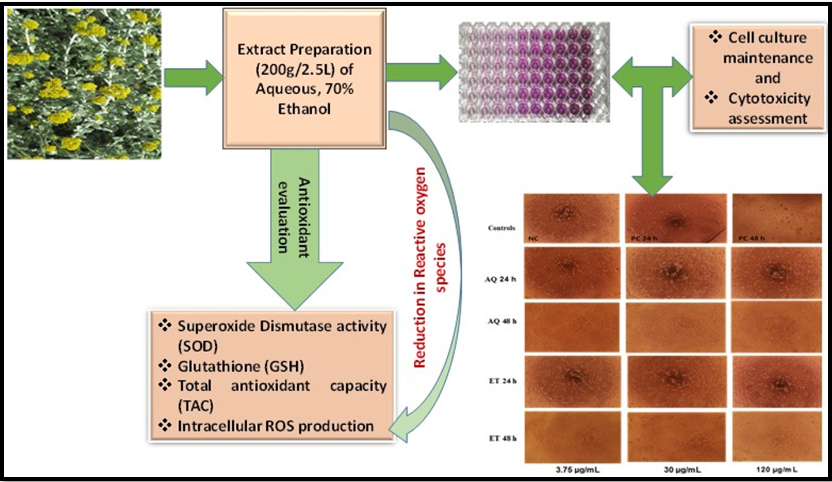
Helichrysum cymosum (L.) D.Don phytocompounds play an integral role in treating various diseases due to their bioactive properties. This study evaluated the protective effects of two H. cymosum extracts (aqueous and 70% ethanol) on cell viability, morphological changes, antioxidant enzyme activities (SOD, GSH and TAC) and ROS production on TM4 Sertoli cells. Results showed no significant effect on cell viability after 24 h but significantly decreased after 48 h (p < 0.05). Cell structure integrity was maintained at all concentrations compared to the positive control (10% DMSO). Antioxidant enzyme activities SOD, GSH and TAC had good scavenging properties, although no statistically difference was recorded. ROS production was maintained with both extracts, albeit with no significant differences. H. cymosum extracts may contain potent bioactive components such as flavonoids that work synergistically to mitigate OS-induced infertility in males. Further investigations including in vivo experiments, are recommended to validate their use in herbal supplements and drug discovery.
Keywords
- Antioxidant capacity, Asteraceae, Cytotoxicity, Glutathione, Helichrysum cymosum (L.) D.Don, Phytocompounds, Superoxide dismutase
References
- Abdillahi, H.S., Van Staden, J. (2012) South African plants and male reproductive healthcare: Conception and contraception. J. Ethnopharmacol. 143(2):475–480. DOI: https://doi.org/10.1016/j.jep.2012.06.047.
- Adewoyin, M., Ibrahim, M., Roszaman, R., Md Isa, M.L., Mat Alewi, N.A., Abdul Rafa, A.A., Anuar, M.N.N. (2017) Male infertility: The effect of natural antioxidants and phytocompounds on seminal oxidative stress. Diseases 5(1):9. DOI: https://doi.org/10.3390/diseases5010009
- Agarwal, A., Baskaran, S., Parekh, N., Cho, C.L., Henkel, R., Vij, S., Arafa, M., Selvam, M.K.P., Shah, R. (2021) Male infertility. The Lancet 397(10271):319–333. DOI: https://doi.org/10.1016/S0140-6736(20)32667-2.
- Aladejana, A.E., Bradley, G., Afolayan, A.J. (2020) In vitro evaluation of the anti-diabetic potential of Helichrysum petiolare Hilliard & BL Burtt using HepG2 (C3A) and L6 cell lines. F1000Research 9:1240. DOI: https://doi.org/10.12688/f1000research.26855.2.
- Alexander, E.C., Faruqi, D., Farquhar, R., Unadkat, A., Ng Yin, K., Hoskyns, R., Varughese, R., Howard, S.R. (2024) Gonadotropins for pubertal induction in males with hypogonadotropic hypogonadism: Systematic review and meta-analysis. Eur. J. Endocrinol. 190(1):S1–S11. DOI: https://doi.org/10.1093/ejendo/lvad166.
- Al-Temimi, A.A., Al-Mossawi, A.E.B., Al-Hilifi, S.A., Korma, S.A., Esatbeyoglu, T., Rocha, J.M., Agarwal, V. (2023) Glutathione for food and health applications with emphasis on extraction, identification, and quantification methods: A review. Metabolites 13(4):465. DOI: https://doi.org/10.3390/metabo13040465.
- Amirahmadi, A., Naderi, R., Afsharian, M.H. (2022) An investigation into the medicinal plants of Semnan province with taxonomic and therapeutic aspects. Trends Phytochem. Res. 6(4):312–338. DOI: https://doi.org/10.30495/tpr.2022.1970840.1282.
- Aoyama, K. (2021) Glutathione in the brain. Int. J. Mol. Sci. 22(9):5010. DOI: https://doi.org/10.3390/ijms22095010.
- Averill-Bates, D.A. (2023) The Antioxidant Glutathione. In Vitamins and Hormones, Academic Press, Vol.121, pp. 109–141. DOI: https://doi.org/10.1016/bs.vh.2022.09.002.
- Bajaj, S., Singh, S., Sharma, P. (2024) Role of Antioxidants in Neutralizing Oxidative Stress. In Nutraceutical Fruits and Foods for Neurodegenerative Disorders, Academic Press, pp. 353–378. DOI: https://doi.org/10.1016/B978-0-443-18951-7.00020-7.
- Banerjee, B., Chakraborty, S., Chakraborty, P., Ghosh, D., Jana, K. (2019) Protective effect of resveratrol on benzo (a) pyrene induced dysfunctions of steroidogenesis and steroidogenic acute regulatory gene expression in Leydig cells. Front. Endocrinol. 10:272. DOI: https://doi.org/10.3389/fendo.2019.00272.
- Bansal, A.K., Bilaspuri, G.S. (2011) Impacts of oxidative stress and antioxidants on semen functions. Vet. Med. Int. 2011(1):686137. DOI: https://doi.org/10.4061/2011/686137.
- Bara, N., Kaul, G. (2018) Enhanced steroidogenic and altered antioxidant response by ZnO nanoparticles in mouse testis Leydig cells. Toxicol. Ind. Health 34(8):571–588. DOI: https://doi.org/10.1177/0748233718774220.
- Brehm, E., Flaws, J.A. (2019) Transgenerational effects of endocrine-disrupting chemicals on male and female reproduction. Endocrinology 160:1421–1435. DOI: https://doi.org/10.1210/en.2019-00034.
- Chang, L., Wang, J., She, R., Ma, L., Wu, Q. (2017) In vitro toxicity evaluation of melamine on mouse TM4 Sertoli cells. Environ. Toxicol. Pharmacol. 50:111–118. DOI: https://doi.org/10.1016/j.etap.2017.01.009.
- Chang, M.S., Kim, W.N., Yang, W.M., Kim, H.Y., Oh, J.H., Park, S.K. (2008) Cytoprotective effects of Morinda officinalis against hydrogen peroxide‐induced oxidative stress in Leydig TM3 cells. Asian J. Androl. 10(4):667–674. DOI: https://doi.org/10.1111/j.1745-7262.2008.00414.x.
- Chua, M.E., Escusa, K.G., Luna, S., Tapia, L.C., Dofitas, B., Morales, M. (2013) Revisiting oestrogen antagonists (clomiphene or tamoxifen) as medical empiric therapy for idiopathic male infertility: A meta‐analysis. Andrology 1(5):749–757. DOI: https://doi.org/10.1111/j.2047-2927.2013.00107.x.
- Di Giacomo, C., Malfa, G.A., Tomasello, B., Bianchi, S., Acquaviva, R. (2023) Natural compounds and glutathione: Beyond mere antioxidants. Antioxidants 12(7):1445. DOI: https://doi.org/10.3390/antiox12071445.
- Dike, C.C., Ezeokafor, E.N., Oparaji, K.C., Amadi, C.S., Chukwuma, C.C., Igbokwe, V.U. (2023) The impact of Annona muricata and metformin on semen quality and hormonal profile in arsenic trioxide-induced testicular dysfunction in male Wistar rat. Magna Scientia Adv. Res. Rev. 8(1):001–018. DOI: https://doi.org/10.30574/msarr.2023.8.1.0053.
- Dutta, S., Sengupta, P. (2018) Medicinal herbs in the management of male infertility. J. Preg. Reprod. 2(1):1–6. DOI: https://doi.org/10.15761/JPR.1000128.
- Ellman, G.L. (1959) Tissue sulfhydryl groups. Arch. Biochem. Biophys. 82(1):70–77. DOI: https://doi.org/10.1177/0748233718774220.
- Ertik, O., Magaji, U.F., Sacan, O., Yanardag, R. (2023) Effect of Moringa oleifera leaf extract on valproate-induced oxidative damage in muscle. Drug Chem. Toxicol. 46(6):1212–1222. DOI: https://doi.org/10.1080/01480545.2022.2144876.
- Falang, D.C., Pierre, B. (2022) In vitro and in vivo antioxidant properties of p-coumaroyl alphitolic acid extracted from fruit of Zizuphus mauritiana. Ann Agric Sci. 67:196-203. DOI: https://doi.org/10.33425/2641-4295.1075.
- Fink, J., Ide, H., Horie, S. (2024) Management of male fertility in hypogonadal patients on testosterone replacement therapy. Medicina 60(2):275. DOI: https://doi.org/10.3390/medicina60020275.
- Forman, H.J., Zhang, H., Rinna, A. (2009) Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Asp. Med. 30(1-2):1–12. DOI: https://doi.org/10.1016/j.mam.2008.08.006.
- Georgiou-Siafis, S.K., Tsiftsoglou, A.S. (2023) The key role of GSH in keeping the redox balance in mammalian cells: mechanisms and significance of GSH in detoxification via formation of conjugates. Antioxidants 12(11):1953. DOI: https://doi.org/10.3390/antiox12111953.
- Giovanelli, S., De Leo, M., Cervelli, C., Ruffoni, B., Ciccarelli, D., Pistelli, L. (2018) Essential oil composition and volatile profile of seven Helichrysum species grown in Italy. Chem. Biodiverse. 15(5):1700545. DOI: https://doi.org/10.1002/cbdv.201700545.
- Greifová, H., Jambor, T., Tokárová, K., Knížatová, N., Lukáč, N. (2022) In vitro effect of resveratrol supplementation on oxidative balance and intercellular communication of leydig cells subjected to induced oxidative stress. Folia Biol. (Kraków) 70(1):19–32. DOI: https://doi.org/10.3409/fb_70-1.03.
- Ho, C.C., Tan, H.M. (2011) Rise of herbal and traditional medicine in erectile dysfunction management. Curr. Urol. Rep. 12:.470–478. DOI: https://doi.org/10.1007/s11934-011-0217-x.
- Ighodaro, O.M., Akinloye, O.A. (2018) First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. med. 54(4):287–293. DOI: https://doi.org/10.1016/j.ajme.2017.09.001.
- Islam, M.N., Rauf, A., Fahad, F.I., Emran, T.B., Mitra, S., Olatunde, A., Shariati, M.A., Rebezov, M., Rengasamy, K.R., Mubarak, M.S. (2022) Superoxide dismutase: An updated review on its health benefits and industrial applications. Crit. Rev. Food Sci. Nutr. 62(26):7282–7300. DOI: https://doi.org/10.1080/10408398.2021.1913400.
- Jadalla, B.M., Moser, J.J., Sharma, R., Etsassala, N.G., Egieyeh, S.A., Badmus, J.A., Marnewick, J.L., Beukes, D., Cupido, C.N., Hussein, A. A. (2022) In vitro alpha-glucosidase, alpha-amylase inhibitory activities and antioxidant capacity of Helichrysum cymosum and Helichrysum pandurifolium Schrank constituents. Separations 9:190. DOI: https://doi.org/10.3390/separations9080190.
- Jambor, T., Zajickova, T., Arvay, J., Ivanisova, E., Tirdilova, I., Knizatova, N., Greifova, H., Kovacik, A., Galova, E., Lukac, N. (2022) Exceptional properties of Lepidium sativum L. extract and its impact on cell viability, ROS production, steroidogenesis, and intracellular communication in mice Leydig cells in vitro. Molecules 27(16):5127. DOI: https://doi.org/10.3390/molecules27165127.
- Kaltsas, A. (2023) Oxidative stress and male infertility: The protective role of antioxidants. Medicina 59(10):1769. DOI: https://doi.org/10.3390/medicina59101769.
- Kazeminia, M., Mehrabi, A., Mahmoudi, R. (2022) Chemical composition, biological activities, and nutritional application of Asteraceae family herbs: A systematic review. Trends Phytochem. Res. 6(3):187-213. DOI: https://doi.org/10.30495/tpr.2022.1954612.1248.
- Koshevoy, V., Naumenko, S., Skliarov, P., Fedorenko, S., Kostyshyn, L. (2021) Male infertility: Pathogenetic significance of oxidative stress and antioxidant defence. 24(6):107-116. DOI: https://doi.org/10.48077/scihor.
- Kumbhare, S.D., Ukey, S.S., Gogle, D.P. (2023) Antioxidant activity of Flemingia praecox and Mucuna pruriens and their implications for male fertility improvement. Sci. Rep. 13(1):19360. DOI: https://doi.org/10.1038/s41598-023-46705-9.
- Liew, F.F., Dutta, S., Sengupta, P. (2024) Fertility treatment-induced oxidative stress and reproductive disorders. J. Integr. Sci. Technol. 12(3):756–756. DOI: https://doi.org/10.62110/sciencein.jist.2024.v12.756.
- Lourens, A.C.U., Viljoen, A.M., Van Heerden, F.R. (2008) South African Helichrysum species: A review of the traditional uses, biological activity and phytochemistry. J. Ethnopharmacol. 119(3):630–652. DOI: https://doi.org/10.1016/j.jep.2008.06.011.
- Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J. (1951) Protein measurement with the Folin phenol reagent. J. Boil. Chem. 193(1):265–275. DOI: https://doi.org/10.1016/S0021-9258(19)52451-6.
- Malviya, N., Malviya, S., Jain, S., Vyas, S. (2016) A review of the potential of medicinal plants in the management and treatment of male sexual dysfunction. Andrologia 48(8):880–893. DOI: https://doi.org/10.1111/and.12677.
- Mansour, H.A.E.H. (2023) Infertility diagnosis and management. Beni-Suef Uni. J. Basic Appl. Sci. 12(1):81. DOI: https://doi.org/10.1186/s43088-023-00416-2.
- Marklund, S., Marklund, G. (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 47(3):469–474. DOI: https://doi.org/10.1111/j.1432-1033.1974.tb03714.x.
- Maroyi, A. (2019) Helichrysum cymosum (L.) D. Don (Asteraceae): Medicinal uses, chemistry, and biological activities. Asian J. Pharm. Clin. Res. 12:19–26. DOI: http://dx.doi.org/10.22159/ajpcr.2019.v12i7.33771.
- Matanzima, Y. (2014) Quantitative and Qualitative Optimization of Antimicrobial Bioactive Constituents of Helichrysum cymosum Using Hydroponics Technology. Cape Peninsula University of Technology, Thesis (MTech (Horticulture)). DOI: https://etd.cput.ac.za/handle/20.500.11838/849.
- Monsees, T.K., Franz, M., Gebhardt, S., Winterstein, U., Schill, W.B., Hayatpour, J. (2000) Sertoli cells as a target for reproductive hazards. Andrologia 32(4‐5):239–246. DOI: https://doi.org/10.1046/j.1439-0272.2000.00391.x.
- More, G.K., Makola, R.T. (2020) In-vitro analysis of free radical scavenging activities and suppression of LPS- induced ROS production in macrophage cells by Solanum sisymbriifolium extracts. Sci. Rep.10 (1):6493. DOI: https://doi.org/10.1038/s41598-020-63491-w.
- Nantia, E.A., Moundipa, P.F., Monsees, T.K., Carreau, S. (2009) Medicinal plants as potential male anti-infertility agents: a review. Andrologie 19(3):148–158. DOI: https://doi.org/10.1007/s12610-009-0030-2.
- Nkemzi, A.Q., Ekpo, O.E. (2022) Reproductive, antioxidant, anti-inflammatory, antimicrobial, protective and antidiabetic activities of Helichrysum Mill. species. Plant Sci. Today 9(4):794-801. DOI: https://doi.org/10.14719/pst.1484.
- Nkemzi, A.Q., Okaiyeto, K., Kerebba, N., Rautenbach, F., Oyenihi, O., Ekpo, O.E., Oguntibeju, O.O. (2024) In vitro hypoglycemic, antioxidant, anti-inflammatory activities and phytochemical profiling, of aqueous and ethanol extracts of Helichrysum cymosum. Phytomed. Plus 100639. DOI: https://doi.org/10.1016/j.phyplu.2024.100639.
- Novelli, A., Bianchetti, A. (2022) Glutathione: Pharmacological aspects and implications for clinical use. Geriatric Care 8(2). DOI: https://doi.org/10.4081/gc.2022.10390.
- Olabiyi, F.A., Aboua, Y.G., Popoola, O.K., Monsees, T.K., Oguntibeju, O.O. (2020) Evaluation of antioxidant, antityrosinase activities and cytotoxic effects of Phyllanthus amarus extracts. Nat. Prod. J. 10(2):130–138. DOI: https://doi.org/10.2174/2210315509666190405100745.
- Panner Selvam, M.K., Baskaran, S., Tannenbaum, J., Greenberg, J., Shalaby, H.Y., Hellstrom, W.J., Sikka, S.C. (2023) Clomiphene citrate in the management of infertility in oligospermic obese men with hypogonadism: Retrospective pilot study. Medicina 59(11):1902. DOI: https://doi.org/10.3390/medicina59111902.
- Petricca, S., Carnicelli, V., Luzi, C., Cinque, B., Celenza, G., Iorio, R. (2023) Oxidative stress, cytotoxic and inflammatory effects of azoles combinatorial mixtures in sertoli Sertoli TM4 cells. Antioxidants 12(6):1142. DOI: https://doi.org/10.3390/antiox12061142.
- Philander, L.A. (2011) An ethnobotany of Western Cape Rasta bush medicine. J. Ethnopharmacol. 138(2):578–94. DOI: https://doi.org/10.1016/j.jep.2011.10.004.
- Polycarp, T.N., Obukowho, E.B., Yusoff, S.M. (2016) Changes in haematological param eters and oxidative stress response of goats subjected to road transport stress in a hot humid tropical environment. Comp. Clin. Path. 25:.285–293. DOI: https://doi.org/10.1007/s00580-015-2179-8.
- Ramli, I., Posadino, A.M., Zerizer, S., Spissu, Y., Barberis, A., Djeghim, H., Azara, E., Bensouici, C., Kabouche, Z., Rebbas, K., D'hallewin, G. (2023) Low concentrations of Ambrosia maritima L. phenolic extract protect endothelial cells from oxidative cell death induced by H2O2 and sera from Crohn's disease patients. J. Ethnopharmacol. 300:115722. DOI: https://doi.org/10.1016/j.jep.2022.115722.
- Savira, M., Sari, D.K., Machrina, Y., Widjaja, S.S., Unitly, A.J.A., Ilyas, S., Siregar, J., Pandia, P., Rusda, M., Amin, M.M. (2023) Effect of garlic ethanol extract administration on gluthatione levels to prevent oxidative stress in smoker rat model. Med. Arch. 77(6): 418. DOI: https://doi.org/10.5455/medarh.2023.77.418-421.
- Semenya, S.S., Potgieter, M.J. (2013) Ethnobotanical survey of medicinal plants used by Bapedi traditional healers to treat erectile dysfunction in the Limpopo Province, South Africa. J. Med. Plants Res. 7(7):349–357. DOI: https://doi.org/10.5897/JMPR12.499.
- Siddique, R., Mehmood, M.H., Shehzad, M.A. (2024) Current Antioxidant Medicinal Regime and Treatments Used to Alleviate Oxidative Stress in Infertility Issues. In Fundamental Principles of Oxidative Stress in Metabolism and Reproduction, Academic Press, pp. 287–315. DOI: https://doi.org/10.1016/B978-0-443-18807-7.00018-1.
- Sylvest, R., Fürbringer, J.K., Pinborg, A., Koert, E., Bogstad, J., Loessl, K., Praetorius, L., Schmidt, L. (2018) Low semen quality and experiences of masculinity and family building. Acta Obstet. Gynecol. Scand. 97(6):727–733. DOI: https://doi.org/10.1111/aogs.13298.
- Takalani, N.B., Monageng, E.M., Mohlala, K., Monsees, T.K., Henkel, R., Opuwari, C.S. (2023) Role of oxidative stress in male infertility. Reprod. Fertil. 4(3). DOI: https://doi.org/10.1530/RAF-23-0024.
- Van Haaften, R.I., Haenen, G.R., Evelo, C.T., Bast, A. (2003) Effect of vitamin E on glutathione-dependent enzymes. Drug Metab. Rev. 35(2-3):215–253. DOI: https://doi.org/10.1081/DMR-120024086.
- Van Vuuren, S.F., Viljoen, A.M., van Zyl, R.L., Van Heerden, F.R., Başer, K.H.C. (2006) The antimicrobial, antimalarial and toxicity profiles of helihumulone, leaf essential oil and extracts of Helichrysum cymosum (L.) D. Don subsp. cymosum. S. Afr. J. Bot. 72(2):287–290. DOI: https://doi.org/10.1016/j.sajb.2005.07.007.
- Watcho, P., Mpeck, I.R., Defo, P.B.D., Wankeu-Nya, M., Ngadjui, E., Fozin, G.R.B., Kamtchouing, P., Kamanyi, A. (2019) Cyclophosphamide-induced reproductive toxicity: Beneficial effects of Helichrysum odoratissimum (Asteraceae) in male Wistar rats. J. Integr. Med. 17(5):366–73. DOI: https://doi.org/10.1016/j.joim.2019.07.002.
- Zheng, M., Liu, Y., Zhang, G., Yang, Z., Xu, W., Chen, Q. (2023). The applications and mechanisms of superoxide dismutase in medicine, food, and cosmetics. Antioxidants 12(9):1675. DOI: https://doi.org/10.3390/antiox12091675.

 10.57647/tpr.2025.0903.14
10.57647/tpr.2025.0903.14